Q: Which would you predict to have the lower vapor pressure at a given temperature? A. CH3(CH2)2CH3 B.…
A: Vapor pressure is inversely proportional to molecular weight and directly proportional to…
Q: Draw the product of the E2 reaction shown below. Include the correct stereochemistry. Ignore any…
A: For an E2 reaction to occur the leaving group (Br in this case) and beta-proton should be anti to…
Q: 58. Draw the mechanism for the following reaction. Identify all new asymmetric centers that are…
A: Addition of HCl to an alkene system goes via carbocation formation intermediate. If there is any…
Q: Given the information A+B C 2D D calculate AG at 298 K for the reaction A+B 2C AG = -131.75…
A:
Q: What is the correct term for the type of reaction shown below? A Friedel-Crafts Alkylation B)…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Draw the second best resonance structure for the following aromatic compound.
A:
Q: Give detailed Solution....show work...don't give Handwritten answer...don't use Ai for answering…
A: The objective of the question is to find the equilibrium concentration of F2 gas in a chemical…
Q: The mechanism shown here, along with their corresponding activation energies, is proposed for a gas…
A: The mechanism given is,Step 1: N2O5 NO2 + NO3 Ea = 25 kJ/molStep 2: NO2 + NO3 → NO2+ O2 + NO…
Q: For the molecule shown below, mark all of the correct choices. CH3 CH3 α NH3 Front view The…
A: There are two chiral centers Alpha and Beta
Q: Which of the following represent a correct mechanistic step for a rearrangement? A C ОА Ов Ос OD В D
A: Direction of arrow shows flow of electrons from higher electron density to lower.
Q: student ran the following reaction in the laboratory at 314 K: CH4 (9) + CC14 (9) = 2CH₂C1₂ (9) When…
A: The initial moles of = 0.0377 molThe initial moles of = 0.0715 molThe volume of the container =…
Q: Draw the Newman projection so that it corresponds to the molecule and conformation shown when viewed…
A: In a Newman projection, the line of sight is along a specific bond axis.The front carbon is…
Q: A. 1. LiAlH4 2. H*
A: Lithium Aluminium Hydride is a reducing agent which reduces carbonyl compounds like ketones and…
Q: Part A A sample of a gas is in a sealed container. The pressure of the gas is 695 torr, and the…
A: The formula used here is - PV = nRT(Ideal gas formula)At const n and V, we have relation -
Q: 4. Fill in the missing reagents in the following synthesis. 04-04-0 1) 3) HO
A: To find the reagents for the synthesis of given target molecule
Q: Н С H C + I2 (aq) 1-CH2 H3C O + HI (aq)
A: Definition: Chemical reactions involve the transformation of reactants into products through the…
Q: E = -1.48V 3. For the reduction of Cr(OH)3 by H₂ in basic solution to give Cr and H₂O, write the…
A:
Q: Which of the following is TRUE regarding thioesters? Select three of the following: Hydrolysis of…
A: The three statements that are TRUE regarding thioesters are:b) Acetyl-CoA, a thioester, has more…
Q: best way to describe the final major product? 1. EtMgBr 2. H+ pon secondary alcohol pon ketone Don…
A:
Q: aw the product of the reaction shown below. Ignore inorganic byproducts HO P CrO3 acetone, H3O+ 11 1…
A: This reaction is the oxidation reaction of alcohol,in presence of Cro3 alcohol convert to aldehyde…
Q: Arrange the following solutions in order of increasing hydroxide ion concentration (lowest to…
A: The objective of the question is to arrange the given solutions in order of increasing hydroxide ion…
Q: What is the electron dot structure of PO 3? [Use the following templates, where A and B are…
A: To draw a Lewis structure of any molecule we should know the number of valence electrons of the…
Q: Mechanism. Provide the complete mechanism for the reaction below. You must include appropriate…
A: In the first step of this reaction,benzyne formation takes place,then presence of heat diels alder…
Q: Consider the reaction: C₄H₄(g) +2 H₂(g) ⇌ C₄H₈(g) Kc = 2.35 If at equilibrium the reaction vessel…
A: Given that, the reaction is:
Q: P Identify each of the following compounds as an ester, alc ethers, ketones, aldehydes or…
A: We are given an organic compound, we have to tell it's functional group
Q: Which of the following statements is FALSE? O Vapor pressure increases with increasing temperature…
A: Vapor pressure is a fundamental property of liquids that plays a significant role in phenomena such…
Q: a. b. о - ОН
A:
Q: ОН conc. H₂SO4 Heat
A: Elimination of water is known as dehydration reaction.Alcohols will react with acid to eliminate…
Q: At 367 K, this reaction has a Ke value of 0.0640. X(g) + 2 Y(g) 2Z(g) Calculate K₂ at 367 K. Note…
A:
Q: At the freezing point, the liquid form of a substance Choose... solid form of the substance. the
A: The freezing point is defined as the phase change of the substance from a liquid to the solid state.…
Q: Give detailed mechanism Solution with explanation needed of each steps..don't give Handwritten…
A: The objective of the question is to determine the product of a sequence of reactions involving PPh3,…
Q: CH3 CH OCCH3 CH3 Draw all missing reactants and/or products in the appropriate boxes by placing…
A:
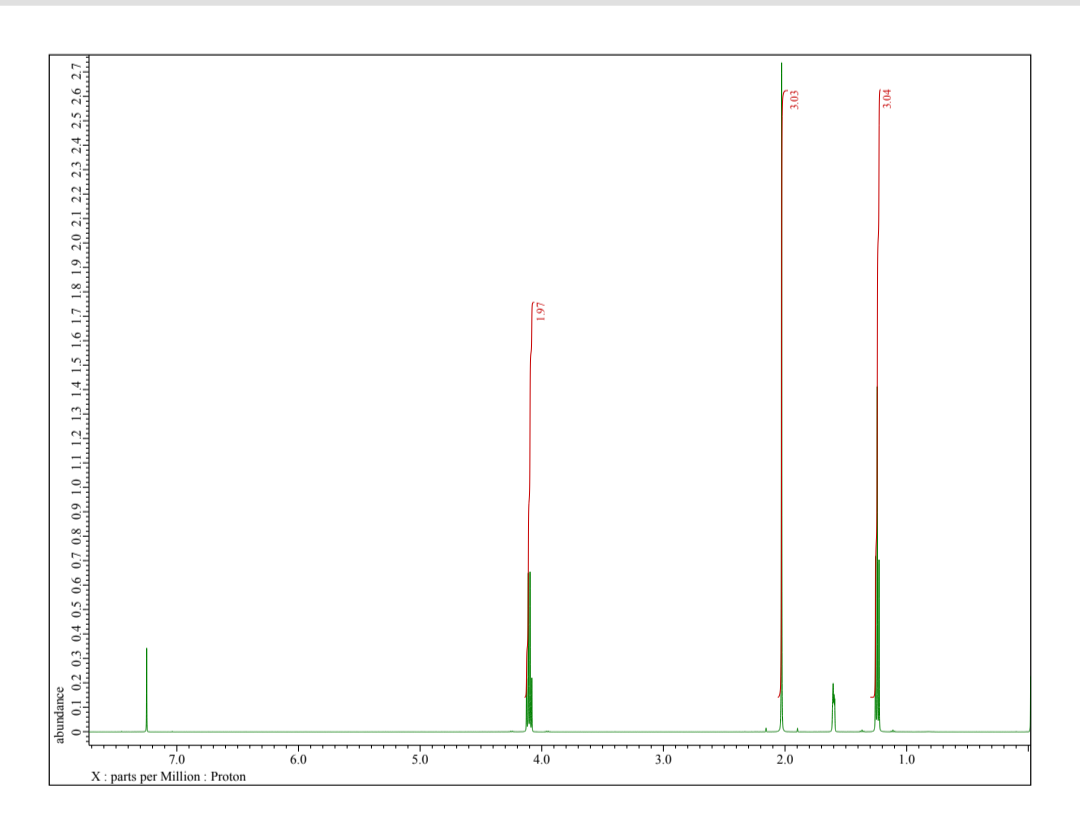
Q: A compound, C₁0H14 shows an IR peak at 745 cm. Its ¹H NMR spectrum has peaks at delta 7.18 (4 H,…
A:
Q: 1. KMnO4, H₂O, heat 2. H+ (workup) the e VON o
A: KMnO4 i.e. potassium permanganate is a strong oxidising agent.which is used for the oxidation of…
Q: 19. The following data was obtained in a chemical kinetics experiment in which the concentration was…
A: Answer:Average rate of formation in a time period is equal to the ratio of change in concentration…
Q: The Ka for lcarbonic acid is 1.8 × 10–5. Which of the following statements best describes the pH of…
A: The objective of this question is to determine the pH of a 0.0010 M solution of carbonic acid, given…
Q: Select the solution below that is the most acidic. [H3O+] = 1.0 × 10–14 M [H3O+]…
A: The objective of the question is to identify the most acidic solution from the given options. The…
Q: Draw the major organic product for the reaction shown Br 1. PPh3 2. n-butyllithium 3. H O
A: The conversion of carbonyl compounds into an alkene by the reaction with the triphenyl phosphonium…
Q: Draw the organic product(s) you would expect from the following reaction. Assume products derive…
A: The reactant is a diene. It is an organic compound. The reagent is hydrochloric acid . Only 1 mol is…
Q: Problem 5.40-Enhanced - with Feedback b. H₂ Pd catalyst
A: Reduction of alkene gives alkane as product. Reduction means addition of hydrogen to the double…
Q: What is the name of the following? CI NO₂
A: The IUPAC rules for naming cyclic compounds are given as follows:For cyclic unsaturated compounds,…
Q: Interpret the attached set of spectra to propose a structure for the compound. Write directly on…
A: NMR spectroscopy is associated with NMR active nucleus. 1H and 13C-NMR spectroscopy is mainly used…
Q: please fill in..
A: Hydrogens present at terminal alkynes are acidic in nature due to alkyne involvement in the sp…
Q: Draw structural formulas for an aldehyde or ketone and alkyl (or aryl) bromide that could be used in…
A:
Q: For the following carbocation determine if it is likely to rearrange, and if so, select the correct…
A: The carbocation rearranges itself when it gains more stability. We know the stability of carbocation…
Q: Draw the structure of the major organic product(s) of the reaction. + • You do not have to consider…
A: The objective is to determine the product formed by the given reaction. Esters on reaction with…
Q: Rank the compounds or ions in order of increasing acidity (or decreasing pK₂). Least acidic H &…
A:
Q: Do not give handwriting solution.
A: All mentioned distances:- A,B,C,D and E are veriable.Explanation:There are 3 steps to solve this…
Q: Give the IUPAC name for the following compound.
A: IUPAC nomenclature is used for naming the organic compound as recommended by the International Union…
Q: Choose the best option for the precursor to the dibromide intermediate. S (Α) E TSO Br OTS CI Br B D…
A:
Step by step
Solved in 3 steps with 1 images

- You dissolve 0.300 g of an unknown organic molecule in 10.0 g of camphor . The freeing point of the camphor decreases by 3.07 degrees Celsius. What is the molar mass of the unknown compound? Kf= 39.7oC/m 94.0 g/mole 97.0 g/mole 194 g/mole 47.0 g/mole 388 g/moleThe amount of urea in ppm [parts per million] present in an aqueous solution containing 45 % by mass of solvent is ______ 8.18 X 10 to the power of 5 space end exponent p p m 1.22 X 10 to the power of 6 space end exponent p p m 4.50 X 10 to the power of 5 space end exponent p p m 5.50 X 10 to the power of 5 space end exponent p p mH2SO4(aq)+NaOH(s)→H2SO4(aq)+NaOH(s)→ (neutralization of both acidic protons) Express your answer as a chemical equation. Identify all of the phases in your answer.
- What is the molar fraction of glycine in an aqueous solution whose concentration is 0.140 molkg-1. Data: Mass H2O = 1 kg; M.M. H2O = 18.01 gmol-1.Calculate for the mass of glyceraldehyde (C3H6O3) needed to prepare 250 mL of each of the following solutions?a. 78% (w/v)b. 1.65 Mc. 890 ppmThe partial molar volumes of acetone and chloroform, in a solution in which the molar fraction of chloroform is 0.4693, are 74.166 cm3mol-1 and 80.235 cm3 mol-1, respectively. What is the volume of 1,000 kg of this solution? Data: M.M. (acetone) = 58.08 gmol-1; M.M. (chloroform) = 119.07 gmol-1.
- Saturated aqueous sodium chloride (d = 1.2 g/mL) is added to the followingmixtures in order to dry the organic layer. Which layer is likely to be on thebottom in each case?a. Sodium chloride layer or a layer containing a high-density organic compound dissolved in methylene chloride (d = 1.4 g/mL)b. Sodium chloride layer or a layer containing a low-density organic compound dissolved in methylene chloride (d = 1.1 g/mL)For the ppm of K2Cr2O7 why did you multiply by 10^6? What is the 10^6 suppose to represent or mean?A U-Tube is divided into left and right compartments by a membrane that exclusively allows water to pass through but not any dissolved particles. Initially, the water level is equal on both sides. Then. the following compounds are added to each compartment: Left compartment: 0.50 mol calcium nitrate and 1 mol of glucose(C6H11O6) Right compartment: 0.75 mol maltose (C12H22O11) and 0.75 mol AlPO4 Select the one statement below that is TRUE. a)Water will stop flowing across the membrane b)The right side compartment will have a higher osmotic pressure c)There will be no change in the water level on either side because water always flattens out d)The water level on the right side will be higher e)The water level on the left side will be higher
- Estimate the osmotic pressure associated with 24.5 g of an enzyme of molecular weight 4.21 x 10-6 dissolved in 1740. mL of ethyl acetate solution at 38.0°CWhat is Brownian motion, and what causes it?15.00 ml of a H3PO4 solution required 21.45 ml of 0.125 N NaOH fpr complete neutraliztion. calculate the normality and the morility o the phosphoric acid solution.

