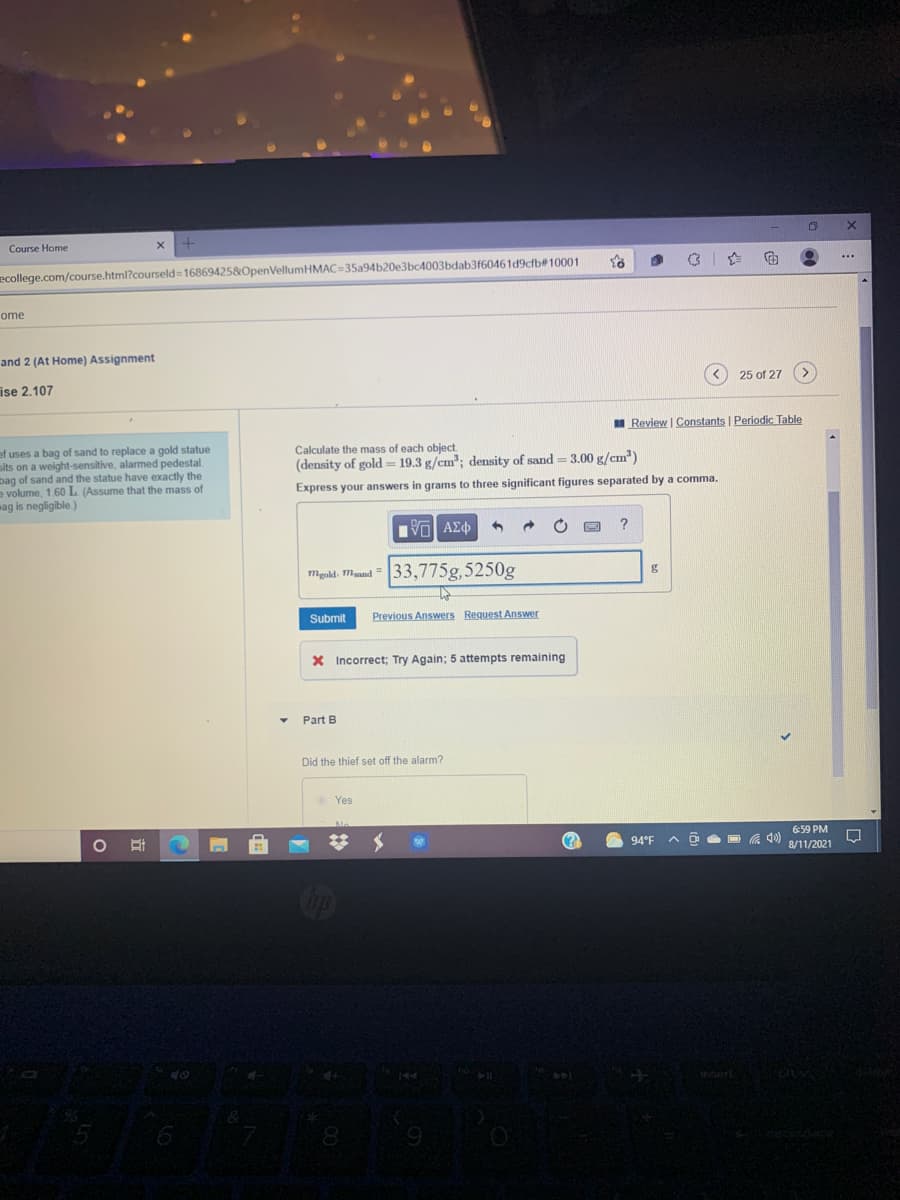
and 2 (At Hom ise 2.107 25 of 27 I Review | Constants | Periodic Table ef uses a bag of sand to replace a gold statue sits on a weight-sensitive, alarmed pedestal. bag of sand and the statue have exactly the a volume, 1.60 L (Assume that the mass of wag is negligible) Calculate the mass of each object. (density of gold- 19.3 g/cm"; density of sand = 3.00 g/cm") %3D %3D Express your answers in grams to three significant figures separated by a comma. mgakd. Mand " 33,775g,5250g Submit Previous Answers Request Answer
and 2 (At Hom ise 2.107 25 of 27 I Review | Constants | Periodic Table ef uses a bag of sand to replace a gold statue sits on a weight-sensitive, alarmed pedestal. bag of sand and the statue have exactly the a volume, 1.60 L (Assume that the mass of wag is negligible) Calculate the mass of each object. (density of gold- 19.3 g/cm"; density of sand = 3.00 g/cm") %3D %3D Express your answers in grams to three significant figures separated by a comma. mgakd. Mand " 33,775g,5250g Submit Previous Answers Request Answer
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter2: Atoms, Molecules, And Ions
Section: Chapter Questions
Problem 90QAP: Calculate the average density of a single Al-27 atom by assuming that it is a sphere with a radius...
Related questions
Question
density of gold = 19.3 g/cm density of sand = 3.00 g/cm
Transcribed Image Text:Course Home
ecollege.com/course.html?courseld= 16869425&OpenVellumHMAC=35a94b20e3bc4003bdab3f60461d9cfb#10001
ome
and 2 (At Home) Assignment
25 of 27
ise 2.107
I Review | Constants | Periodic Table
ef uses a bag of sand to replace a gold statue
sits on a weight-sensitive, alarmed pedestal.
bag of sand and the statue have exactly the
e volume, 1.60 L (Assume that the mass of
wag is negligible)
Calculate the mass of each object.
(density of gold = 19.3 g/cm; density of sand = 3.00 g/cm")
Express your answers in grams to three significant figures separated by a comma.
Πνα ΑΣφ
33,775g,5250g
mgold. mnd
Submit
Previous Answers Request Answer
X Incorrect; Try Again; 5 attempts remaining
Part B
Did the thief set off the alarm?
O Yes
%23
6:59 PM
6- a q0 8/11/2021
94°F
Cip
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax