Q: Draw all possible resonance structures for the following cation, and indicate which structure makes ...
A:
Q: Like carbocations, radicals formed from compounds that contain another functional group can undergo ...
A: During the reaction of an alkene with NBS in the presence of light generates a free radical on the a...
Q: A 1.2 m aqueous solution of an ionic compound with the formula M2X3 has a boiling point of 102.8°C. ...
A: Elevation of boiling point: Boiling point of the solution is higher than the pure solvent, it is kno...
Q: increase the rate of the forward reaction.
A: I am supposed to answer only one question according to the rules of bartleby.
Q: The deuterium kinetic isotope effect (kH2O/kD2O) for the hydrolysis of aspirin is 2.2. What does thi...
A: The kinetic isotope effect is greater than 1.0 when the ortho-carboxylate ion act as a general–base ...
Q: As we will learn in Chapter 28, styrene derivatives such as A can be polymerized by way of cationic ...
A: a. Figure 1
Q: Identify the structure of a minor product formed from the radical chlorination of propane, which has...
A:
Q: Draw a stepwise mechanism for the following reaction.
A: Deuterium is the isotope of hydrogen atom. Deuterium is replaced by hydrogen atom in 1,1-dideuterocy...
Q: What is the edge length in pm for a simple cubic with a radius of 145 pm?
A:
Q: A 20 years old athlete (racer) asked you togive opinion on whether predominantly eating food rich wi...
A: INTRODUCTION: Fat is the essential component for the human body when it is consumed in small amount....
Q: 21. For the reaction, CO, + H0- H;CO, which of the following statements are incorrect? a. CO, would ...
A: The given reaction is shown below. According to Bronsted and Lowery, the compound that can donate a...
Q: Which relationship correctly compares the rates of the following reactants and products?2 NOCl(g) → ...
A:
Q: Draw the products of each reaction. Indicate the stereochemistry of Diels–Alder products.
A:
Q: Predict the products of the following reactions. I figured out the three parts on the left. I just n...
A:
Q: Determine the amino acid sequence of a polypeptide from the following data: Complete hydrolysis of t...
A: We can use the above given info to write the primary structure of the original polypeptide. First we...
Q: Instead of adding to the 4a position and protonating N-5, the thiolate ion could have added to the 1...
A:
Q: jo IKIrios ennanhr u Lo porodinna emsn s Aayng v Write the two species lor the bufler systerm cmnpos...
A: Buffer solutions: Buffer solution is defined as a solution that oppose changes in while adding litt...
Q: A sample of gas has an initial volume of 158 mL at a pressure of 735 mm Hg and a temperature of 34°C...
A: By applying ideal gas equation: P1 and P2 is the pressures of the gas. T1 and T2 is the temperature...
Q: Provide suitable reagents to effect the following transformations.
A:
Q: The specific rotation of a-D-galactose is 150.7 and that of B-D-galactose is 52.8. When an aqueous m...
A: The specific rotation of α-D-galactose is 150.7˚ and the specific rotation of β-D-galactose is 52.8˚...
Q: A 1.00 L solution saturated at 25 °C with calcium oxalate (CaC2O4) contains 0.0061 g of CaC2O4. a. W...
A: Calcium oxalate salt solubility in water is very less. It is around 6.1mg in one litre of water.
Q: Answer the following questions about the eight aldopentoses: a. Which are enantiomers? b. Which are ...
A: a. The aldopentoses are eight in number which consists of D and L form of ribose, arabinose, xylose...
Q: Consider the following reaction: C2H8+ 4 O2→2 CO2+ 4 H2OIf 2.00 g of CO2is collected when 10.0 g of ...
A: The calculations for percent yield of any specified reaction is possible using the product's amount ...
Q: Which one of the following correctly ranks the compounds in order of lowest vapor pressure to highes...
A:
Q: What is the overall order of the following reaction, given the rate law? 2 A+3 B → 2 C Rate = k[A][B...
A: Given: 2A + 3B -----> 2C Rate = k [A] [B] Concept: a rate law is an equation which r...
Q: please help with all parts of this question. DOUBLE CHECK YOUR ANSWERS AS PREVIOUS TUTORS GOT IT WRO...
A: The flow chart can be completed as: In 100 mL beakers, obtain 30 mL of the 0.1 M weak acid and 30 mL...
Q: Give an example for making a buffer with a pH of 11. You can choose the chemicals needed and figure ...
A: The buffer solution having pH 11 can be prepared from the solution of a substance whose pKa value is...
Q: Eu-151 decays via alpha emission. What else is the product of this radioactive decay?
A: Alpha decay is a radioactive decay in which a heavier nucleus emits alpha particle. During alpha dec...
