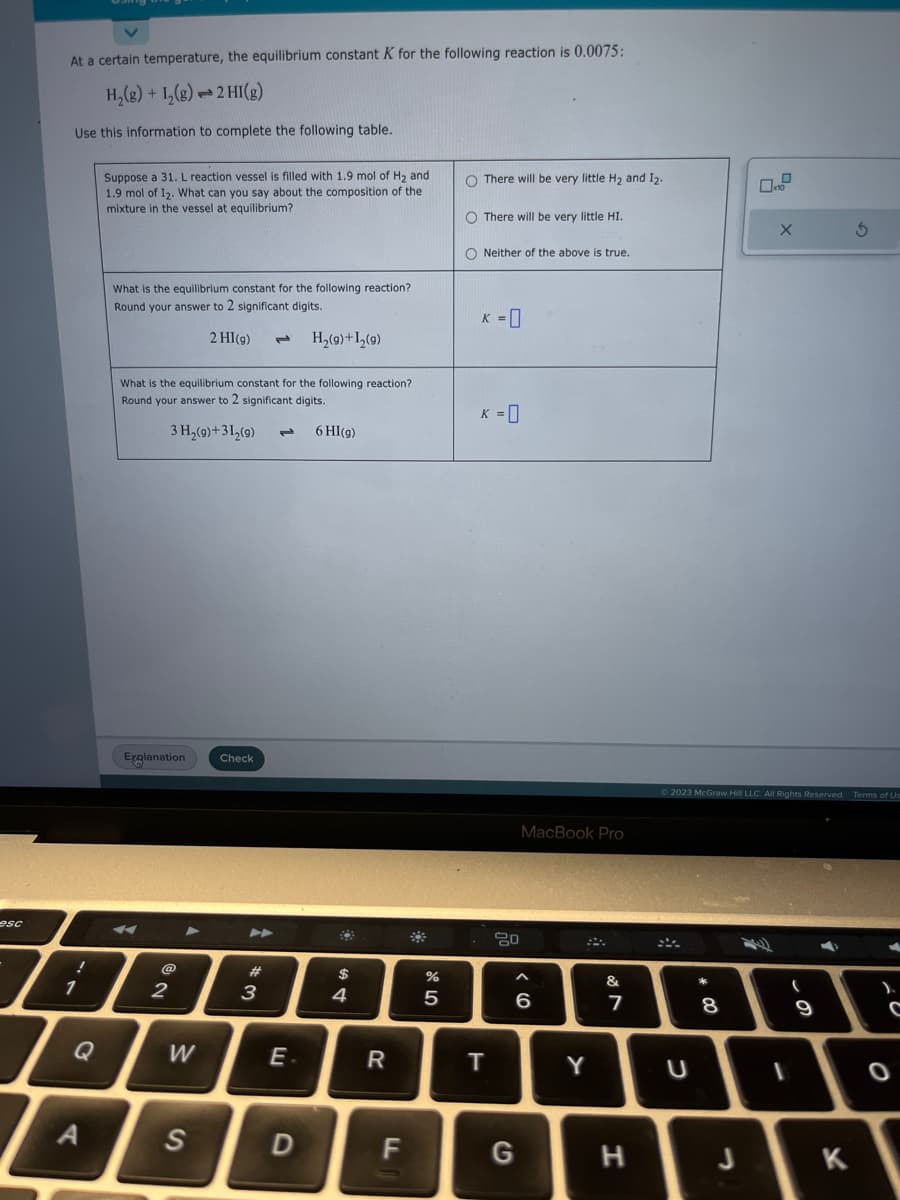
At a certain temperature, the equilibrium constant K for the following reaction is 0.0075: H₂(g) + 1₂(g) 2 HI(g) Use this information to complete the following table. Suppose a 31. L reaction vessel is filled with 1.9 mol of H₂ and 1.9 mol of 12. What can you say about the composition of the mixture in the vessel at equilibrium? What is the equilibrium constant for the following reaction? Round your answer to 2 significant digits. 2 HI(g) H₂(9)+1₂(9) What is the equilibrium constant for the following reaction? Round your answer to 2 significant digits. 3 H₂(9)+31₂(9) 6 HI(g) O There will be very little H₂ and 1₂. O There will be very little HI. O Neither of the above is true. K = 0 K = 0
At a certain temperature, the equilibrium constant K for the following reaction is 0.0075: H₂(g) + 1₂(g) 2 HI(g) Use this information to complete the following table. Suppose a 31. L reaction vessel is filled with 1.9 mol of H₂ and 1.9 mol of 12. What can you say about the composition of the mixture in the vessel at equilibrium? What is the equilibrium constant for the following reaction? Round your answer to 2 significant digits. 2 HI(g) H₂(9)+1₂(9) What is the equilibrium constant for the following reaction? Round your answer to 2 significant digits. 3 H₂(9)+31₂(9) 6 HI(g) O There will be very little H₂ and 1₂. O There will be very little HI. O Neither of the above is true. K = 0 K = 0
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter12: Chemical Equilibrium
Section: Chapter Questions
Problem 60QRT
Related questions
Question
100%
Please use correct significant digits (in this case it is 2), and use scientific notation, if needed.
Transcribed Image Text:esc
At a certain temperature, the equilibrium constant K for the following reaction is 0.0075:
H₂(g) + 1₂(g) → 2 HI(g)
Use this information to complete the following table.
1
Q
A
Suppose a 31. L reaction vessel is filled with 1.9 mol of H₂ and
1.9 mol of 12. What can you say about the composition of the
mixture in the vessel at equilibrium?
What is the equilibrium constant for the following reaction?
Round your answer to 2 significant digits.
2 HI(g) 1 H₂(g) + 1₂ (9)
What is the equilibrium constant for the following reaction?
Round your answer to 2 significant digits.
3 H₂(9)+31₂(9)
6 HI(g)
Explanation
2
W
S
Check
3
E.
D
*
$
4
R
FL
do 5
%
O There will be very little H₂ and 1₂.
O There will be very little HI.
ONeither of the above is true.
K = 0
K = 0
T
MacBook Pro
20
<CO
6
G
Y
&
7
H
U
© 2023 McGraw Hill LLC. All Rights Reserved. Terms of Us
00
8
X
J
1
(
9
4
S
K
C
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps with 15 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning