Q: 1) Classify the following compounds as R or S? Br H H. 2. .....Br 1. F C CH3 CI CN |CH, NH2 H. 4. 3.…
A: According to Cahn-Ingold system, the chiral carbon has either R or S configuration. The atoms…
Q: OMg +o -> MgO Mg = Mg = %3D %3D 0 = %3D %3D
A: Complete the reaction by using coefficient---
Q: 1. Provide the name for the following compounds. CI F "CI (a) (b) (c) Br (d) (e) (f)
A:
Q: Mg(NO3)2 Na2CO3→ Ca(NO3)2 Na2CO3 → -> Sr(NO3)2 NazCO3 → Ba(NO3)2 Na2CO3 → ->
A: The group 2 metals like Mg, Ca, Sr, Ba reacts with Sodium carbonate to give the corresponding metal…
Q: 6. F AICI3 -OCH3 7. AICI3 -F + Cl 8. Br2 — ОН CHC!3 9.
A:
Q: CI с. AICI3 1) HNO3, H2SO4 d. 2) Zn / HCI CI 1) СО, НСI е. 2) SO3, H2SO4
A: Acetanilide -HN- COCH3 group is ortho – para directing. Because of steric hinderance and the…
Q: Q-10. Vinegar (a) + Baking Soda (b) → Sodium Acetate + Carbon dioxide + Water. Write the molecular…
A: (a) Vinegar = CH3COOH (b) Baking Soda = NaHCO3 CH3COOH + NaHCO3 ----> CH3COONa + CO2 + H2O…
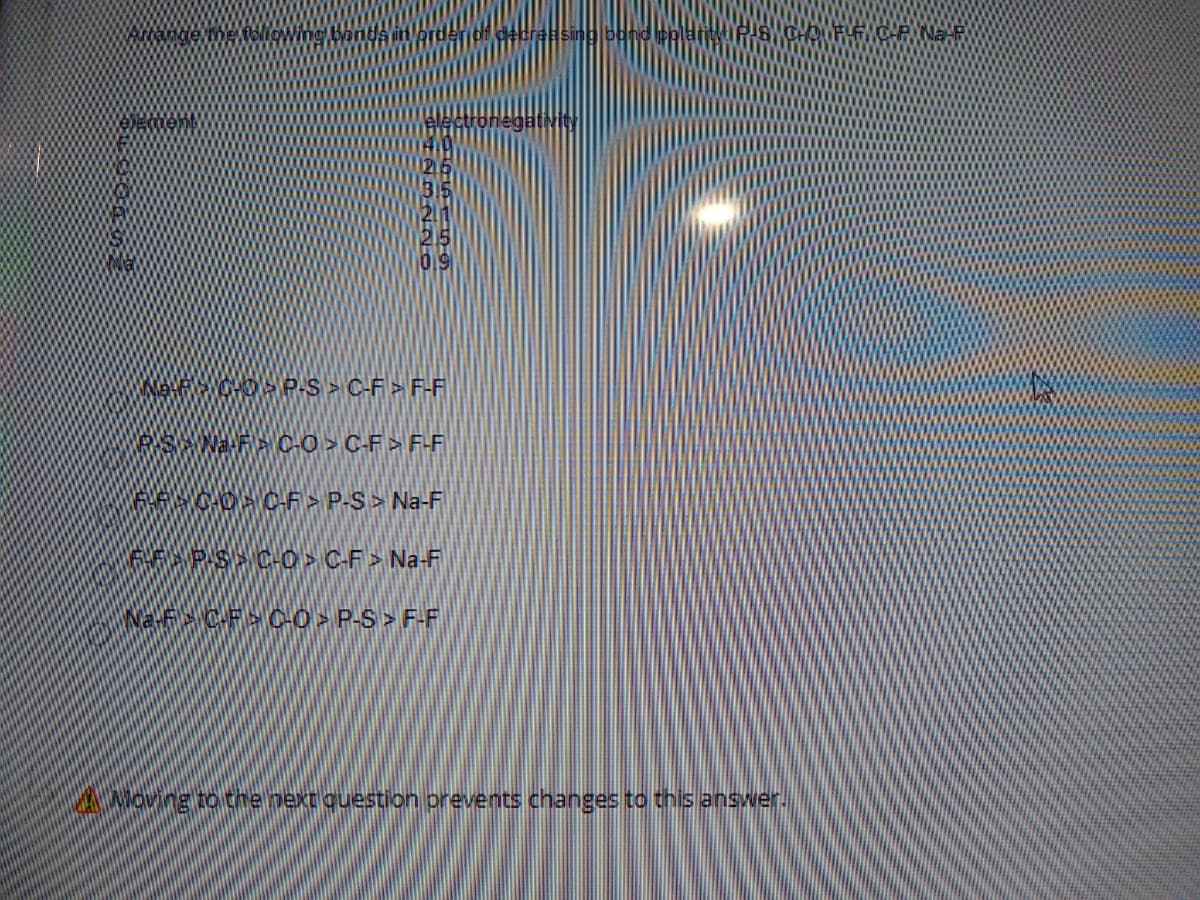
Q: Using the information given in Table 4, fill the blanks with > or <. IE1 –-- IE2 -- IE3 Li: IE3 IE1…
A: The data given is,
Q: INORGANIC CHEMISTRY MODULE 2 ASSIGNMENT: Write the Formulas OF Compound in which combining ratios…
A: The formula of a compound gives information about the elements that are present in the compound. The…
Q: 1. 2 Li, Et,0 2. 0.5 equiv. Cul. 3. CH3(CH2)½Br -CI
A:
Q: oms. Be sure to follow the Law of Conservation of Mass. 0 Zn + HCI ZnCl2 + H2 2. C12H22011 C + H2O…
A: A balanced reaction is a reaction in which the number of atoms of each element is same on both…
Q: Solve the attached Problem.
A: The given reaction is the reduction of alkynes to trans alkenes in the presence of liq. NH3. The…
Q: NABHz CN NH3, Hg ot H3 Ot Por3, KoC (cH3h. 0s \. BH3 2. +202, O4- crqele DCC
A:
Q: Table 1. Tabulated data from Figure 2. Rel. Abundance m/Z Proposed Identity ~1 45 100 44 ~11 28 ~2…
A:
Q: Fe 1.) 2.) 3.) 4.) 5.) 6.) 7.) 1s 2s'2p 8.) 9.) 10.) 11.) 12.) Bal. 13.) 14.) 15.) 16.) 17.) 18.)…
A: Element Electronic configuration Noble gas configuration n…
Q: 10. Which of the following subs CBr CBr F CBr,F2 CBRF CF 11. Which of the following liq CH;OCH;…
A: CF4 has lowest boiling point Explanation: The higher the intermolecular forces, the more the boiling…
Q: Which of the following pairs represent isotopes? A) ²⁸Si and ²⁸Si⁴⁺ B) ²⁸Si and ²⁸Al C) ²⁸Si and…
A: Isotopes basically mean the same element having the same number of proton but with the different…
Q: 6) Circle the set or sets offquontum.nucmbers: thatis orare erroneous. a)n=4l=2 me=-2 (6)h=22=2…
A:
Q: Fill in the blanks for the 1sr image . For the second image it’s filled in which is an example to…
A: Molar mass of the compounds is equal to the sum of the masses of all the elements.
Q: KPO, (ag) • 3AGNO, (ag) -- Ag,PO, (s) + 3KNO, (ag) 3K"(ag) • PO,(ag) • 3Ag'(aq) • 3NO, (aq) -->…
A: Given-> K3PO4(aq) + 3AgNO3(aq) ---> Ag3PO4(s) + 3 KNO3(aq)
Q: 15- Two compounds formed by iodine and fluorine showed the following data, for a fixed mass of…
A: GIVEN: The mass of I in compound I = 1.0000. The mass of F in compound I = 0.1497. The mass of I…
Q: if the RD of HFO is said to be 0.84, what is its mass for 800m3? 67 200 tons 672 tons…
A:
Q: c. Al + Cu2+ -> Al3+ + Cu С. d. Zn + 2HNO3 -> Zn(NO3)2 + NO2 + H20
A: Balanced the redox reaction ---
Q: isotope 0-16 % abundance relative mass mass contribution 99.7572 15.9949146 0-17 0.0381 16.9991317…
A: Average atomic mass is the average of atomic masses of all the isotopes of an element found in…
Q: Mg + O2 -> MgO Mg = Mg = %D O = %3D
A:
Q: 4. НОСН2CH2ОН 5.
A: The International Union of Pure and Applied Chemistry (IUPAC) nomenclature of organic chemistry is a…
Q: 10 Which of the following sets, are all compounds? a) Water, Salt, Ammonium nitrate b) Carbon…
A: All the matters are composed of atoms. The matter that formed by only one type of atom is known as…
Q: Determine possible chemical formulas for the following M* A M+ = 98 m/z В M+ = 72 m/z C M+ = 88 m/z
A: Hello. Since the question contains more than three sub-parts the first three sub-parts shall be only…
Q: 5.) The atomic weights of three compounds are shown. Use a linear system and Cramer's rule to find…
A:
Q: Calculate the densHy of Zinc using the atomic masS Gf Zinc and m dimensions of 3.5 x 2.5 x 25mm
A:
Q: Which of the following pairs would be isotopes? A) 46X & 46X B) 52X & 53X C) 62X & 63X D) none is a…
A: Which of the following pairs would be isotopes?
Q: Match the following tems with their correct definition. Empareja los siguientes téminos con su…
A: Chemical property ----> A characteristic of matter that can be observed or measured when changing…
Q: Use data from Appendix C, Figure 7.10, and Figure 7.12 to calculatethe lattice energy of RbCl.
A: The Born–Haber cycle is purposed the calculation of reaction energies with the help of ionization…
Q: For numbers 1-6, refer to the given choices below. A. Isotopes B. Isobars C. Isotones 1. 23892U and…
A:
Q: Fill in what is missing in the ZApen table. Use a separate sheet of paper for your answer. Element…
A: All things are provided in handwritten solution
Q: Group 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 Period 2 He 2 4 8782 № 10 Ne LI Be N 13 14 11 12…
A: Amount of positive and negative charge can be figure out from the number of protons and electrons.
Q: Which pair could be used to demonstrate the law of multiple proportions? A. CuCl2 and CuCl B.…
A:
Q: What is the chemical formula for a molecular model with 2 nitrogen atoms and 4 hydrogen atoms? Also…
A: a) Since the model is having 1 black and 4 white balls. Hence it is having 1 carbon i.e C and 4…
Q: МОМОМОМ OMOM BF3-OEt, A RO" OTBS ÓTBS 2 R= H (52%) 3 R= MOM (30%)
A:
Q: 25 1dentify which is E or Z 25 deity which ins Er CH,"
A: As per the CIP rules, if high priority groups are on the same side of the C=C then it is named as Z…
Q: 43 100 80 60 27 29 40 28 41 42 39 20 58 15 10 20 30 40 50 60 Mass/charge (mle) Relative abundance…
A: Mass spectrometry is the most accurate method for determining the molecular mass of the compound and…
Q: Which is the correct order of size (from smallest to largest) of K, K+, Ca, and Ca2+? a)Ca2+ < K+…
A: The correct order of size (from smallest to largest) of K, K+, Ca, and Ca2+ has to be arranged…
Q: 2H зн 3H 2H 2H IS7.44 2.85-
A:
Q: In each of the following equations, what hybridization change, if any, occurs for the underlined…
A: Hybridization is a phenomenon which involves the mixing of atomic orbitals of similar energy in…
Q: 3235 92U > 234 90TH + 4 2HE2+ (a) -- true False
A: Alpha decay: In alpha decay a radioactive nucleus disintegrates by ejecting an particle. In alpha…
Q: 4. Complete the table below Symbol A p* Representation e Li 1 1 eomes 8. C 12 6. 19 39 00
A: 4. (a) Atomic number (Z) = Number of protons Mass number (A) = Number of protons + Number of…
Q: Write
A: Molecular formula denotes total number of atoms present in one molecule
Q: Give the noble gas shorthand notation (ex: [He] 2s2 2p1) for Br Give the noble gas shorthand…
A: Electronic confirmation of heavier element is expressed in various ways. Their internal shell is…
Q: Which of the following atomic symbols is incorrect? a) 16C14 b) 32P15 c) 39K19 d) 17 e) N14
A:
Q: lon Nat Ca2+ Fe2+ Al³+ 02- Na20 NO so AsO
A: An ionic compound can be defined as combination of positively charged ion and negatively charged…
Step by step
Solved in 2 steps

- Really hoping for solutions since I’m having a hard time with this. Pls. skip if unsure or not willing to answer the subitems (these are all connected for one item). Thanks in advanced. When the compounds analyzed by Robrob were passed on to the next test, anotherscientist, Kikoko, needed to determine the molecular weight of the active compound. They created a solution with a concentration of 20.0%w/w from the solid active compound and the solution had a density of 1.40 g/mL. From this solution, 10μL was taken and mixed with 190μL of reagent and water. It was analyzed and showed a concentration of 5x10-4 M compound.a. What is the concentration (in molarity) of the 20%w/w solution? b. Why was the concentration of the solution expressed as %w/w initially? c. What is the molecular weight of the active compound?direct to the point answers plsso what is the answer whcih later