Q: When apples that have been cut are exposed to oxygen, an enzyme-catalyzed reaction causes them to tu...
A: The macromolecular biological substances by which chemical reaction is accelerated is termed as an e...
Q: 3. In an acidic solution the pH is (grcater than less than?) 7. In a basic solution the pH is (great...
A: According to Bronsted-Lowry concept, A base is a proton acceptor and an acid is a proton donor. Acco...
Q: Does the amount of radiation (cpm) increase or decrease as the distance between the sorce and the se...
A: Amount of radiation between the source and sensor is determine using the inverse square formula: In...
Q: A 20 years old athlete (racer) asked you togive opinion on whether predominantly eating food rich wi...
A: Saturated carbohydrates help in the absorption of fat-soluble vitamins and also required to build ce...
Q: PLEASE help me with all parts of this problem. DOUBLE CHECK YOUR ANSWERS PREVIOUS TUTORS GOT IT WRON...
A:
Q: Drawing Resonance Structures Draw two more resonance structures for each species.
A: Resonance structures: a).
Q: Complete the table below relating the values. Each value is =0, >0, or <0
A: We have knowledge about two equations: One is Nernst Equation according to this, Second is Relation...
Q: A mixture of helium, neon, and argon has a total pressure of 558 mm Hg. If the partial pressure of h...
A: In a mixture of non-reacting gases, the total pressure exerted is equal to the sum of the partial pr...
Q: The copper(II) ion reacts with phosphate ion to form an insoluble ionic compound, Ksp = 1.40×10–37. ...
A: The dissociation of solid copper (II) phosphate is as follows,
Q: Why does the vapor pressure of a liquid decreases as the strength of its intermolecular forces incre...
A: The vapor pressure is defined as the pressure a gas exerts when the gas is in equilibrium with the ...
Q: 3. Does u buffered solution always naintsiu a solution at pH ?7 Please explain. At what pH is huffer...
A: Buffer solutions: Buffer solution is defined as a solution that oppose changes in while adding litt...
Q: For the reaction 2NOCI(g) =2NO(g) + Cl2(g), Ke = 5.0 at a certain temperature. What concentration of...
A:
Q: Write the IUPAC and common names, if any, of the carboxylate salts produced in the reaction of each ...
A:
Q: When glucose undergoes base-catalyzed isomerization in the absence of the enzyme, mannose is one of ...
A:
Q: Draw orbital diagrams for the ground states of: (a) Terbium, Tb, a lanthanide element. (b) T...
A:
Q: Chemistry Question
A: In an exothermic reaction heat is released (absorbed/released), and heat appears on the product reac...
Q: Given the initial rate data for the reaction A + B → C, determine the rate expression for the reacti...
A: The rate law relates the concentration of each reactant present in the reaction to the rate of that ...
Q: Devise a synthesis of each compound using CH3CH3 as the only source of carbon atoms. You may use any...
A: a. The alkynes acts as nucleophile by removing its terminal proton. This brings negative charge on t...
Q: What is the pKa value of Tris-HCl?
A: The molecular formula of Tris-HCl is: NH2C(CH2OH)3 ·HCl
Q: At 15o C water’s Kw = 6.02 x 10–14. Write the equilibrium expression forwater’s self-ionization. Wha...
A: The equilibrium expression for self ionization of water is : or Equilibrium constant: pH =-log[H+...
Q: What is the molarity of the following solutions? A solution containing 0.95 moles of I2in enough wa...
A: The concentration formula that establishes relationship among moles dissolved and solution volume is...
How can I write a equation showing how acetic acid buffer system responds to the addition of H3O+ ions?
Acetic acid buffer consists of weak acid acetic acid and the salt of weak acid (for example – Sodium acetate). The pH of the buffer system can be determined as follows-
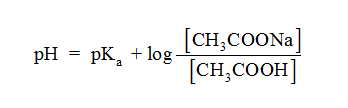
Step by step
Solved in 2 steps with 2 images

- Acetic acid buffer system responds to the addition of OH- ions. How would I write the equation showing the response of the acid?Write equations for the reaction of the buffer CH3COOH/CH3COO- buffer reacting with HCl and NaOH.show the equation for the equation for the reaction of an acetate buffer with a strong base represented by OH-
- 8. Write an equation showing how the acetic acid buffer system responds to theaddition of H3O+ ions. Write an equation showing how the acetic acid buffer system responds to theaddition of OH- ions.How do buffers work? Explain using H2CO3-NaHCO3 buffer by writing the equations for: (a) dissociation with water (b) addition of HCl (c) addition of NaOHWrite an equation for the neutralization reaction of acetic acid in solution upon addition of NaOH.

