Assume all gases in this problem are ideal. What is the total translational kinetic energy of the air molecules (in J) in a room of volume 26 m³ if the pressure is 9.3 X 105 Pa (the room is at fairly high elevation) and the temperature is 22°C? J Is any item of data unnecessary for the solution? (Select all that apply.) volume npressure temperature none of the above
Assume all gases in this problem are ideal. What is the total translational kinetic energy of the air molecules (in J) in a room of volume 26 m³ if the pressure is 9.3 X 105 Pa (the room is at fairly high elevation) and the temperature is 22°C? J Is any item of data unnecessary for the solution? (Select all that apply.) volume npressure temperature none of the above
Chapter2: The Kinetic Theory Of Gases
Section: Chapter Questions
Problem 42P: What is the total translational kinetic energy of the air molecules in a room of volume 23 m3 if the...
Related questions
Question
please answer v
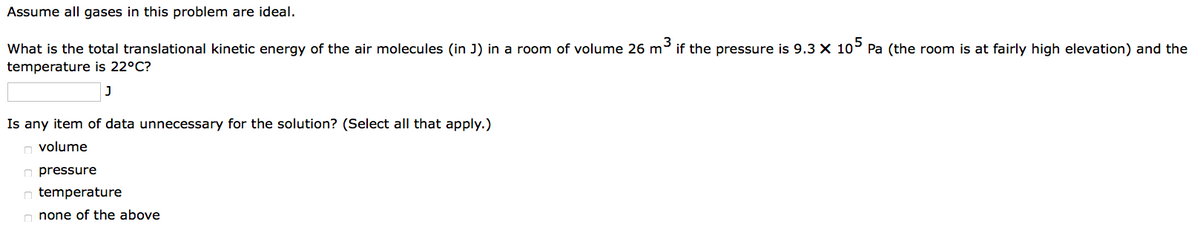
Transcribed Image Text:Assume all gases in this problem are ideal.
What is the total translational kinetic energy of the air molecules (in J) in a room of volume 26 m³ if the pressure is 9.3 X 105 Pa (the room is at fairly high elevation) and the
temperature is 22°C?
J
Is any item of data unnecessary for the solution? (Select all that apply.)
volume
pressure
temperature
none of the above
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you


University Physics Volume 1
Physics
ISBN:
9781938168277
Author:
William Moebs, Samuel J. Ling, Jeff Sanny
Publisher:
OpenStax - Rice University

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning


University Physics Volume 1
Physics
ISBN:
9781938168277
Author:
William Moebs, Samuel J. Ling, Jeff Sanny
Publisher:
OpenStax - Rice University

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning