Q: How many grams of bromine are required to completely react with 22.1g of titanium
A: Titanium on heating with bromine to form titanium tetra bromide. The reaction given below
Q: b. How much oxygen is
A:
Q: heat
A:
Q: Answer the following questions. _____1. Which of the ff you can use for mixing liquids? a) test tube...
A: 1) The tool which is used for mixing liquids is stirring rod. Therefore, the correct answer is optio...
Q: Calculate the fluid intake in milliliters (mL) for the following lunch. (Assume a glass holds 8 oz.)...
A: 1 glass = 8 Oz 1 Oz = 29.57 mL
Q: e. Cuts) + S,(5)→ 3. Write the products for each decomposition reaction. Balance the chemical equati...
A: Decomposition reactions are those in which chemical species break up into simpler parts.
Q: How many liters of a 3.70 M K2SO4 solution are needed to provide 91.8g of K2SO4 (molar mass 174.01...
A: Definition: Molarity is a concentration term for a solution. The molarity of a given solution is def...
Q: m mass c
A:
Q: Which is more acidic, p-methylphenol or phenol? why? Why is water more acidic than ethanol but sligh...
A: Which is more acidic, p-methylphenol or phenol? why? Why is water more acidic than ethanol but sligh...
Q: 7. Considering the bond formed for MgO and NaCl, which among the two is easier to use to conduct ele...
A: The small size of cation or anion forms easily bond with water.
Q: : Calculate the molecular weight of urea, CO (NH2)2 Show your pertinent solution.
A: Molecular mass of any compound is equal to the sum of atomic masses constituent atoms in the compoun...
Q: Which of the following compounds are aromatic? circle them. WEN
A:
Q: A 5.0M solution of acetic acid (pKa=4.75) has a pH of 6.75. The ratio of acid to conjugate base is?
A: The pH of a buffer is calculated by Henderson-Hasselbach equation. pH=pKa+log[conjugate base][acid]w...
Q: Complete these following reactions: Ex. #1: Ex. #3: Li CI (2 equiv) Mg Et,0 sometimes written " Li O...
A: These are some of the examples of the formation of alkyl compounds attached to active metal centres ...
Q: hydroxide
A:
Q: aw the major products for the ff. reactions: МСРВА --> cat. OsO4 NMO + HBr –- H20 + Br2 --> 4-
A:
Q: Draw an expanded structural formula of a hydrocarbon that contains sp³, sp?, and sp hybridized carbo...
A:
Q: (Q90) A second order reaction has an initial concentration of 1.46 M. If the half-life of the reacti...
A: Given: Initial concentration = 1.46 M The half-life of the reaction = 32.8 minutes
Q: 4. Draw the direction vector in the unit cell for the tor the following cubic directions. Identify t...
A:
Q: Arrange the following compounds from most soluble to least soluble in water. I. methanol II. n-bu...
A: A solubility of alcohols in water mainly depends on hydrogen bonding between water. As hydrogen bond...
Q: Draw the Lewis structures with the correct molecular geometry using VSEPR Theory for C2H2.
A:
Q: Name the different oils that can be used in making soaps. Describe the physical appearance of oil an...
A: Soap are the sodium or potassium salts of fatty carboxylic acids which results in the chemical react...
Q: 9. Distance of atoms from each other (only an example): MgO= 2 nm and the bond length/distance are 1...
A:
Q: What is the hydronium ion concentration in an aqueous hydrobromic acid solution that has a pOH of 12...
A: Hydrogen ion concentration is an important parameter of an aqueous solution. The concentration of th...
Q: Classify each of the following compounds. The possible classifications are as follows: alcohol keton...
A: The possible classification for the given compound = ?
Q: moles
A:
Q: Henry's Law describes the solubility of a gas in a liquid. Rank the following gases in order of incr...
A:
Q: 3. Write the mechanism for the reactions below. Pd cat, base Br heat Et0OC Et0OC- Et0OC Et0OC
A: The mentioned conversation involves two major steps such as coupling process mediated by palladium f...
Q: Identify the electrophilic or nucleophilic sites in the chemical species shown below. Clearly label ...
A: Nucleophile is nucleus loving and has a region of high electron density. Electrophile is electron l...
Q: Draw an expanded structural formula of a hydrocarbon that contains sp, sp?, and sp hybridized carbon...
A:
Q: 3.47 Draw an energy diagram for a two-step reaction that releases energy and whose first step is fas...
A: As during the reaction, the energy is released. Thus the reaction is exothermic reaction. Transitio...
Q: If 2.5 mols of oxygen gas was available to react with excess hydrogen, how many grams of water would...
A: The balance chemical reaction : 2H2 + O2 ---> 2H2O Number of moles O2 = 2.5 moles In presence...
Q: 1. An isothermal process is performed on 10 moles of an ideal gas. The gas temperature is 400 K. The...
A:
Q: considerer the following reaction: NiCl2 + 6NH3 + 2 KClO4 --> Ni(NH3)6(ClO4)2 + 2KCl if you mix 1....
A:
Q: Which of the following requires minimum energy for electronic transition among atomic orbitals of H-...
A:
Q: .CI AICI;
A: It is an example of friedel craft alkylation.
Q: What mass in grams of TNT can be made from 598 g C7H8?
A:
Q: How many electrons are there in an atom of 1?
A: The element can be represented as zXA or zAX Where z= atomic number it indicates number of protons ...
Q: The line formula for a branched alkane is shown below: a. What is the molecular formula of this comp...
A:
Q: 1. A) Calculate the volume of Nickel atoms in an FCC unit cell, given that the atomic radius of Ni i...
A: Number of atoms in FCC, Z = 4 Atomic Radius = 163 pm or 1.63 × 10-8 cm
Q: Write the electron configurations for the elements below. Use the following format with spaces betwe...
A: Electronic configuration is the distribution of electrons in an atom or molecule.
Q: Question 8 OH Но НО OH Но HO OH OH OH HOʻ Но What are the monosachharides that make up this trisacch...
A:
Q: OH но но OH What is the Fischer formula of this monosaccharide? H-C-OH HO-C-H HO-C-H H-C-OH CH;OH O ...
A: A Fischer projection formula is a convention used to depict a stereoformula in two dimension without...
Q: poly-(3-hexylthiophene
A: In this question we have to explain separation scheme for the purifiy of poly-(3-hexylthiophene).
Q: Which is the is the correct structure for a-D-galactopyranose? OH OH HO HO O HOCH2 0. OH OH OH OH OH...
A: α-D-galactose is d-Galactopyranose having alpha-configuration at the anomeric centre. It has a role ...
Q: Counting Atoms Fill in the table Pb C CI Mass (g) 0.543 1.834 Molar Mass g/mol) 0.685 Moles Total Nu...
A: We can solve these questions by knowing some of the basic mole concepts, like conversion of number o...
Q: OH HO + Catechol Oxidase 1/202 H2O + Catechol Benzoquinone
A: Catechol oxidase is a copper containing oxidizing enzyme which is used for he oxidation of catechol ...
Q: Below are some objectives. Check if each objective possesses the SMART characteristics. If not, plea...
A: In the given questions some objectives are given. 1. Position of an element in periodic table gives ...
Q: solution is prepared by mixing 7.05 grams of benzene (C6H6) in water to create a solution total volu...
A: Mass of benzene = 7.05 g Volume of solution = 100 mL = 0.1 L Molar mass of benzene= 78.11 g/mol
Q: D D m) 3-hexyne to each of the folowing deuterated alkenes: CH;CH, CH, CH3 CH3 CH2 D and D CH, CH3 n...
A: Lindlar catalyst - It is heterogeneous catalyst that consist of pd deposited on calcium carbonate...
Which rxn/s is/are unfavorable? Briefly explain why.
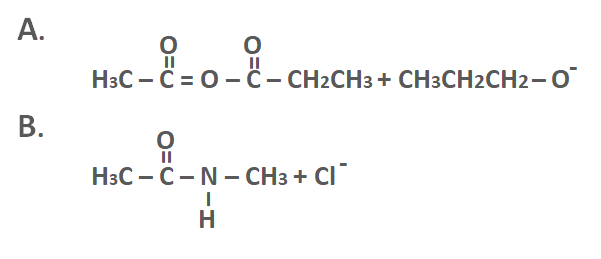
Step by step
Solved in 2 steps with 2 images

- Methanogenic bacteria convert acetic acid (CH3COOH) to CO2(g) and CH4(g). ΔH°f, (kJ/mol) ΔG°f, (kJ/mol) CH3COOH -484.5 -389.9 CO2(g) -393.5 -394.4 CH4(g) -74.8 -50.8 Calculate ΔH°rxn. ΔH°rxn = ____ kJ Calculate ΔG°rxn. ΔG°rxn = ___kJLithocholic acid is an A–B cis steroid found in human bile. Draw lithocholic acid showing chair conformations, as in Figure 27-11, and tell whether the hydroxyl group at C3 is axial or equatorial.Rank the following groups in order of decreasing priority. −CH2CH3, −CH3, −H, −CH(CH3)2
- Most naturally occurring amino acids have chiral centers (the asymmetric a carbon atoms)that are named (S) by the Cahn–Ingold–Prelog convention (Section 5-3). The commonnaturally occurring form of cysteine has a chiral center that is named (R), however.(a) What is the relationship between (R)-cysteine and (S)-alanine? Do they have the oppositethree-dimensional configuration (as the names might suggest) or the same configuration?(b) (S)-Alanine is an l-amino acid (Figure 24-2). Is (R)-cysteine a d-amino acid or anl-amino acid?give a name of any compound which is isostructural with XeF2O2CCH,CH,-C-CO2 + CH2 NH3 NH3 O2CCH,CH,-C-CO2 + CH2 CH2-C-CO2 + H. phenylalanine -CO2 0,CCH,CH2—CO2 CH2-C-CO2
- If a chiral carbon has the following substituents: -OH, -NH2, -COOH, and -OCH3 arrange them in order of prioritization (highest priority to lowest priority)? a -OCH3 > -OH > -NH2 > -COOH b -OCH3 > -NH2 > - OH > -COOH c -OH > - OCH3 > -NH2 > -COOH d -COOH > -NH2 > -OH > -OCH3The chiral catalyst (R)-BINAP-Ru is used to hydrogenate alkenes to give alkanes . The products are produced with high enantiomeric excess. An example is the formation of (S)-naproxen, a pain reliever. Q.How can one enantiomer of naproxen be formed in such high yield?Arrange the following groups in order of increasing priority. Q.) -CH3 -CH2OH -CH2NH2 -CH2Br

