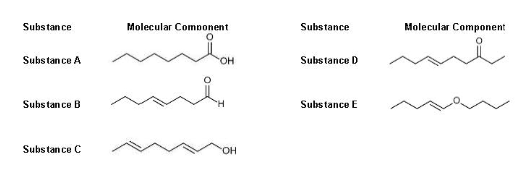
Aside from London dispersion forces, which substance do you expect dipole interactions to be effective? a. Substance A and B b. Substance B and D c. Substance A and C d. Substance B and E -Which substances can react with potassium borohydride, converting the product into alkene alcohol (assuming complete reaction)? a. Substance A and B b. Substance B and D c. Substance C and D d. Substance D and E -The presence of trans pi bonds can influence molecular stacking due to the limitation of bond rotation. Based on this information, which substance is characterized by molecules that have effective molecular stacking? a. Substance A b. Substance B c. Substance C d. Substance D e. Substance E
-Aside from London dispersion forces, which substance do you expect dipole interactions to be effective?
a. Substance A and B
b. Substance B and D
c. Substance A and C
d. Substance B and E
-Which substances can react with potassium borohydride, converting the product into
a. Substance A and B
b. Substance B and D
c. Substance C and D
d. Substance D and E
-The presence of trans pi bonds can influence molecular stacking due to the limitation of bond rotation. Based on this information, which substance is characterized by molecules that have effective molecular stacking?
a. Substance A
b. Substance B
c. Substance C
d. Substance D
e. Substance E
-Which substance CANNOT decolorize bromine water?
a. Substance A
b. Substance B
c. Substance C
d. Substance D
e. Substance E
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images







