At 800 K hydrogen iodide can decompose into hydrogen and iodme gases 2HIg)2)+ H2g) At this temperature, Kp 0.0169. What is the partial pressure of hydrogen iodide, HI, if initially a sealed flask at 800 K contains only Hi at a pressure of 0.200 atm? 2.06 x 10 atm 1.59x 10 atm 1.79x 10 atm 1.30x 10 atm none of these
At 800 K hydrogen iodide can decompose into hydrogen and iodme gases 2HIg)2)+ H2g) At this temperature, Kp 0.0169. What is the partial pressure of hydrogen iodide, HI, if initially a sealed flask at 800 K contains only Hi at a pressure of 0.200 atm? 2.06 x 10 atm 1.59x 10 atm 1.79x 10 atm 1.30x 10 atm none of these
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter13: Fundamental Equilibrium Concepts
Section: Chapter Questions
Problem 12E: Show that the complete chemical equation, the total ionic equation, and the net ionic equation for...
Related questions
Question
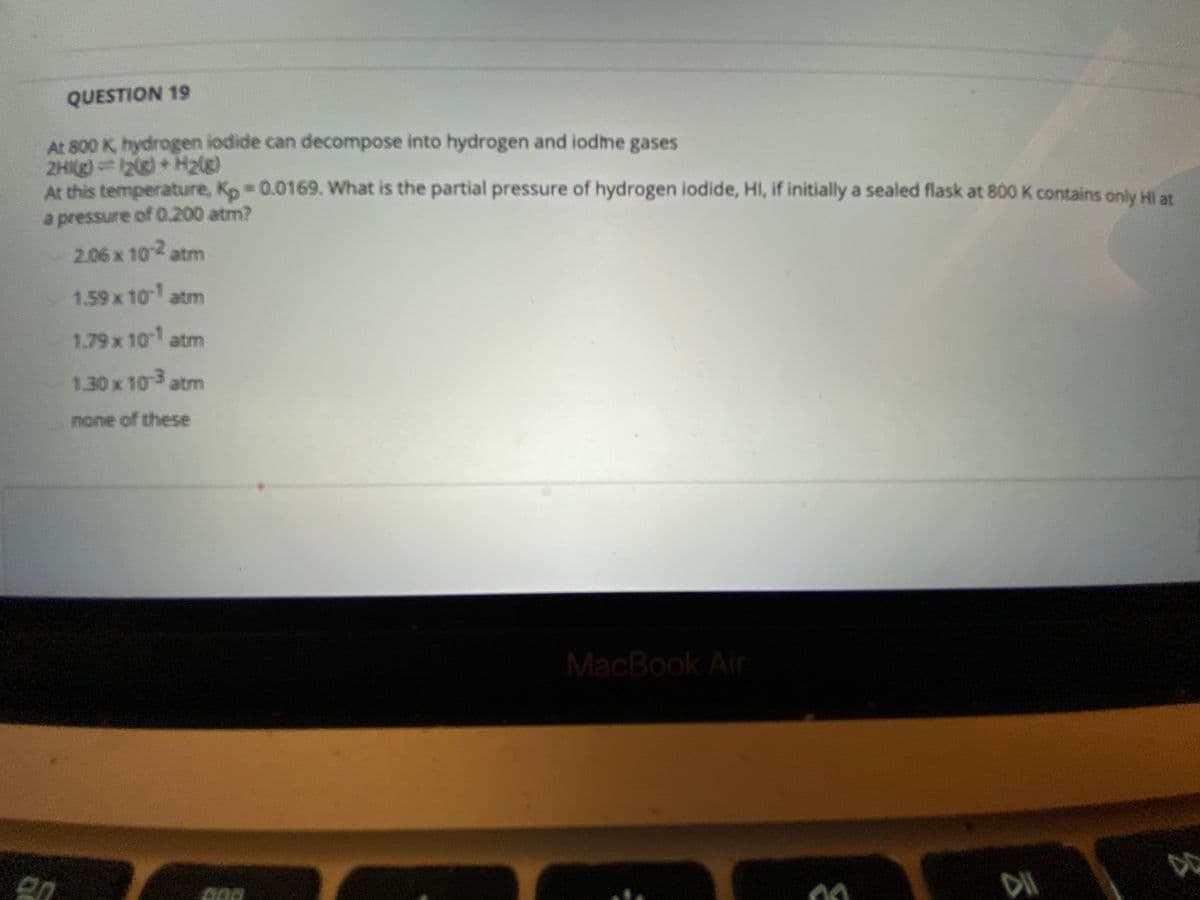
Transcribed Image Text:QUESTION 19
At 800 K, hydrogen iodide can decompose into hydrogen and iodme gases
2HI(g)2) + H2(g)
At this temperature, Kp 0.0169. What is the partial pressure of hydrogen iodide, HI, if initially a sealed flask at 800 K contains only Hi at
a pressure of 0.200 atm?
2.06 x 10 atm
1.59 x 10 atm
79x.ו10 am
1.30x 10 atm
none of these
MacBook Air
DO
DII
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax