Q: Which one of these molecules is the least soluble in water? CH3-CH2-CH2-CH2-CH2 OH CH3-CH-CH3…
A: The given compounds are alcohols. The alcohols are soluble in water as they form hydrogen bonding…
Q: 1. The following are solvents used in Biochemistry Laboratory. Lipids are expected to dissolve in?…
A:
Q: Explain why beeswax is insoluble in H2O, slightly soluble in ethanol (CH3CH2OH), and soluble in…
A: Beeswax is insoluble in water, slightly soluble in ethanol, and soluble in chloroform.
Q: what intermolecular forces Allura red (C18H14N2Na2O8S2) have 1: with distill water 2: with ethyl…
A:
Q: Explain why beeswax is insoluble in H2O, slightly soluble in ethanol (CH3CH2OH), and soluble in…
A: Beeswax is mainly consisting of lipids in which two long alkyl groups are joined together by a…
Q: what intermolecular forces Brillant blue (C37H34N2Na2O9S3) have 1: with distill water 2: with…
A: (C37H34N2Na2O9S3) is a synthetic dye.
Q: Is the following structure soluble in water? Yes or No? H3C-CH2-0-CH3
A:
Q: Are the London dispersion forces the same for all molecules or are they different and why?…
A: The forces of attraction that exist between atoms of the different molecules (or two same molecules)…
Q: Draw (using skeletal form) the chemical equation for the INTERMOLECULAR dehydration of 1-ethanol at…
A:
Q: 1. List the reasons why pentanol has a higher boiling point when compared to its corresponding…
A: Pentanol is an alcohol which means it contains a hydroxyl group (-OH) in its structure. On the other…
Q: Which compound has higher solubility in water? A polar compound or a nonpolar compound? A compound…
A: We'll answer the first question since the exact one wasn't specified. Please submit question…
Q: Circle one of the following substances which would be most soluble in H2O?
A:
Q: CH3-CH2-C-CH3 Which intermolecular attraction is present in the solvent MEK, whose structure is…
A: The intermolecular forces of attraction operate between the atoms which include the force of…
Q: Which of the following would be the most soluble in water? a) CH3(CH2);CH2OH b) CH3(CH2)½CH2OH c)…
A: The given compounds are alcohols. Alcohols form H-bonding with water molecules. Thus they are…
Q: those that do hydrogen bond, draw a diagram of two molecules using a dotted line to indicate where…
A:
Q: what intermolecular forces cause ethyl acetate to be insoluble in water, but soluble in hexane?
A: The rule is that : like dissolve like. The predominant intermolecular force present in ethyl acetate…
Q: OH
A: Carboxylic acid has strongest boiling acid as it show maximum hydrogen bonding and thus show strong…
Q: Give the IUPAC name or common name of the isomer of 1-hexanol that has the highest vapor pressure.
A: Given statement is : Give the IUPAC name or common name of the isomer of 1-hexanol that has the…
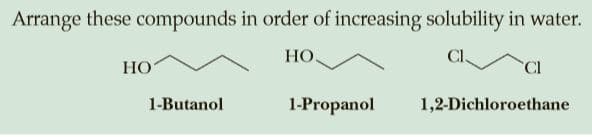
Q: Arrange these compounds in order of increasing solubility in water.
A:
Q: What kind of solvent ingredients is usually used in the concentrations of 4-10 percent in skin care…
A: Some chemical are used in skin care product ;some of their function is to soften the skin cell and…
Q: What is being broken apart when ethanol, CH3CH2OH, dissolved in water to make liquor?
A:
Q: other don't
A: This question is related to dissolution.
Q: Write the letter of the following compound(s) that fit the criteria
A: The molecule which will have H present in the molecule connected to the high electronegative atoms…
Q: Which one has the highest boiling point? O propane ethanol diethylether O l-propanol O Chloroethane
A: Given Propane Ethanol Diethylether 1-Propanol Chloroethane Highest Boiling point = To be…
Q: Which of the following would be MOST soluble in water? CH3CH2CH2CH2CH2NH2 All of these would have…
A:
Q: Which of the following would be MOST soluble in water? Select one: A. chloropropane B. ethanol C.…
A: As we know, Water is a polar molecule . And , we know polar dissolve polar and non- polar dissolve…
Q: Following are three alcohols with the molecular formula C4H10O. Their boiling points, from lowest to…
A: SOLUTION: Step 1: The boiling points of alcohols are much higher than those of alkanes with similar…
Q: In the previous problem, you compared the water solubility of the following molecules. H2N. HO, H.…
A: Intermolecular forces refer to the type of interactions which are present in between atoms or…
Q: Regarding t-butanol and n-butanol, which is/ are correct statements of the following |- Both are…
A: SOLUBILITY tert butanol have higher solubility than n butanol. We can interpret the reason as water…
Q: Predict which of the following molecules will have the least water solubility. OH OH Но, CH3 H3C…
A: Solubility in water depends upon various factors like the composition of the substance being…
Q: Which of the following situations will alcohol be at the lowest percentage? A. Equal amounts of…
A: Alcohol can be defined as an compound in which a hydroxyl group is bound to a carbon atom
Q: Soluble in: Cold Hot 3M 3M water HCI NaOH water Yes No Benzoic acid Mg(OH)2 NazSO4 Zn(OH)2 No No Yes…
A: Answer According to given table Mg(OH)2 does not react with 3M HCl…
Q: OH I II III IV
A: Solubility depends upon the effectiveness of hydrogen bonding.
Q: (a) Which of the following molecules can hydrogen bond to another molecule like itself? (b) Which of…
A: a) The molecules which will have H attached to high electronegative elements like O, N, Cl, and F…
Q: Which of these five-carbon aloohols would you expect to be most water soluble? OH OH OH A в D OA
A: Alcohols are soluble in water because they form intermolecular hydrogen bonding with water…
Q: Explain the intermolecular forces between water and cyclohexane?
A:
Q: At 25°C, the vapor pressure of diethyl ether, (CH3CH2)2O, is higher than the vapor pressure of its…
A: Given that, At 25°C, the vapor pressure of diethyl ether, (CH3CH2)2O, is higher than the…
Q: Draw a diagram to explain Hydrogen bonding between two ethanol molecules.
A: Hydrogen bonding is a special type of dipole - dipole interaction between molecules, not a covalent…
Q: (1) Isopropyl alcohol and 1-propanol are two names for the same compound. (2) Ether molecules cannot…
A:
Q: Rank the following compounds in order of increasing boiling point.
A: The extent of a boiling point depends upon the molecular forces existing among the molecules. Higher…
Q: Define Solubility of Alcohols ?
A: Alcohols are the organic compounds which contains the -OH functional group. The general formula of…
Q: Which of the following compounds cannot form hydrogen bonds with water? CH3-CH2-CH3 0 0 CH3C -O-CH3…
A: Each molecule of H2O has two hydrogen atoms and two oxygen atoms with two lone pairs of electrons.…
Q: Arrange the following compounds in the order of increasing solubility in water (least first).…
A:
Q: Based on intermolecular forces, predict the ordering from LOWEST boiling point to HIGHEST boiling…
A: compounds boiling point in degree celcius CH3CH2CH2OH 97 Ne -246 CH3COCH3 56 CH4 -161…
Q: Arrange the following alcohols by increased boiling point: a.) 1-hexanol, b.) 1-pentanol, c.)…
A: Boiling point of alcohols is directly proportional to molecular mass of alcohols. Boiling point of…
Q: Choose the water solube molecules H -OH он
A:
Q: Name and describe the intermolecular forces that are responsible for methylated spirits to dissolve…
A:
Q: Rank the attached compounds in order of increasing boiling point. Which compound is the most water…
A: All the given compounds have polar nature and they show dipole-dipole interactions. The hydrogen…
Q: Boiling point of Ethyl acetate 2-propanol What’s sodium benzoate solubility in water?
A: Solubility refers to the ability of a substance (solute) to dissolve in a liquid (solvent) to form…
Q: Arrange the following compounds in order of increasing boiling point. Clear All CH;CH2CH2C-H Lowest…
A: Increasing order of boiling point: CH3CH2CH2CH2CH3 < CH3CH2CH2CHO < CH3CH2COOH…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Explain why ethanol (CH3CH2OH) is more soluble in water than is ethane (CH3CH3 )List each set of a compound in order of increasing boiling point.a) hexane, octane, and decanewhat intermolecular forces Allura red (C18H14N2Na2O8S2) have 1: with distill water 2: with ethyl acetate 3: with cyclohexane 4: with 2-propanol
- Among CH₃OH, HOCH₂CH₂OH, CH₃CH₂OCH₂CH₃, which will have the highest vapor pressure?Which would be the most important type of intermolecular force between fat molecules and petroleum ether? Explain your reasoning. Also note that petroleum ether is a mix of hydrocarbons (C6-16H12-34).Rank the attached compounds in order of increasing boiling point. Which compound is the most water soluble? Which compound is the least water soluble?

