As shown in the figure below, for a given change in volume expansion, the higher the temperature of the gas, the larger the exported work. Work, -w/RT 5 1 O True Increasing 37 temperature 3 Expansion, V/V VIV 5
As shown in the figure below, for a given change in volume expansion, the higher the temperature of the gas, the larger the exported work. Work, -w/RT 5 1 O True Increasing 37 temperature 3 Expansion, V/V VIV 5
Chapter3: The First Law Of Thermodynamics
Section: Chapter Questions
Problem 90AP: A cylinder containing three moles of a monatomic ideal gas is heated at a constant pressure of 2...
Related questions
Question
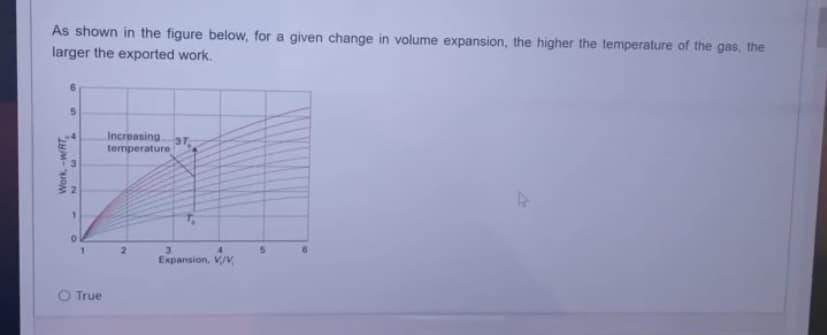
Transcribed Image Text:As shown in the figure below, for a given change in volume expansion, the higher the temperature of the gas, the
larger the exported work.
Work, -w/RT
5
1
O True
Increasing 37
temperature
3
Expansion, V/V,
VIV
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

