Q: Denise Guinn THIRD EDITION presented by Macmillan Learning Write the structure of the products forme...
A:
Q: Discuss the physical examination of materials in material science
A:
Q: Provide the name of the compound shown below. N' N°
A:
Q: An example of using chemical energy is O a walking O b looking at the sun O c running d. eating
A:
Q: The following thermochemical equation is for the reaction of iron(s) with chlorine(g) to form FeCl3(...
A: You have given the chemical reaction - 2Fe + 3Cl2 => 2FeCl3 dH = -799 KJ
Q: Write the symbols and charges for the atoms given below and then identify it as anion/cation and met...
A: Metals have natural tendency to lose electrons whereas non-metal have natural tendency to gain elect...
Q: What type energy is an battery? O a Thermal Energy Chemical Energy Potential Energy Electric Energy
A: Battery is a device which can convert chemical energy to electrical energy.
Q: 0.145 mol C2 H6 Express your answer using three significant figures. VC = mol Submit Request Answer ...
A:
Q: What reagent/reagents is/are necessary to transform the starting molecule into the desired product? ...
A:
Q: assessment-delivery/start/4732295265?action=Donresume&subm 1. Draw the electrons for a simple atomic...
A:
Q: A catalyst can increase the rate of a reaction by (choose all) a. By providing an alternative mechan...
A: By using catalytic action diagram energy vs reaction coordinate.
Q: This reaction is an example of conjugate addition of a nucleophile to an a,ß-unsaturated carbonyl. L...
A: The alpha hydrogen to the carbonyl group is acidic in nature and this hydrogen is removed in presenc...
Q: Hydrochloric Acid + Sodium Hydroxide none Observation Molecular Equation Complete Ionic Equation Net...
A: (a) The reaction of hydrochloric acid with sodium hydroxide results in the formation of sodium chlor...
Q: QUESTION 21 Which of the following would be the unit for the rate constant of the reaction reaction ...
A: Given, rate of reaction is r= 12 M/min This reaction is 2nd order with respect to A and zero order w...
Q: Indicate the element and the # of atoms of that element present in 2 molecules of acetic acid (CH3CO...
A: Given :- chemical formula of acetic acid is CH3COOH To calculate: number of atoms of each element i...
Q: Chemistry Question
A: According the the first law of thermodynamics, ∆U = q + w Where : ∆U = internal energy q = heat ...
Q: Calculate the overall enthalpy change for the reaction
A: *In H2O, two O-H bond is present and stoichiometry is 2 so bond energy =2×2×464 KJ/mol=928KJ/mol *In...
Q: What is the major organic product obtained from the following reaction? HO, H. 1. NaOH, H20 1 OCHS 2...
A: Given: Reaction To find: The correct product formed Solution: when esters react with sodium hydroxid...
Q: Use the References to access important values if needed for this question. What is the energy change...
A: Given mass of solid iodine = 11.9 gms Change in temperature∆T = (37.7 °C- 23.6°C) = (310.7 K- 296.6 ...
Q: What are catalysts?
A: The explanation of catalysts has to be given.
Q: Are these 4 quantum numbers possible? 3, 3, 3, 1/2
A: Start by making sure that you're familiar with the valid values each quantum number can take. A...
Q: 2. You perform some experiments for the reaction A - B + Cand determine the rate law has the form Ra...
A: Ans The rate law for the given reaction is as follows Rate =k[A]x a). if concentration of A is trip...
Q: What is the major organic product obtained from the following reaction? 1. NaOCHCHs CH,CH,OH CHCH O....
A: The given reaction is an example of the Dieckmann reaction. In this reaction, the diester reacts wit...
Q: A 9.00 L tank at 4.56 °C is filled with 8.55 g of chlorine pentafluoride gas and 9.34 g of dinitroge...
A: Number of moles of any Substance can be calculated by - Moles = mass/Molar mass Here, Mass of Chlori...
Q: Balance the equation below then use it to answer the following questions.
A: a) In the Balance chemical equation, the no of elements of each type on reactant and product side ...
Q: What is the pE value in a solution in equilibrium with air (21% O2 by volume) at pH 6.00? а. 16.6 b....
A:
Q: An aqueous solution of aluminum nitrate is mixed with an aqueous solution of lithium carbonate. What...
A: An aqueous solution of aluminum nitrate is mixed with an aqueous solution of lithium carbonate. then...
Q: QUESTION 7 What is the Henry's Law constant for 0.1966 grams of a substance dissolved in 22.9 mL of ...
A: Mass of the substance = 0.1966 g Volume of the water = 22.9 mL Partial pressure = 777 ...
Q: at what pressure is the boiling point of water the same as its freezing point
A:
Q: How many half-lives are required for the concentration of reactant to decrease to 25% of its origina...
A: Given:Let the initial concentration be=N∘Final concentration= 25% of initial concentrationN=25100N∘t...
Q: What is the 3rd resonance structure?
A: various resonating structures are formed when there occur a conjugating system in the molecule. for ...
Q: 1. Draw the electrons for a simple atomic model of Oxygen. 2. Draw a square (0) around the valence e...
A: Oxygen (O) Atomic number = 8 Number of electrons = 8 Number of protons = 8 Protons are present i...
Q: Classify each of the following molecules as polar or nonpolar: Drag the appropriate items to their r...
A: Bond have dipole moment is polar whereas with zero dipole moment is non-polar.
Q: Of the choices below, which two pieces will undergo a Robinson annulation to giv che product shown?
A: The Robinson annulation is reaction where the Michael addition followed by Aldol condensation takes ...
Q: when ions are formed when solid MgSO4 dissolves in water? Mg2+, SO42- Mg2+, 4 SO- Mg2+, S2-,4O2- Mg+...
A: Hence th answer is Mg2+ So42-
Q: The osmotic pressure of an aqueous solution of a protein at 22degree celcius was used to determine t...
A: Given , Temperature (T) = 22°C = (22+273.15) K = 295.15 K Mass of protein (m) = 7.43 mg = 7.43×10-3 ...
Q: The father of thermodynamics is: O Sadi Carnot O John Dalton O Linus Pauling O John Adams
A: Nicolas Leonard Sadi Carnot is known as the father of thermodynamics.
Q: Question 4 By clearly showing your working steps, the combustion of which of the following fuels wo...
A:
Q: Which energy transformation occurs with a toaster? (The toaster is plugged in and turned on) O a Pot...
A: Introduction: Electric energy is one of the type of kinetic energy. The energy is produced due to mo...
Q: of energy to trigger a series of impul= is wavelength, 2.9 um, are needed?
A: Energy = N • h • c / ( wavelength ) Where : N = number of photons h = 6.626 • 10^-34 Js c = 3.0 •...
Q: 7. One reason gold is so prized is because it seldom reacts. However, a mixture of nitric acid and h...
A: Gold reacts with a mixture of nitric acid and hydrochloric acid to produce the salt of gold and nitr...
Q: 2. Compound P, C4H9Br reacts with aqueous solution of sodium hydroxide to form compound Q, C4H10O. C...
A: Oxidation of alcohol KMnO4 is used as an oxidizing agent. When alcohol react with KMnO4, primary alc...
Q: Determine the pH of the 1.5M HNO2. The Ka = 4.0 x 10^-4 . HNO2(aq) + H2O(l) ↔ NO2 - (aq) + H3O ^+ (a...
A: Given that Ka for HNO2 = 4.0 x 10^-4 write the ICE Table and solve for the x by writing the Ka expre...
Q: which compound below is a strong electrolyte when dissolved in water? none of these compounds is a s...
A: A strong electrolyte is a solution in which a large fraction of the dissolved solute exists as ions....
Q: The radiation R (t) in a substance decreases at a rate that is proportional to the amount present; t...
A:
Q: Given that a reaction is Exothermic in the foward direction and has a forward activation energy of 5...
A:
Q: Chemistry Question
A:
Q: In a certain instant during the course of the reaction below the rate formation of NO2F was 0.16M/s....
A:
Q: Your body transforms the used for movement. Thermal Energy, Radiant Energy
A: Solution- Your body transforms the chemical energy form your food into the kinetic energy used for m...
Q: How many moles of Fe2O3Fe2O3 are in 171 g171 g of the compound?
A: Molar mass of Fe2O3 = 2 • ( molar atomic mass of Fe ) + 3 • ( molar atomic mass of Oxygen atom ) Mo...
Draw the electrophile formed by the following reagents/conditions below.
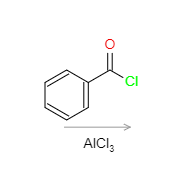
Step by step
Solved in 2 steps with 1 images


