(A) The literature reports that when alcohol 1 (see structure below) is treated with thionyl chloride in diethyl ether, the single product is (35,5S)-3- chloro-5-methylcyclohexene (2) (no enantiomer of diastereomer of 2 is formed). Explain by writing a detailed mechanism. Look at the solution to LC-39 (B) Using a mechanism, predict what single stereoisomer of 2 should form when pyridine is substituted for the diethyl ether. (C) Finally, predict mechanistically what additional stereoisomers of 3- chloro-5-methylcyclohexene should form when 1 is treated with Lucas reagent (ZnCl₂/H₂0 CI).
(A) The literature reports that when alcohol 1 (see structure below) is treated with thionyl chloride in diethyl ether, the single product is (35,5S)-3- chloro-5-methylcyclohexene (2) (no enantiomer of diastereomer of 2 is formed). Explain by writing a detailed mechanism. Look at the solution to LC-39 (B) Using a mechanism, predict what single stereoisomer of 2 should form when pyridine is substituted for the diethyl ether. (C) Finally, predict mechanistically what additional stereoisomers of 3- chloro-5-methylcyclohexene should form when 1 is treated with Lucas reagent (ZnCl₂/H₂0 CI).
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter22: Reactions Of Benzene And Its Derivatives
Section: Chapter Questions
Problem 22.59P
Related questions
Concept explainers
Organomagnesium compounds
The interaction of alkyl halide with Mg metal in a suitable ether solvent is the common method for the synthesis of the Grignard reagent.
Hydrolysis Grignard Reactions and Reduction
Organomagnesium halides are Grignard reagents. Francois Auguste Victor Grignard, a French chemist who received the Nobel Prize in Chemistry in 1912, created these highly useful reagents.
Question
Can you make sure to include what the reactions follow SN1, SN2, SNi etc
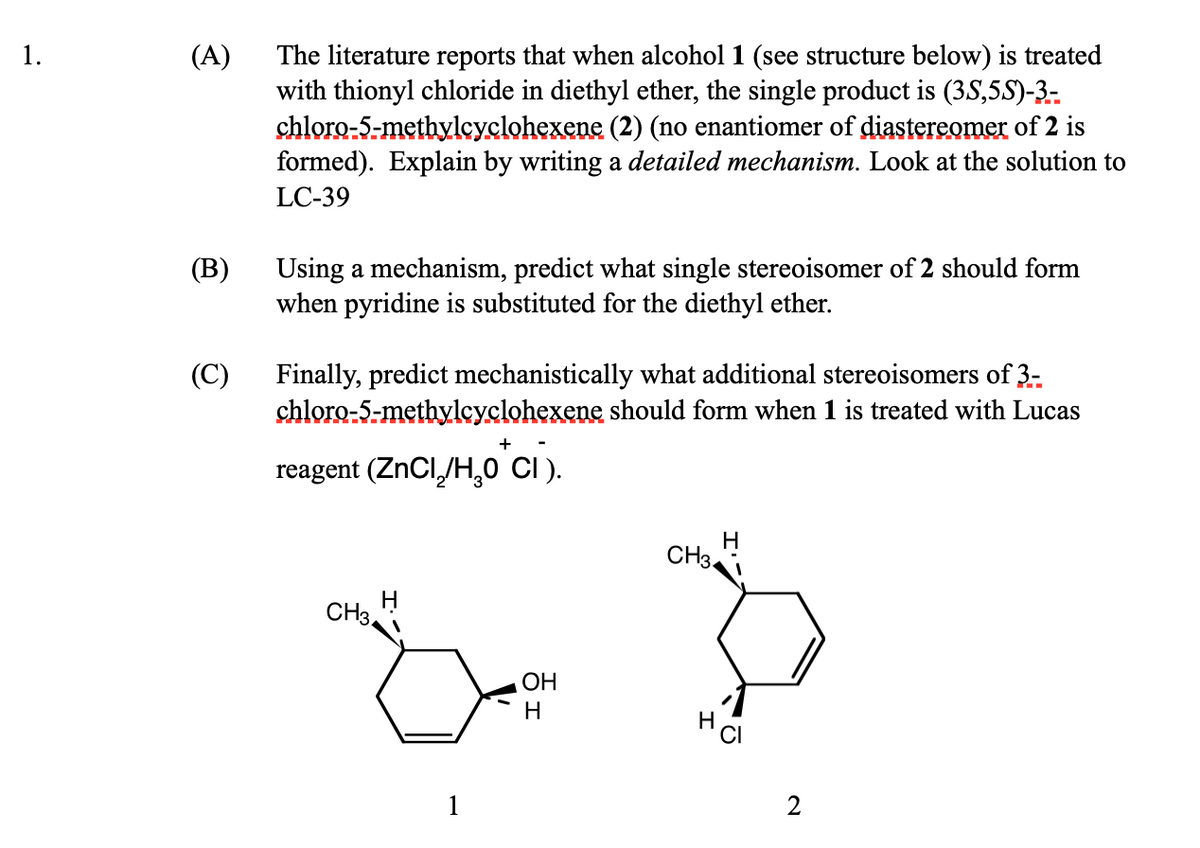
Transcribed Image Text:1.
(A)
(B)
(C)
The literature reports that when alcohol 1 (see structure below) is treated
with thionyl chloride in diethyl ether, the single product is (35,5S)-3-
chloro-5-methylcyclohexene (2) (no enantiomer of diastereomer of 2 is
formed). Explain by writing a detailed mechanism. Look at the solution to
LC-39
Using a mechanism, predict what single stereoisomer of 2 should form
when pyridine is substituted for the diethyl ether.
Finally, predict mechanistically what additional stereoisomers of 3-
chloro-5-methylcyclohexene should form when 1 is treated with Lucas
reagent (ZnCl₂/H₂O*CI).
CH3
OH
H
CH3
H
2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning