Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter3: Measurement And Chemical Calculations
Section: Chapter Questions
Problem 93E
Related questions
Question
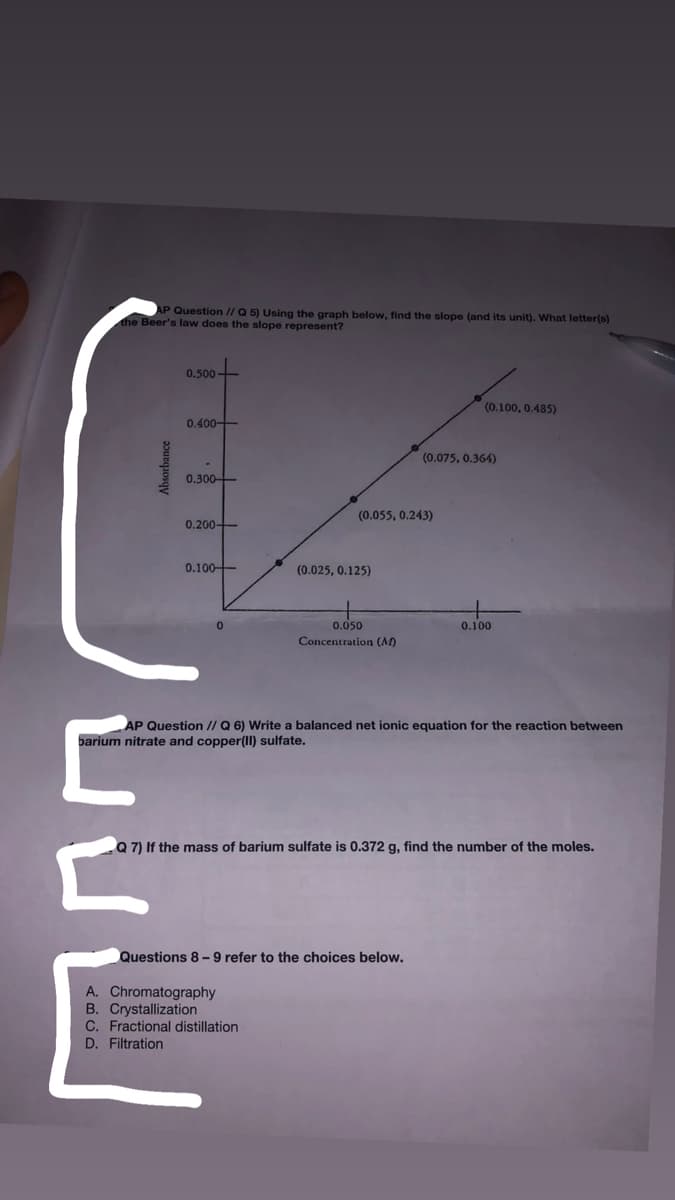
Transcribed Image Text:AP Question // Q 5) Using the graph below, find the slope (and its unit). What letter(s)
the Beer's law does the slope represent?
0.500
(0.100, 0.485)
0.400-
(0.075, 0.364)
0.300-
(0.055, 0.243)
0.200-
0.100-
(0.025, 0.125)
0.050
0.100
Concentration (M)
AP Question // Q 6) Write a balanced net ionic equation for the reaction between
barium nitrate and copper(II) sulfate.
Q 7) If the mass of barium sulfate is 0.372 g, find the number of the moles.
Questions 8- 9 refer to the choices below.
A. Chromatography
B. Crystallization
C. Fractional distillation
D. Filtration
Absorbance

Transcribed Image Text:8. Method to isolate ethanol from ethanol and water mixture
9. The different colors in black ink
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning