Answer questions a-c about the Bronsted acid-base reaction below using the identifying letters A-D below each structure. The pka's for the acids of interest are: dimethylsulfone (pK₂ = 28), and ethanol (pka = 16.0). H₂ HO-CH₂ CH₂ CH3- -CH₂ -CH₂ НО CH3- D B A ethoxide с ethanol dimethylsulfone dimethylsulfone anion a) The stronger base is b) Its conjugate acid is c) The species that predominate at equilibrium are (two letters, e.g. ac)
Answer questions a-c about the Bronsted acid-base reaction below using the identifying letters A-D below each structure. The pka's for the acids of interest are: dimethylsulfone (pK₂ = 28), and ethanol (pka = 16.0). H₂ HO-CH₂ CH₂ CH3- -CH₂ -CH₂ НО CH3- D B A ethoxide с ethanol dimethylsulfone dimethylsulfone anion a) The stronger base is b) Its conjugate acid is c) The species that predominate at equilibrium are (two letters, e.g. ac)
Chapter20: Carboxylic Acids And Nitriles
Section20.4: Substituent Effects On Acidity
Problem 9P
Related questions
Question
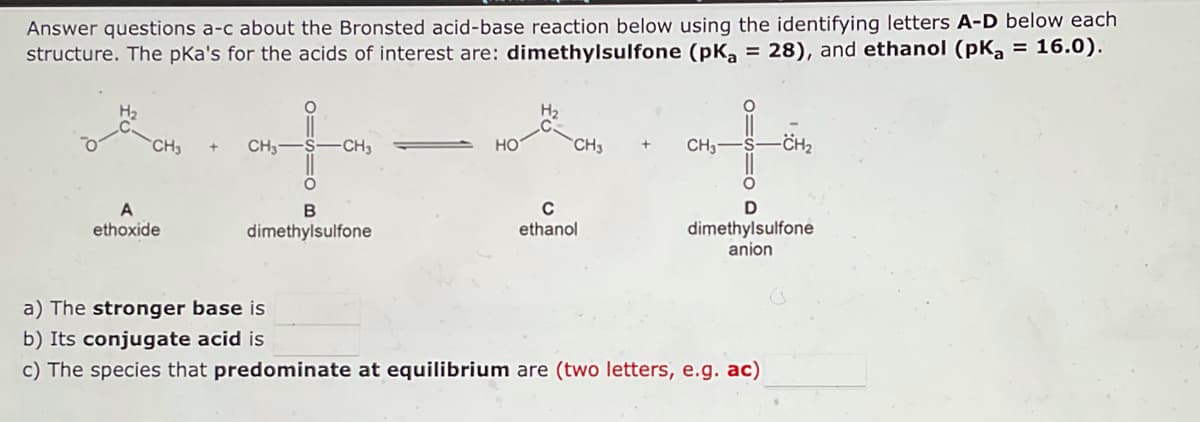
Transcribed Image Text:Answer questions a-c about the Bronsted acid-base reaction below using the identifying letters A-D below each
structure. The pka's for the acids of interest are: dimethylsulfone (pK₂ = 28), and ethanol (pK₂ = 16.0).
HO-CH₂
CH₂
CH3-
-CH3
НО
-CH₂
CH3-
D
B
A
ethoxide
с
ethanol
dimethylsulfone
dimethylsulfone
anion
a) The stronger base is
b) Its conjugate acid is
c) The species that predominate at equilibrium are (two letters, e.g. ac)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

