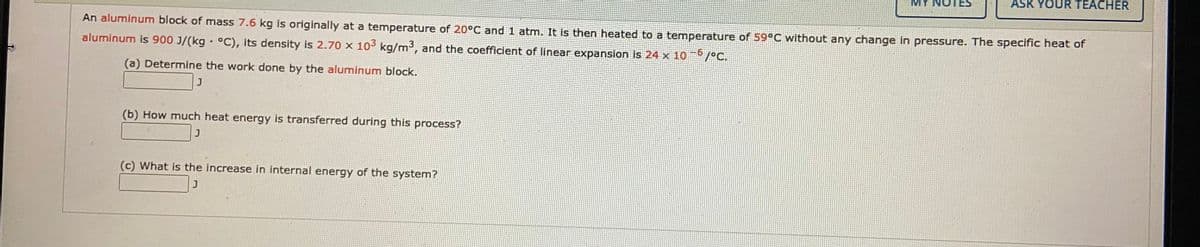
An aluminum block of mass 7.6 kg is originally at a temperature of 20°C and 1 atm. It is then heated to a temperature of 59°C without any change in pressure. The specific heat of aluminum is 900 J/(kg - °C), Its density is 2.70 x 10 kg/m3, and the coefficient of linear expansion is 24 x 10 -6 /ec. (a) Determine the work done by the aluminum block. (b) How much heat energy is transferred during this process? (c) What is the increase in internal energy of the system?
An aluminum block of mass 7.6 kg is originally at a temperature of 20°C and 1 atm. It is then heated to a temperature of 59°C without any change in pressure. The specific heat of aluminum is 900 J/(kg - °C), Its density is 2.70 x 10 kg/m3, and the coefficient of linear expansion is 24 x 10 -6 /ec. (a) Determine the work done by the aluminum block. (b) How much heat energy is transferred during this process? (c) What is the increase in internal energy of the system?
Chapter3: The First Law Of Thermodynamics
Section: Chapter Questions
Problem 97CP: The insulated cylinder shown below is closed at both ends and contains an insulating piston that is...
Related questions
Question
Transcribed Image Text:NOTES
ASK YOUR TEACHER
An aluminum block of mass 7.6 kg is originally at a temperature of 20°C and 1 atm. It is then heated to a temperature of 59°C without any change in pressure. The specific heat of
aluminum is 900 J/(kg · °C), its density is 2.70 x 103 kg/m3, and the coefficient of linear expansion is 24 x 10 /°C.
(a) Determine the work done by the aluminum block.
(b) How much heat energy is transferred during this process?
(c) What is the increase in internal energy of the system?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

