Q: What mass of Li3P would you measure out to make 2.6 liter of 5.7 M Li3P solution?
A:
Q: 90 NANO 80 70 Pb(NO)2 KNOS 60 CaC 50 O 40 Nacl 30 KCI 20 KCIOS 10 Ce,(SO 0 10 20 30 40 50 60 70 80…
A: We have to find out the salt (solute) which is least soluble at 0℃
Q: I want to make 500 mL of HCl solution with MHCl concentration = 0.1 moL/L from HCl solution which…
A: Given that - Volume of HCl Solution = 500 mL Concentration of this Solution = 0.1 mol/L = 0.1 M…
Q: (a) A solution is made by dissolving 13.5 g of glucose (C6H12O6) in 0.100 kg of water. What is the…
A: a) Since mass percentage is defined as the percentage of solute in the solution i.e % m/m = mass of…
Q: You have 125.0mL of 4.10M aqueous Mg(CIO3)2. Which of the following statements are true? O Mg(CIO3)2…
A: Which of the following statement is/are true ?
Q: If 1.50 mL of a stock solution of 12.0 M HCl is diluted to 25.0 mL, what is the new concentration?
A:
Q: What volume of 6.0 M H2SO4 is needed to prepare 500.0 mL of a solution that is 0.25 M in H2SO4?
A: On dilution of a substance the number of moles of solute always remains constant. The number of…
Q: 2.2 A concentrated perchloric acid solution has a density of 1.67 g/mL at 25°C and is 11.7 M. What…
A: Perchloric acid is a strong monoprotic acid. It readily deprotonates in aqueous solution.
Q: If water is added to 150 mL of a 0.25 M naOH solution until the final volume is 250mL, what will the…
A: Dilution of the solution: When we dilute a concentrated solution with pure water, the mole of solute…
Q: 1. a) What volume of 8.40 N Al(OH)3 will be used to prepare 350.0 mL of 2.10 M solution? b) How many…
A: Solution -
Q: Heat of Neutralization = Q/moles of limiting reactant = Q/moles of NaOH titrated at neutralization =…
A:
Q: ) Solution Concentration and Solution Stoichiometry a) What is the molarity of a solution made by…
A:
Q: In order to prepare 10% 100 mL of HCl solution from 37% HCl solution, how many mL of concentrated…
A: For Concentrated HCl solution : Concentration, C1 = 37 % Volume, V1 =? For New HCl solution :…
Q: You have 125.0mL of 4.10M aqueous Mg(CIO3)2. Which of the following statements are true? O Mg(CIO3)2…
A:
Q: 1. Preparation of hydrochloric acid solution To perform the work, 500 ml of hydrochloric acid…
A: Given: Volume of HCl (V1) = 500 mL Concentration of HCl (C1) = 7.449 mol/L Concentration of HCl (C3)…
Q: 1)lf 25mL of a 1.5M KOH solution is mixed with 15mL of a 0.5M H3PO4 solution, how many grams of…
A:
Q: Alcoholic beverages come in many different concentrations denoted by their weight percent (w/w). If…
A:
Q: 12. What is the molarity of a solution containing 1.4mole of acetic acid (HC2H3O2(aq)) in 250.0mL of…
A: Hey, since there are multiple questions posted, we will answer first question. If you want any…
Q: The stock solution of 13.60 M NH4OH is used to make 500.00 mL of a 0.001000 M solution. How many mL…
A:
Q: Q27 (C) 200 grams of K2SO3 is mixed with water to make a 0.75 M solution. How much volume (ml) of…
A:
Q: Analytical Chemistry Q5- Calculate the number of grams of solute in: a - 1 L of 0.2 N Ba(OH)2…
A: it is defined as number of gram equivalent divided by volume of solution in liter
Q: 90 NANO, 80 70 60 CaCl 50 Pb(NO,)2 40 NaCi 30 KCI 20 KCIO, 10 Ce,(SOJa O 10 20 30 40 50 60 70 80 90…
A: Given data: The temperature of a saturated solution of Pb(NO3)2 = 30°C. The mass of Pb(NO3)2 that is…
Q: A 32.5 mL sample of an 5.6 % (m/v) KBr solution is diluted with water so that the final volume is…
A: We have to predict the final concentration.
Q: A solution of 250.0 mL of a 0.200 M NaHCO3 solution is to be prepared. The solution is diluted by…
A: In the given problem, the molarity of the serially diluted solution can be calculated by using the…
Q: To 14.8 cm3 of a 1.5 M H2SO4 , enough water is added to produce a 0,18 M H2SO4 solution. How much…
A: During the process of dilution, the number of moles of the solute H2SO4 remains the same. Thus, we…
Q: A solution is prepared by taking 7 ml of 5.9 M HCl and diluting it to 113 ml. Then 15 ml of that…
A: Given that, Volume of initial 5.9M HCl = 7ml
Q: 6 mL of a 1.5 % CuSO4 solution is diluted to 10 mL with pure water, what is the final concentration…
A: NOTE : 1) since density is not given, we have to assume % concentration as % (m/v). 2) Since it is…
Q: b) 1.881x10-3g of copper (II) sulfate in 122mL of solution
A:
Q: 1(a) Define the term concentration. [Takrifkan istilah kepekatan.] 1(b) What is the different…
A:
Q: 150 140 130 120 110 100 NaNO 90 80 70 60 NHC KCI Naci 50 40 30 20 TRCIÓ 10 Cey(so 10 20 30 40 s0 60…
A: The solubility curve is given as,
Q: how many moles of Ca(NO3)2 are there in 75 mL of 0.25M solution?
A:
Q: Calculate the mass (in g) of NaCl needed to make 100.0 mL of a 1.54 M solution (to 3 sig figs).
A: Definition of Molarity- Molarity is the number of moles of solute dissolve in one litre of solution.…
Q: 100 90 80 NANO 70 60 CaCl, Pb(NO.)2 50 40 Naci KCI 30 20 KCIO, 10 Ce,(SOa O 10 20 30 40 50 60 70 80…
A: The solubility graph is : At 50oC the solubility of KNO3 is approximately 88g/ 100 ml of the…
Q: You are standardizing a NaOH solution and find that it requires 21.71mL of NaOH to neutralize 1.589g…
A:
Q: A 20.0 % by mass solution of phosphoric acid (H3PO4) in water has a density of 1.114 g/mL at 20°C.…
A: Molarity is calculated by using formula : molarity = moles of solute / volume of solution ( L) So,…
Q: 5. 1f you diluted 50.0 mL of 0.380 M aluminum sulfate solution to 100.00 ml., what would be the…
A: Given data: Volume V1 = 50.0 mL Molarity M1 = 0.380 M Volume V2 = 100 mL Molarity M2 = ? Formula…
Q: Solution X 100 mL of 0.10 M NaOH(aq) is mixed with 100 mL of 0.10 M HBr(aq) Solution Y 100 mL of…
A: In this question, we have to find out the correct answer of given problem by the help of the…
Q: How many iters of a 0.200 MKI solution is needed to completely react with 243 g of Cu(NO,ccording to…
A:
Q: 38.5.85g NaCI dissolve in water to make 200.mL of volume of solution. Density of solution is found…
A: The Molarity of solution is calculated by the formula, Molarity=Given weightMolecular weight×Volume…
Q: What is the concentration of a solution made by diluting 65 mL65 mL of 6.0 M HCl6.0 M HCl to a final…
A: Given information: Volume of HCl (V1) = 65 mL Concentration of HCl (C1) = 6.0 M New volume of HCl…
Q: 1.) How Many grams of NaOH (MW=40.0) are in 200 ml of a 0.175 M solution of NaOH?
A:
Q: What volume of concentrated 15.0 M nitric acid is required to make 300.0 mL of a 2.50 M nitric acid…
A:
Q: 8. Consider the regular soda solution above as a stock solution that you will use to make 240 mL of…
A: Given: Concentration of Stock solution (regular soda) (M1) = 0.39 M Concentration of working…
Q: 6.00 mol of C6H12O6 in 250.0 mL of solution M
A:
Q: 2- Calculate the weight of solute in grams for each of the followings solutions: (a) 43.5 mL of…
A: Here i have calculated the weight of solute in grams
Q: 100 90 NANO, 80 70 CaCl, Pb(NO)2 60 50 40 NaCI 30 KCI 20 KCIO, 10 Ce,(SO,) O 10 20 30 40 50 60 70 80…
A: The solubility graph is given below :
Q: What volume of 3.25 mol/al stock solution is needed to make 2.5L of 1.50 mol/L solution?
A: Given: Stock solution concentration = 3.25 mol/L. Final solution concentration = 1.50 mol/L. And…
Q: 1. A 0.10 M HNO3 solution will be prepared from concentrated HNO3. a. Using the method outlined in…
A: Given, density =1.42 g/ml 70% by mass HNO3 is given Molarity =? Molar mass of HNO3 = 63g/mol
Q: When 20 grams of potassium chlorate, KCIO3, is dissolved in 100 grams of water at 80 °C, the…
A:
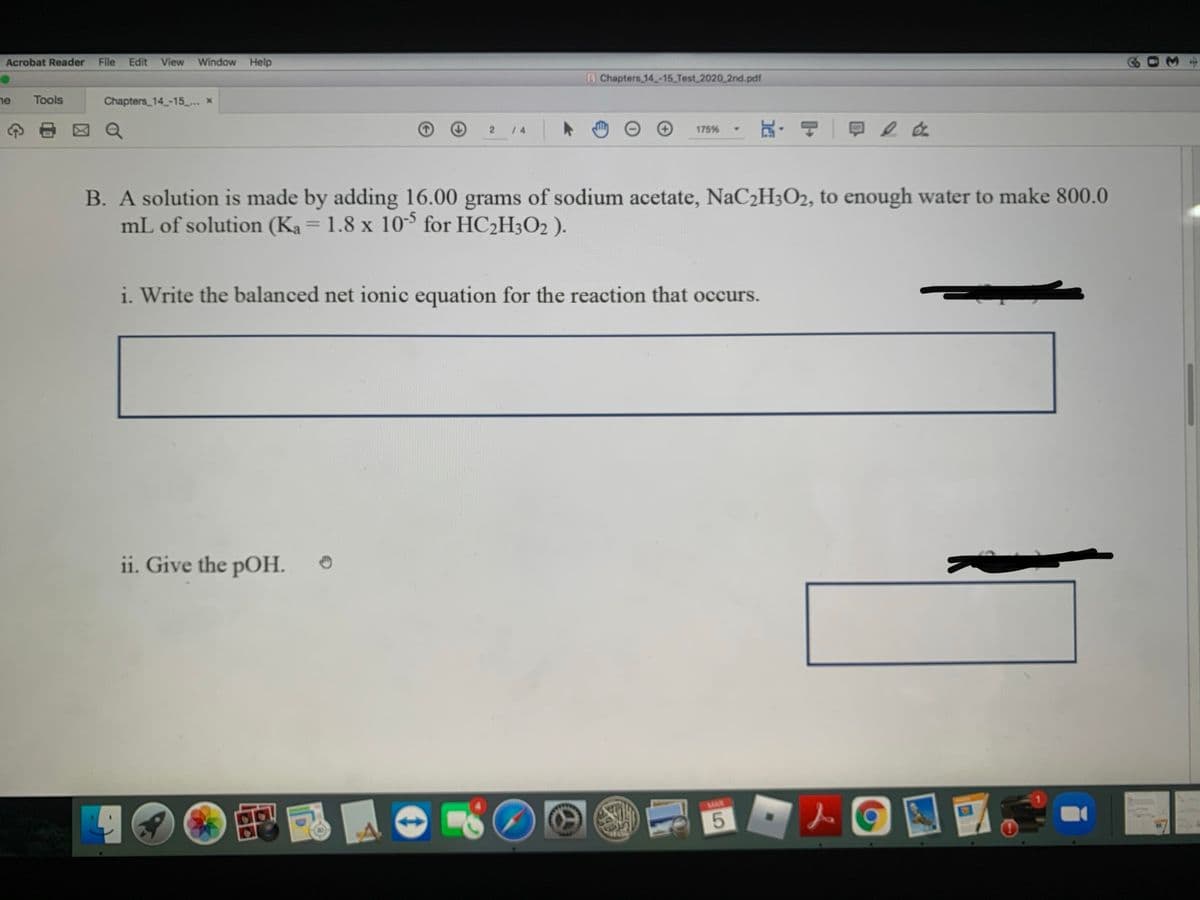
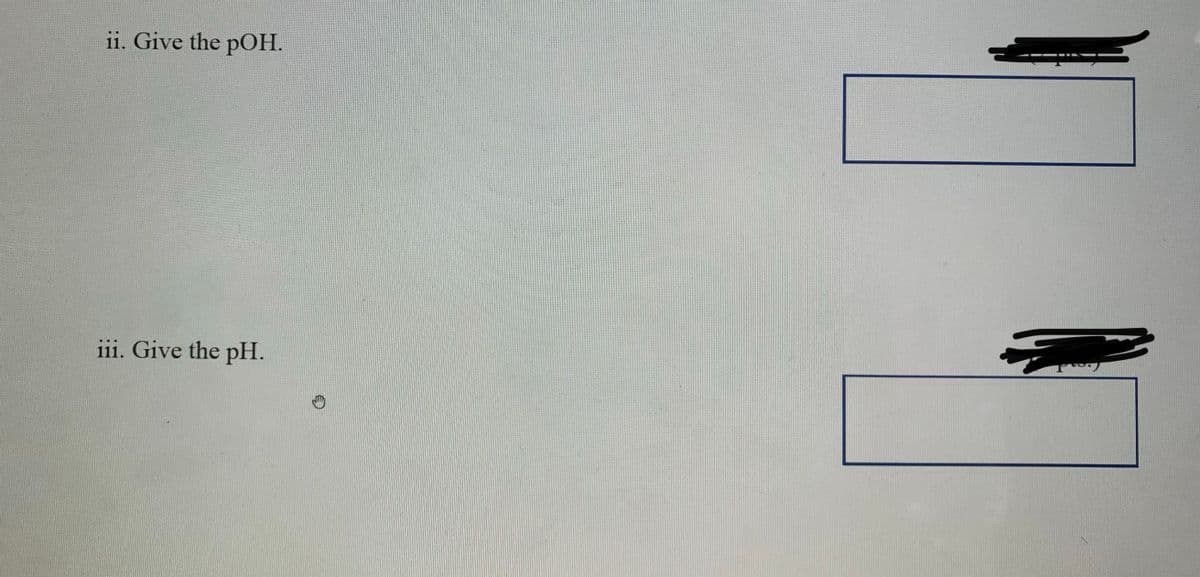
Step by step
Solved in 2 steps

- how many grams of barium carbonate BaCO3 are dissolved in: a) 1 liter of clean water b) 1 liter of solution 0,1 N Ba (NO3) 2 c) 300 ml of 0.01N BaCl2 solution during the calculations take into account that Ksp of BaCO3 = 4 × 10-⁹In which situation(s) is it necessary to use a volumetric flask for maximum accuracy? to prepare a 0.150M solution of sodium chloride to measure ~25mL of water to prepare 0.1M HCI from 36-38% HCI a and c all of the above (Please type answer no write by hend)1. Calculate the mass required to prepare a 250 mL 3 M NaCl in water.Mass = M x MM x V 10002. Calculate how to prepare the following dilutions, ½, ¼, 1/8 and 1/16 of the initial concentration.There is a helpful equation that is used to determine how much stock solution and how much diluent to combine to get the final solution of a desired concentration
- Prepare 0.1 M solutions of NaOH and 0.1 M ethyl acetate using high-purity distilled water. So, weight desired amount of NaOH and ethyl acetate and dissolved in dH2O to prepare stock solution in equal molarity. Mw (NaOH) = 40.0 g/mol , Mw (EtOAc)= 88.1 g/mol, Density(EtOAc): 0.898 g/cm3The first goal is to make the oxalic acid standard solution. You measure 1.5232 g of oxalic acid on an analytical balance, add it to a 250-mL volumetric flask and add deionized H2O to a final volume of 250.0 mL. Molar mass of H2C2O4•2H2O = 126.07 g/mol Mass of H2C2O4•2H2O = 1.5232g what is the Number of moles of H2C2O4•2H2O?1) How much total sea salt is present in a 95 Kg person? (a normal person (70 kg BW) has approximately 43L body fluid). 2) Please express the normal blood glucose level into molarity (mM). 3) Commercial HCl is 36%. Please convert it into molarity. 4) Find out the Volume (dm3) of product at STP when 0.28 M, 200 mL NaOH (aq.) reacts with 250 mL 85% H3PO4 (Sp. G.-1.067). 5) The above reaction has the product Ammonia, which when dissolved in 300 mL water makes an alkaline solution. Find its molarity (M). 6) Please re-calculate the child dose as per the BW of the baby. (Adult dose-750 mg and the BW of the baby is 50 lb (British pound) (1 lb=0.453 Kg).
- Mass solute needed to prepare 0.500L of 0.2 M NaCO3 from a solid NaCO3 with a purity of 92.0wt%1. Find the number of millimoles of solute in a. 226 ?? of 0.320 ? HClO4. b. 25.0 ? of 8.05 ? 10−3 ? K2CrO4. c. 6.00 ? of an aqueous solution that contains 6.75 ??? of AgNO3. d. 537 ?? of 0.0200 ? KOH. 2. Describe the preparation of a. 5.00 ? of 0.0500 ? KMnO4 from the solid reagent. b. 4.00 ? of 0.250 ? HClO4, starting with an 8.00 ? solution of the reagent. c. 400 ?? of a solution that is 0.0250 ? in I2, starting with MgI2. d. 200 ?? of 1.00% (?/?) aqueous CuSO4 from a 0.365 ? CuSO4 solution. e. 1.50 ? of 0.215 ? NaOH from the concentrated commercial reagent [50% NaOH (w/w), ?? ?? = 1.525]. f. 1.50 ? of a solution that is 12.0 ??? in K+, starting with solid K4Fe(CN)6. 3. Exactly 75.00 ?? of a 0.3132 ? solution of Na2SO3 were treated with 150.0 ?? of 0.4025 ? HClO4 and boiled to remove the SO2 formed. a. What was the mass in grams of SO2 that was evolved? b. What was the concentration of the unreacted reagent (Na2SO3 or HClO4) after thereaction was complete?The mass of lead chromate that is dissolved in 250 mL of a saturated solution is _____ grams. (Ksp = 1.8 x 10-14) The attached photo is an example of how to solve the problem
- 5) 15.00 mL of 0.620 M potassium chloride solution is combined with 25.00 ML of 0.870 M potassium phosphate solution. What is the concentration of potassium ions in the final solution (in M)? b) I would ask you to perform a Material Balance on the mixing - find all concentrations ( of all ions ) and the mass of the PPT.What is the solubility of AuBr (Ksp= 5.0 × 10-17 at 25 °C) in g/100mL? A. 7.07×10-9 g/100 mL B. 1.96×10-7 g/100 mL C. 1.96×10-6 g/100 mL D. 2.55×10-11 g/100 mLdescribe how you can prepare 500mL of 0.10 M Fe2O3 (159.69g/mol) from the primary-standard-grade solid.

