Chapter20: The Representative Elements
Section: Chapter Questions
Problem 6RQ
Related questions
Question
4
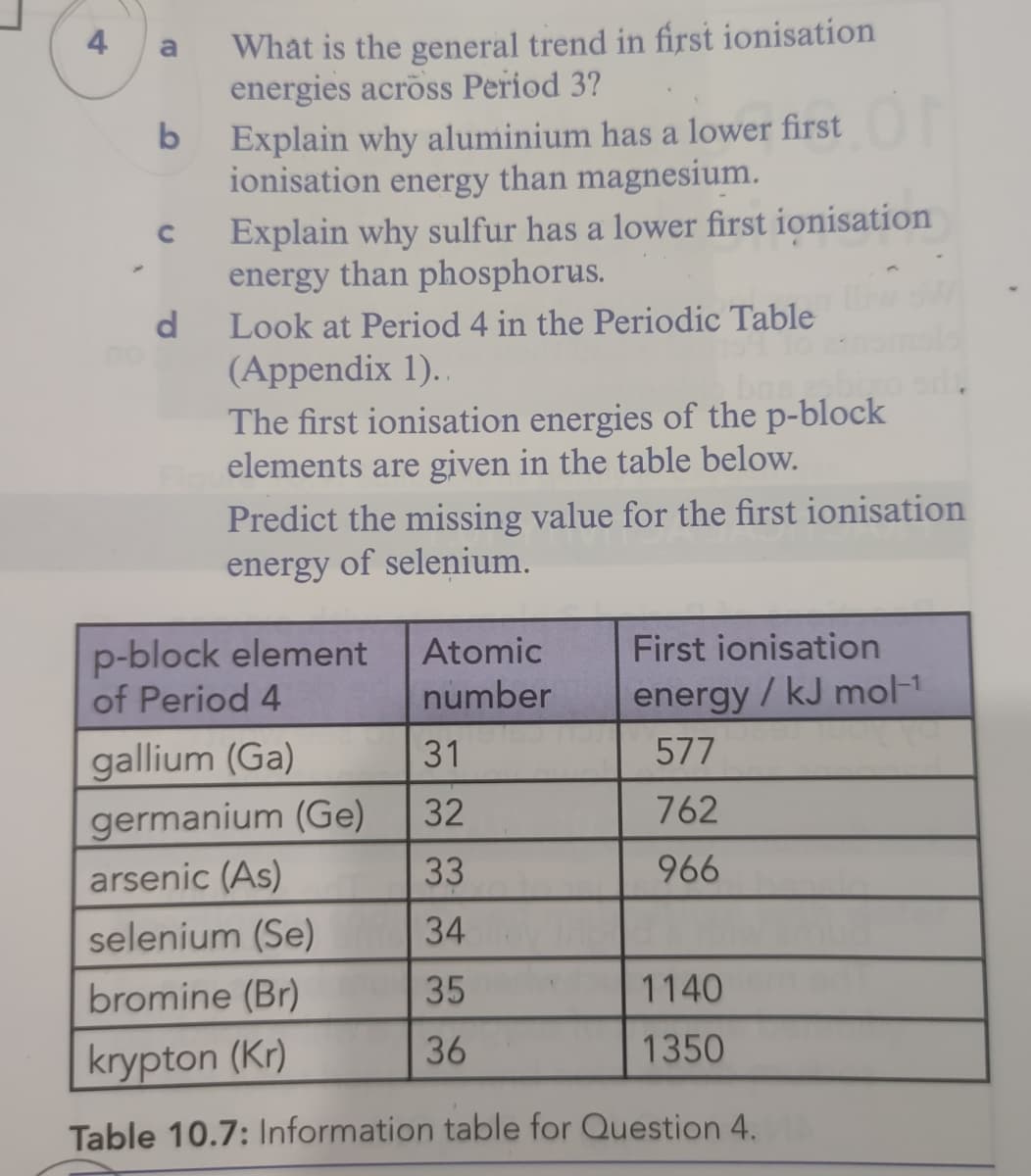
Transcribed Image Text:What is the general trend in first ionisation
energies across Period 3?
4
a
Explain why aluminium has a lower first
ionisation energy than magnesium.
Explain why sulfur has a lower first ionisation
energy than phosphorus.
Look at Period 4 in the Periodic Table
(Appendix 1)..
The first ionisation energies of the p-block
elements are given in the table below.
Predict the missing value for the first ionisation
energy of selenium.
d
bas
p-block element
of Period 4
Atomic
First ionisation
number
energy/ kJ mol-1
gallium (Ga)
31
577
germanium (Ge)
32
762
arsenic (As)
33
966
selenium (Se)
34
bromine (Br)
35
1140
36
1350
krypton (Kr)
Table 10.7: Information table for Question 4.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning