as an Ideal gas at temperatures above Its boiling point of -24. °C. For many purposes we can treat dimethyl ether (C, H,O) Suppose the pressure on a 310. mL sample of dimethyl ether gas at -14.0°C Is doubled. O yes Is It possible to change the temperature of the dimethyl ether at the same time such that the volume of the gas doesn't change? O no If you answered yes, calculate the new temperature of the gas. Round your answer to the nearest °C. °C
as an Ideal gas at temperatures above Its boiling point of -24. °C. For many purposes we can treat dimethyl ether (C, H,O) Suppose the pressure on a 310. mL sample of dimethyl ether gas at -14.0°C Is doubled. O yes Is It possible to change the temperature of the dimethyl ether at the same time such that the volume of the gas doesn't change? O no If you answered yes, calculate the new temperature of the gas. Round your answer to the nearest °C. °C
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter8: Properties Of Gases
Section: Chapter Questions
Problem 8.BCP
Related questions
Question
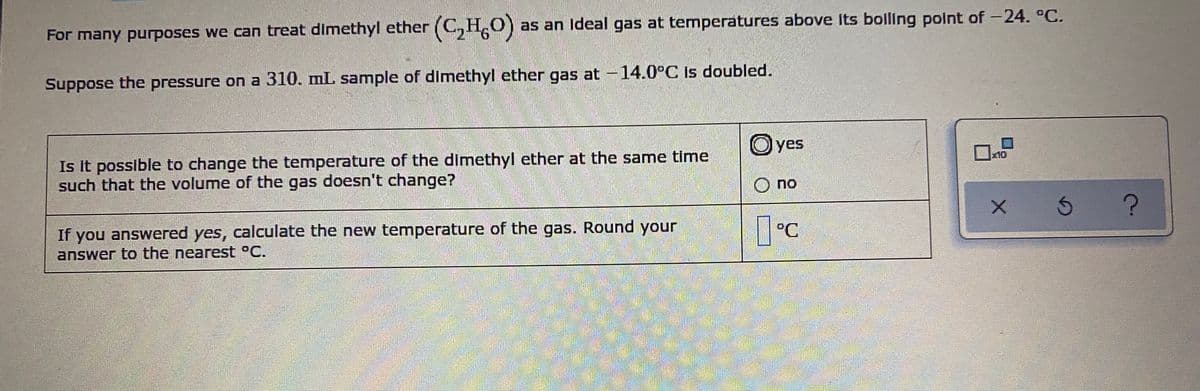
Transcribed Image Text:For many purposes we can treat dimethyl ether (C,H,0)
as an Ideal gas at temperatures above its bolling point of -24. °C.
9.
Suppose the pressure on a 310. mL sample of dimethyl ether gas at -14.0°C is doubled.
Oyes
Is it possible to change the temperature of the dimethyl ether at the same time
such that the volume of the gas doesn't change?
X10
O no
If you answered yes, calculate the new temperature of the gas. Round your
answer to the nearest °C.
°C
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning