Q: 9. Write chemical equations to show what happens when each of the following substances is mixed with…
A: According to guidelines i can answer only 1st three sub parts.
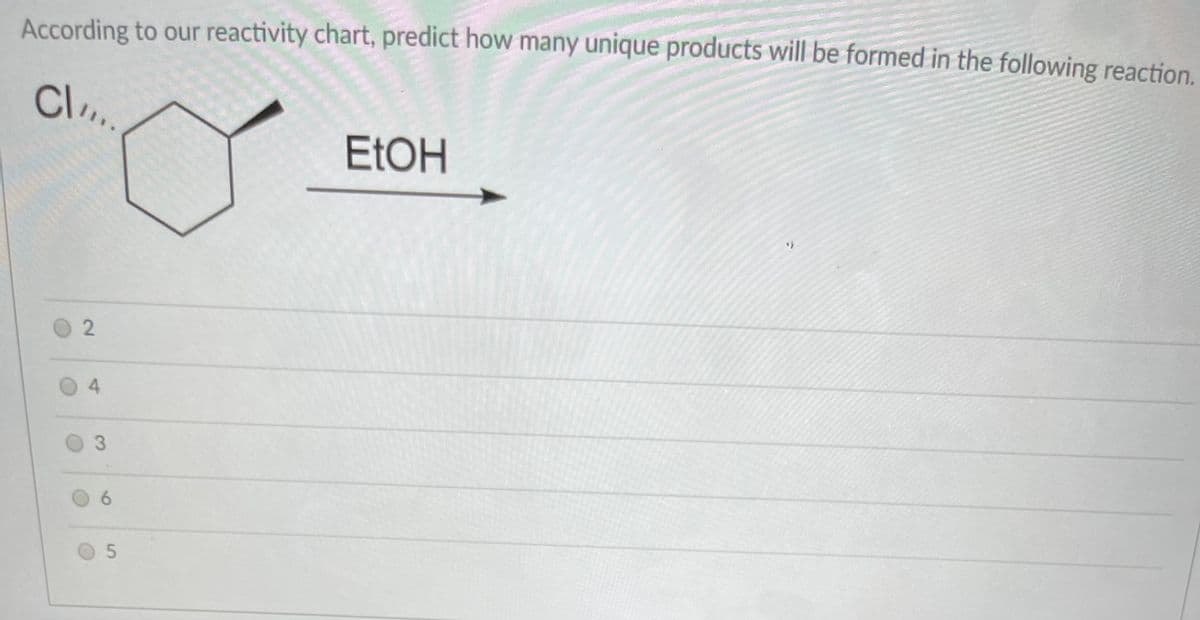
Q: Which of the statements below is true about the reaction? H OH NABH4 CH3 CH3 ELOH m wt. = 120.2
A: In the above reaction, Acetophenone is treated with NaBH4 and the product obtained is 1-phenyl…
Q: 1. Which action between Mg ribbon and HCl will increase the rate of reaction? A. Crumple the Mg…
A: Rate of the reaction can be defined as the speed at which chemical reaction proceeds.
Q: What is the [ OH 1j in a 0.01 M KOH solution? O a. 1 x 10-2 M Ob.1X 10-14 M Oc1X 10-7 M O d.1x10-12…
A: Given, concentration of KOH solution =0.01 Mwe are asked to find the concentration of OH- ions.
Q: 1.) Describe what do you expect to observe when the following reactions take place. FeSO4(aq) +…
A: When two compound react and result into new compounds or element is called as chemical reaction.
Q: If the concentration of H* ions in a solution is 3.16 x 10-4 mal/1, then what is the concentration…
A:
Q: Which of the following compounds, when dissolved in water will produce an acidic solution? X.…
A: Given salts: KNO3 CH3NH3Cl NaHSO4 We have to find the salts that will produce acidic solutions.
Q: 21: Which of the following equations explains why this reaction forms a hydroxide upon the addition…
A: Coordination compounds are those addition molecular compounds that retain their identity in…
Q: According to s principle, what would happen to the chemical balance of the ollowing equilibrium when…
A: Le Chatelier's principle states that if a dynamic equilibrium is disturbed by changing the…
Q: Vrite the balanced NET IONIC equation for the reaction that occurs when hydroiodic acid and ammonia…
A: Net ionic equation of HI + NH3
Q: H3C—N̈H2 reacts with BF3 in a combination reaction, forming H3C—NH2—BF3 with a covalent bond between…
A: Lone pair donor is base and lone pair accepter is acid. Proton donor is acid and proton accepter is…
Q: Write a chemical equation for the hydrolysis reaction that explains why an aqueous solution of…
A: Ionisation of CH3NH3Cl :CH3NH3Cl (aq) --------------> CH3NH3+ (aq) + Cl- (aq)
Q: What is the product of this reaction? 02, NaOH Aliquat 336
A:
Q: Predict the products of the following reactions by completing and balancing the reaction equation.…
A: fourth reaction is not possible because more acidic product is formed.
Q: What is the equilibrium concentration of hydroxyl ions present in a solution of HClO4 whose…
A:
Q: Given these reactions, answer the questions (one answer can be used twice) NaOEt Br EtOH YBr Br…
A:
Q: 12. Which one has the lowest concentration of H3O* ions? A. 0.10 M HNO3 B. 0.10 M acetic acid C.…
A: Strong acids dissociate in solution completely and H+ ions are formed. Similarly, strong bases…
Q: Explain how Compound A and Compound B will dissolve in water using the correct vocabulary including…
A: Compound B is KBr which is an ionic compound formed by transfer of 1 electrons from K to Br atom…
Q: 4. What are the reaction products (ions) formed when solid potassium bromide is dissolved in water?…
A: To find: reaction products when solid potassium bromide is dissolved in water.
Q: CH3OH OMs OМе This is an example of a solvolysis reaction.
A: CH3OH is a basic nucleophile and -OMs is a good leaving group. Since the leaving group is attached…
Q: If the concentration of H ions in a solution is 3.16 x 10-4 mel, then what is the concentration of…
A: Given, Concentration of the H+ ion = [H+] = 3.16 × 10-4 mol/L Concentration of the OH- ion = [OH-] =…
Q: 5. A reaction between sodium borate and acetic acid is a neutralization reaction (just like a…
A: Given, Neutralisation reaction between sodium borate and acetic acid. The balanced chemical equation…
Q: Consider the reaction below: K2O(aq) + MgBr2(aq) ----> _ Compound A + _ Compound B (OR K2O(a q) +…
A:
Q: a. OH H2SO4 b. HBr
A: Detail mechanistic pathway is given below
Q: How many millimoles of phosphoric acid (a triprotic acid, H3A) can be completely neutralized by 20.0…
A: From given 20 ml of 0.252 M KOH = (20x0.252/1000) moles KOH=0.00504 moles KOH
Q: How do I fill out this chart Reactions: NaHCO3(s) + HCl(aq) → NaCl(s) + CO2(g) + H2O(l)…
A: Mass of the mixture = 0.5093 g Mass of residue remaining after first weighing = 0.4537 g Mass of…
Q: What is the concentration of chloride ions in a 0.250M MgCl2 solution? O 0.250M 0.500M O 0.667M O…
A: Concentration of MgCl2 = 0.250 M Concentration of chloride ions = ?
Q: NaOH Option 1 Option 2 Option 3 O Option 4
A: Option 2 is correct
Q: 1. Solid NH4NO3 dissolves in water and then reacts with KOH (aq). The container releases a smell as…
A: When aqueous solution of NH4NO3 treated with KOH, acid - base reaction occurs.Here we have to give…
Q: What will happen to this equilibrium when NaOH is added? NH3 + H+ NH4+
A: Given-> NH3 + H+ <------> NH4+
Q: H3C—N̈H2 reacts with BF3 in a combination reaction, forming H3C—NH2—BF3 with a covalent bond between…
A: Bronsted-Lowry acid-base theory: The Bronsted-Lowry acid-base theory states that the acid is a…
Q: Question is attached
A: Given:
Q: Which of the following correctly completes the reaction? Na + Al(OH)3 → ? Na + Al(OH)3 → NaAl(OH)3…
A: We know that Balanced chemical equation : A chemical equation in which the number of each type of…
Q: Identify the concept that best fits each of the following reactions. + OH (ag) (aq) 1. 2H,O H,O* (6)…
A: Since you have posted question with multiple sub-parts, as per guidelines we will solve only the…
Q: Which reactant is the Acid in the previous reaction? HCl Mg(OH)2
A: According to Arrhenius theory, the substance which give H+ ions in water upon dissociation is called…
Q: Fill in the box to complete the reactions: 1. ВН3:THF ОН 2. Н2О2, NaBHA H2, Pd HO,. + en HO
A: Answer given below
Q: 11 of 48 This reaction must have a AG>0 at SATP: 2Li(s)+Cl2(g)→2LİCI(s) NHẠC(s)¬NH3(g)+HCI(g)…
A: Note: Since you have posted multiple independent questions in the same request, we will solve the…
Q: n this reaction: [CoCl2(ETOH)4] + 6H2O [Co(H2O)6]2++ 2Cl- 4EtOH A. what is the chemical reaction B.…
A: According to Le Chatlier principle- When an external stress is applied, the equilibrium of the…
Q: hat is the equilibrium constant expression for the following reaction? 2 NaHCO3 (aq) ⇌ Na2CO3…
A: Let , consider a general reaction aA +bB →cC +dDThe equlibrium constant in term of…
Q: 1. Determine the concentration of NaOH 2. Detremine the concentration of acetic acid 3. Calculate…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: Write a chemical equation for the hydrolysis reaction that explains why an aqueous solution of…
A: As CH3NH3Cl upon hydrolysis produces hydronium ions, the solution is acidic.Hydrolysis reaction of…
Q: Write an equation for the reaction of HNO3 with water.
A: In the reaction of HNO3 and Water (H2O), HNO3 will act as a bronsted acid (donates H+) and water act…
Q: Complete the acid/base reaction below. The first reactant given is the acid. HNO2 + H20
A: According to Bronsted Lowry concept:An acid releases H+ in solution. A base accepts H+ from the…
Q: Why do we use sodium hydroxide instead of a weaker base? What is the practical reason for the use of…
A: Sodium hydroxide is a strong base and sodium bicarbonate is a weaker base. The practical reason,…
Q: HS .COOH COOH b oxidation HS соон reduction SH HS COOH COOH d
A: In this question, what Compound ( in b, c, d and e ) will give Compound a How we can identify the…
Q: In the grignard reaction, why do you need to add HCI in this reaction? What would happen if you did…
A: Grignard reaction is used to convert ester or carbonyl group into alcohol.
Q: 3 CH3I NaOH solution NH2 [7] (11) (i) AGOH (replaces I with "OH) (ii) 180 °C [8] (14) (12) (13) 95%…
A: This question belong to reaction mechanism of organic chemistry.In the reaction 7 ,the NaOH solution…
Q: Which factor will not increase the concentration of NH3 (ammonia)? N2(g) + H2(g) ⇄ NH3(g) + heat…
A: When a reaction proceeds, the reactants convert to products. The amounts of reactants decrease with…
Q: CoCl42-(aq) + 6H2O(l) >< Co(H2O)62+(aq) + 4Cl-(aq) 1) What was the original color of the…
A: Given data : Le Chatelier’s Principle: According to the principle that if a system is in chemical…
Step by step
Solved in 4 steps with 1 images

- Which characterization accurately describes the reaction shown below?Choose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) KMnO4, H3O+ CH3Cl, AlCl3 HNO3, H2SO4 Cl2, FeCl3 fuming sulfuric acidbeginning with the compound shown, suggest a synthesis for each of the compounds shown below. Clearlyshow all intermediates and reaction conditions. Several steps are required for each synthesis conversion
- Below is a schematic representation of possible reactions that Compound X can undergo. Use the scheme to answer the following questions. Illustrating with reaction mechanisms, show how compounds [1], [2], [3] and [4] are formed. Which of the compounds in the following pairs will occur in relatively higher yields and why? [1] and [2] [3] and [4]Write the products of the compound and the following reagents. If the reaction would not work for your compound, write "no reaction" and explain the problem. CrO_3Complete the following reaction equations :(i) C6H5Cl + CH3COCl →(ii) C2H5NH2 + C6H5SO2Cl →(iii) C2H5NH2 + HNO2 →
- Determine the correct reagent(s) from the list below to complete the following reactions.Write in the correct letter (a, b, c, or d) for each box provided.I recovered 3 mL of water, how many grams of water was produced in the reaction please show how? Cyclohexanol can attack the carbocation intermediate formed during the synthesis of cyclohexene. Draw this side product with the molecular formula C12H22O.Rank (1-4) from most reactive to least reactive (in decreasing reactivity in an elimination reaction with NaOH in DMF at 100 degrees celsius)
- Rank in terms of reactivity towards the nucleophile. 1=least 4=mostChoose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) Br2, FeBr3 Na/NH3, -33 degrees C NBS, light KMnO4, H3O+ Mg metal, ether KOH, EtOH, heatThe following two step synthesis reduces the amount of waste generated by incorporating most of the atoms in the reagents into the products. Predict the products of each step.

