b. Account for this fact: Mixing 50 mL of methanol with 50 mL of water produces a solution with a volume slightly less than 100 mL. OMethanol evaporates during the mixing. ODuring hydration, methanol molecules decrease in volume. Owater evaporates during the mixing. Owater molecules interact extensively with methanol molecules. Submit Answer Try Another Version 2 item attempts remaining
b. Account for this fact: Mixing 50 mL of methanol with 50 mL of water produces a solution with a volume slightly less than 100 mL. OMethanol evaporates during the mixing. ODuring hydration, methanol molecules decrease in volume. Owater evaporates during the mixing. Owater molecules interact extensively with methanol molecules. Submit Answer Try Another Version 2 item attempts remaining
Chapter7: Solutions And Colloids
Section: Chapter Questions
Problem 7.86E
Related questions
Question
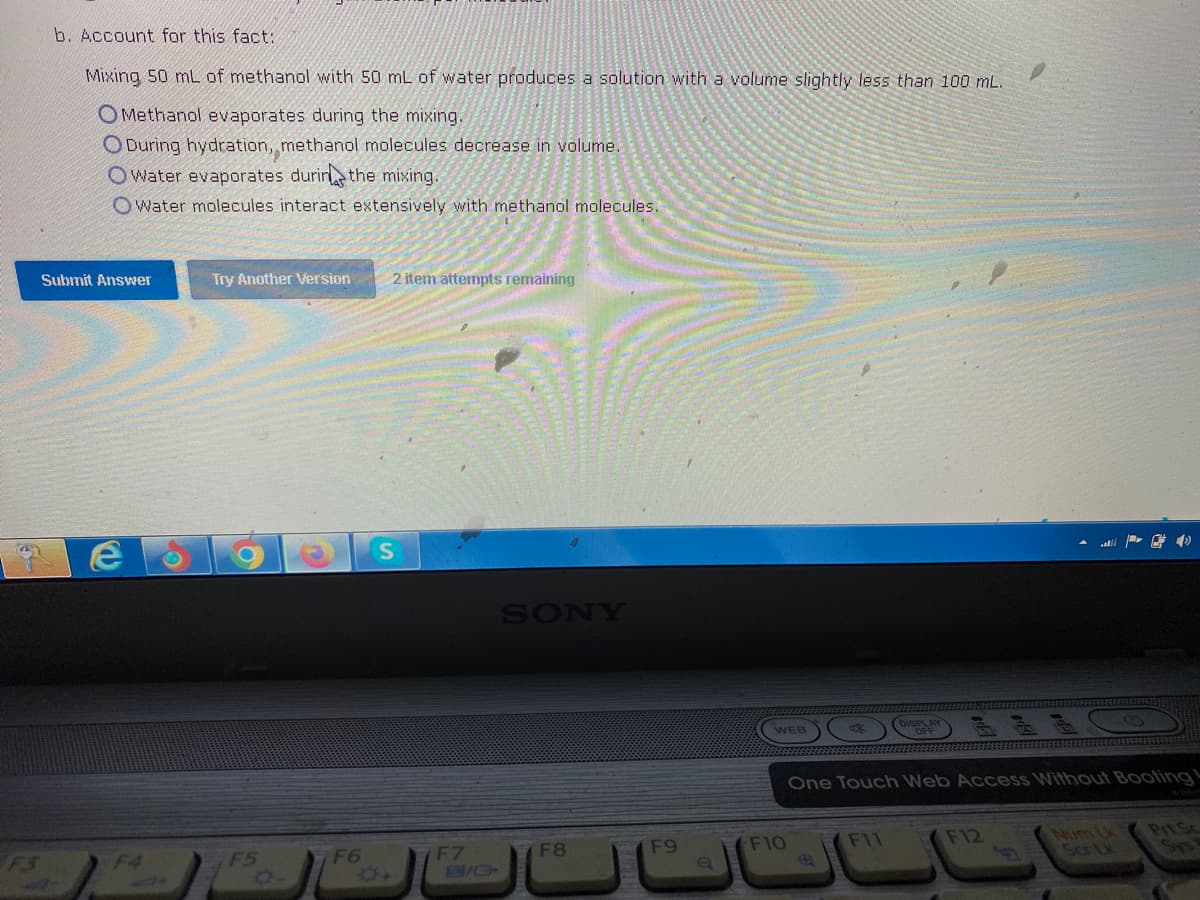
Transcribed Image Text:b. Account for this fact:
Mixing 50 mL of methanol with 50 mL of water produces a solution with a volume slightly less than 100 mL.
OMethanol evaporates during the mixing.
ODuring hydration, methanol molecules decrease in volume.
Owater evaporates during the mixing.
Owater molecules interact extensively with methanol molecules.
Submit Answer
e
Try Another Version
F5
n
F6
2 item attempts remaining
S
F7
C/C+
SONY
F8
F9
e
WEB
F10
OFF
One Touch Web Access Without Booting
F11
F12
Num Lk
PrtSc
Sys
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT
