b. CH CH2 | H;Č CH3 - Polypeptide backbone H3C CH3 ! CH C=OH -CH2+S-S CH2– а. CH2 -CH2-CH2-CH2–CH2-NH;* 0-C-CH2- d.
Q: A protein is allosterically regulated by a molecule. This molecule enhances the binding affinity, an...
A: Regulation in science refers to the adapted form or change in structure or function of any molecule ...
Q: 9. Compare the energy cost, in ATP equivalents, of synthesizing stearate from mitochondrial acety...
A: Stearate is a 18 carbon saturated fatty acid. Lets first look at the amount of ATP equivalents requi...
Q: 1.Would you expect a solution of high salt to be able to denature ribonuclease? Why or why not?
A: As there are multiple question you have asked that are not much interrelated to each other so accord...
Q: 1. Differentiate starch from cellulose and carb 2. In which solvents or solutions will a lipid be a....
A: Since, you have posted a question with multiple sub-parts, we will solve first three sub-parts for y...
Q: Please help me answer this in 10 sentences only. How can biotechnology help preserve the endangered...
A: The importance of wildlife to human welfare cannot be overstated. Wild animals are sources of money,...
Q: Classify characteristics of skeletal muscle energy metabolism by dragging each statement to its corr...
A: Oxidative phosphorylation This pathway requires oxygen Produces a significant fraction of ATP durin...
Q: cell wants to synthesize the a(1→4) dimer from two glucose molecules. Show the mechanism.
A:
Q: Identify the amino acid shown below. (Note: single letter code is provided as answer) H,N-C-COOH CH2...
A: Amino acids are the building blocks of proteins. The building blocks of living organisms are amino a...
Q: Two proteins bind the same ligand, and the data is shown below. Select the best answer based on this...
A: Lowest the KD value, greater will be the affinity of ligand to the protein. Highest the KD value, we...
Q: As homocysteine levels increases A. cardiac risk decreases B. cardiac risk increases C. cardia...
A: Heart along with the blood vessels (arteries, veins and capillaries) and blood comprises the cardiov...
Q: Explain why, glucose-6-phosphate will prefer to go via the pentose phosphate route. What additional ...
A: Metabolism includes biosynthesis/ reduction (an anabolic process) and oxidation (catabolic p...
Q: Define
A: Enzyme kinetic is a branch of Biochemistry. There are several factors that enzyme catalysis are depe...
Q: Briefly discuss the functions of the four types of biological molecules.
A: Four types of biological molecules include carbohydrates, proteins, fats/lipids, and nucl...
Q: 4. Which of the following substances is associated with Na+/K+ balance in the human body? a. Cortiso...
A: Thanks a lot for submitting the questions. As you have asked to answer only 4, 5, and 6. Please find...
Q: 7. Which set of the following amino acids are all polar? a. W K Q b. H M N c. T N Y d. M T C e. P C...
A: The side chains of amino acids are used to categorize them. Glycine (Gly), alanine (Ala), valine (Va...
Q: Kindly refer to the photo attached. thank you
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any ...
Q: 6. Which of the following statements concerning bile acids is incorrect? They are... a. Insoluble in...
A: Bile acids are steroid acids present in bile of mammals and vertebrates. These bile acids are conju...
Q: polecules used during process, purpose Fermentation & or Occurs when? cules produced during
A: The question is all about the fermentation cycle and glycolysis cycle. Both cycle contain some gases...
Q: Give one physiological c
A: pH is the measure of the strength of H+ ion or Hydronium ions in solution. pOH is the ...
Q: QUESTION 2 In a lab, you take 5 mL of of Stock solution and mix it with 5 mL of water to make soluti...
A: 5 ml stock solution present in solution A(10 ml) Solution B volume = 10 ml solution A+ 990ml water =...
Q: make the test principle, materials required for the analysis, sample preparation, test procedure, in...
A: A variety of organic compounds exist in nature. Among them, the nitrogen-containing organic compound...
Q: Why is there a need to filter the alkaline solution after adding the Barium chloride solution?
A: In an aqueous solution, barium chloride acts as a simple salt. However when dissolved in water, it ...
Q: The compound tetraethylammonium (TEA) blocks the voltage-gated changes in potassium permeability tha...
A: Action potentials are known to be the source of neuronal transmission, in the form of electrical sig...
Q: please provide a mechanism for how Pro-Thr-Pro-Ser amide could rearrange/molecule into a different m...
A: Amino acids are the building unit of proteins/polypeptide chain, consisting of amine group, carboxyl...
Q: The only sugar structure that does NOT contain chiral carbon atom a. Erythrose b. Erythroluse c. Gly...
A: Multiple questions asked. I can answer the first question, following the guidelines. Kindly repost o...
Q: SEE EXAMPLE IN THE IMAGE A mixture of 0.20M acetic acid and 0.30M sodium acetate is given. Calculate...
A: The Henderson Hasselbalch equation for calculation of the pH of a solution of weak acid and its conj...
Q: B. Calculate the concentration of the reducing sugar in the 50.0 mL sample in mg/mL. Apply Beer-Lamb...
A: A reducing sugar is any sugar that is capable of acting as a reducing agent.
Q: Sickle cell anemia results from a substitution of a valine for a glutamic acid. What do you expect t...
A: Sickle cell anemia: Normal red cells are round like doughnuts shape, but in sickle cell anemia, it ...
Q: Which of the following types of bonds are present in the primary structures of proteins? O Hydrogen ...
A: Proteins are composed of twenty standard amino acids attached together through amide bonds. These tw...
Q: ndicate what you would do and the observations you would expect for each case. Be explicit in your e...
A: Phenols are family of organic compounds characterized by a hydroxyl group attached to a carbon atom ...
Q: 3% red cell suspension using 0.5 mL of pRBC
A: The question is all about the preparation of 3% red cell suspension from 0.5 ml pRBC from the patien...
Q: Make a conclusion about the qualitative test on lipids You can use this as your reference: https:/...
A: Qualitative tests help to detect the presence of an unknown substance in a given sample. Presence of...
Q: If the sequence of mRNA is 5'-AAUCGUACGGAUGCCGAAAUACCCAUUAGGGAUUGCAUAGCGAGCAACGGAC-3' , what is the ...
A: The genetic code is the set of rules used for translation of the information encoded within genetic ...
Q: This is a reducing tetrasaccharide and is made up of 3 different monosaccharides True False
A: Given tetrasaccharide - Alpha(1,6) glycosidic linkage between 1st and 2nd monomer;2nd and 3rd monome...
Q: Draw the C-2 epimer of Ribose
A: Epimers are a pair of diastereomers, at one stereogenic carbon center of two epimers have at least...
Q: Explain how blood glucose can be maintained through the different pathways of carbohydrate metabolis...
A: Carbohydrates are the main source of energy for plants and animals. In plants carbohydrates are form...
Q: A 50.0 ml juice extract is colorimetrically assayed using Nelson's test. One milliliter (1.00 mL) of...
A: Answer: Principle: Nelson Somogyi method is a version of Somogyi'stitrometric method for use with ...
Q: Short-chain fatty acids such as butyrate are absorbed by the mammalian intestine and used as metabo...
A: Short chain fatty acids are the molecules having fatty acids with fewer than six carbon atoms.
Q: Why are some metabolic reactions coupled to the hydrolysis of ATP? To drive the nonspontaneous...
A: Metabolic reactions are involved during the metabolism of biomolecules like carbohydrates, proteins,...
Q: Why methylated spirit is used? Why the beaker with its content was kept in the dark raped Why methyl...
A: Methylated spirit is made up of ethyl alcohol and methyl alcohol. The composition of methylated spir...
Q: what is the purpose and objectives on doing nitrious acid test?
A: Amines are the compounds and functional groups having a basic nitrogen atom with a lone pair of elec...
Q: TRUE OR FALSE?
A: Metabolic pathways in living organism include series of enzyme-mediated chemical reaction that inclu...
Q: When combining 0.5 mL of Lysozyme solution (with a concentration of 10 mg/ml) and 0.5 mL of water, w...
A: Lysozyme is an enzyme which catalyzes the hydrolysis of peptidoglycans present in cell walls of bact...
Q: Why do steroid hormones not require signal transduction and second messengers to exert their actions...
A: Steroid hormones:- It is a group of hormones , belong to the class of chemical compounds known as st...
Q: From the complete oxidation of glucose (glucose → 6CO2), how many total nucleotide triphosphates are...
A: Glucose is metabolized through the glycolytic pathway to yield energy in the form of ATP and NADH. T...
Q: Ninhydrin Test Samples used: Egg Albumin Gelatin Dispersion Added reagent: Ninhydrin Solution ...
A: Ninhydrin is a general test for proteins and amino acids. It degrades amino acids into aldehydes, am...
Q: 1. A mother went to a drugstore to purchase a vitamin supplement for her baby. She found out from t...
A: Amino acids are the primary molecules that combine together to form the biomolecule proteins. These ...
Q: Which statement about N-linked glycosylation is correct? N-linked oligosaccharides are attach...
A: There are two types of glycosylation of protein – N Linked and O Linked In N-Linked glycosylation, o...
Q: Legend: Blue – wild-type β-galactosidase Red – mutant β-galactosidase _________ a. What is the...
A: The amino acid sequence of a protein determines the structure and hence the function of a protein. T...
Q: Phospholipase A2 causes the release of what fatty acid from membrane phospholipids? What chemical cl...
A: Phospholipase is an enzyme that breaks down phospholipids into fatty acids and other lipophilic comp...
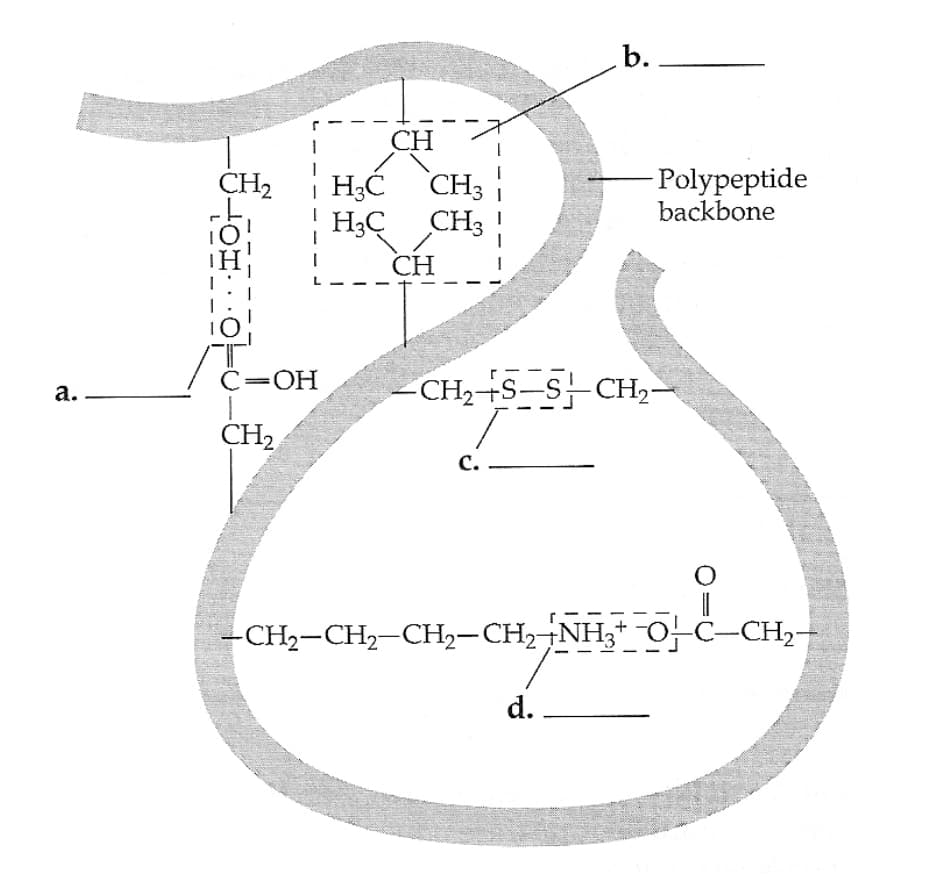
In the following diagram of a portion of a protein, label the types of interactions that are shown.
- What level of protein structure are these interactions producing?
Step by step
Solved in 3 steps with 1 images

- why is CH3-CH-CH2-CH2-CH3 non imposable ? I OHShow where trypsin and chymotrypsin would cleave the following peptide Tyr-Ile-Gin-Arg-Leu-Gly-Phe-Lys-Asn-Trp-Phe-Gly-Ala-Lys-Gly-Gin GinWhich peptide would absorb the most UV light at 280nm? (Can you show work because I mainly want to know how to solve this type of problem. Thank you!)Leu-Trp-Tyr-Ala Tyr-Lys-Tyr-Cys Glu-Tyr-Ile-Arg Ala-Trp-Trp-Ala Thr-Ala-Ile-Thr
- The primary structure of b-endorphin, a peptide containing 31 amino acids synthesized by the body to control pain, is shown here: Tyr-Gly-Gly-Phe-Met-Thr-Ser-Glu-Lys-Ser-Gln-Thr-Pro-Leu-Val-Thr- Leu-Phe-Lys-Asn-Ala-Ile-Ile-Lys-Asn-Ala-Tyr-Lys-Lys-Gly-Glu What fragments are obtained as a result of treatment withcyanogen bromide?The primary structure of b-endorphin, a peptide containing 31 amino acids synthesized by the body to control pain, is shown here: Tyr-Gly-Gly-Phe-Met-Thr-Ser-Glu-Lys-Ser-Gln-Thr-Pro-Leu-Val-Thr- Leu-Phe-Lys-Asn-Ala-Ile-Ile-Lys-Asn-Ala-Tyr-Lys-Lys-Gly-Glu What fragments are obtained as a result of treatment with trypsin?Hello! Can you please explain why C is correct?
- Draw the peptide at a pH @1 of Cys-His-Glu-Met-Ile-Ser-Thr-Arg-Tyr - do it on this formatCan you please help me how to draw tripeptide ile-leu-arg and indicate the torsion angles phi and psi for leucine?HindII --- 5' GTC - GAC 3', HaeIII --- 5' CC - GG 3', EcoRI --- 5' G - AATTC 3' and BamI --- 5' CCTAG - G 3' 5' AGAATTCTTACGCCGGACGTACCTAGGTTTAGTCGACTC CGCCGCCCCTAGGGTCATCA 3' 3' TCTTAAGAATGCGGCCTGCATGGATCCAAATCAGCTGAGGCGGCGGGGATCCCAGTAGT 5' Number of pieces of DNA , and blunt end fragment (s), and sticky end fragment(s)





