Q: Draw the structure of the compound used to form the enolate for the reaction.
A: Please see explanation part.Explanation:Step 1: Step 2: Hope you get the mechanism of making…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. CH3CH2CH2C(O)CH2CH₂CH pH 4-5…
A:
Q: 2. Predict the products for the following transformations. CH3 HNO3 OH A. H2SO4 CH3 Cl₂ B. AICI 3 Br…
A: Benzene rings are identified to be electron-rich in nature due to the presence of 3 pi bonds in…
Q: A.) Which C–O bond has the smallest bond energy? CO32– CO2 CO B.) Which reaction is…
A: C---O bond energy can be analysed by bond order calculation. More bond order more strength, more…
Q: 6. Predict the product for the subsequent reaction: 요 hv NC
A: The objective of the question is to find the product of the reaction in which given carbonyl…
Q: The diene shown below will NOT react in a Diels-Alder reaction. Why not? Select one: O A. The…
A: The objective of the question is to choose the correct option for the molecule given ragarding…
Q: The compound shown below is the product of a Claisen condensation. Draw structural formulas for the…
A:
Q: 3. Give the reagents needed to form the indicated products: CN CN
A: In the given example the first step is elimination reaction to form a double bond. In the second…
Q: In which process is w > 0 at constant pressure? 3 H2(g) + N2(g) à 2 NH3(g)…
A: Work done (w) is considered positive when the system expands against the surrounding pressure. In…
Q: Lithium reacts with nitrogen to form lithium nitride. 6 Li(s) + N2(g) → 2 Li3N(s) In one experiment…
A: The % yield of the reaction is 25%.Explanation:
Q: What is the major product of the following reaction? EIO OEI 1. NaOEt 2. H₂O+ Give detailed…
A: The Dieckmann condensation is a reaction that occurs when diesters react with a base to produce…
Q: EtO OEt NH2
A:
Q: only typed solution
A: The objective of the question is to determine which among Ethene, Propane, Propene, and Ethane has…
Q: The gas-phase reaction 2N2O5 = 4NO2 +O2 has an activation energy of 103 kJ/mol, and the rate…
A: The objective of this question is to find the rate constant of a gas-phase reaction at a different…
Q: CI VaNH2 heat
A: reaction is known as a bimolecular elimination reaction.It is a concerted process or a single step…
Q: A 0.409 M solution of the salt NaA has a pH of 8.83. Calculate the Ka value for the acid HA. Record…
A: pH = 8.83Molarity of NaA = 0.409 MKw = 1.010-14
Q: (please correct answer and don't use hend raiting) What is the point group of AlCl4-?
A: The ion given is tetrachloroaluminate ion (AlCl4-).It is required to answer the point group for this…
Q: In a reaction, 1.00 mole of H2 is produced at a constant pressure of 1.00 atm and at 298 K (1 L atm…
A: +481 kJ closest is +483.6 kJ Explanation:Recall that at constant pressure:dU=q+w and q=dHand we can…
Q: Classify the following configurations as A, E, or T in complexes having Oh symmetry. Some of these…
A: t2g4eg2 as 'T' - The pair in t2g orbitals can be in any of the three.t2g6 as 'A' - There is…
Q: A chemistry graduate student is given 450. mL of a 0.40M acetic acid (HCH3CO2) solution. Acetic acid…
A:
Q: What should be the volume of concentrated ammonium hydroxide that should be measured to prepare 100…
A: Volume of the concentrated ammonium hydroxide to be used is 2.75 mL or approximately 3 mL.This…
Q: Draw a major resonance contributor of this enolate anion. Include all lone pairs in your structure.…
A: The objective of the question is to find the major resonance contributor of the given enolate…
Q: What is the pH of 2.11E-4 M KOH(aq) at 25 °C? (Kw = 1.01E-14) -10.3 9.41 10.3 11.1 3.68
A: Concentration of KOH = 2.1110-4Kw = 1.0110-14
Q: a. Choose the complete Lewis structure for the following partially completed Lewis structure. F F…
A: In Lewis structure, all the bonds as well as lone pairs (non-bonding electrons) are represented in…
Q: Consider the molecular orbital model for these diatomic species: CN+ CN CN- Fill in the correct…
A: The electrons in each molecular orbital in case of CN+, CN and CN- are: CN+,…
Q: 2 significant digits, and Each row of the table below describes an aqueous solution at about 25 °C.…
A: Solution [H3O+]PHA3.2×10−98.49B9.12×10−54.04C5.2×10−87.28Explanation:
Q: Write the systematic (IUPAC) name for each of the following organic molecules: HO structure OH name…
A:
Q: CI + 2 N-H
A: Given that a reaction scheme.We have to drawthe structure of tetrahedral intermediate.The structure…
Q: Select the reagent for the reaction. Леон OH 00 Od + NH N=C=N بعد
A: The given reaction is an example of dehydration reaction in which a carboxylic acid is reacted with…
Q: Complete the following reaction:
A: Alkene gives hydration reactions in the presence of an acid catalyst. The reaction follows an…
Q: 8. Write stepwise mechanism for the following reaction. CH3 + HC1 CH3 C1
A:
Q: What would the complete mechnism be ? (I.e including intermediates, etc) OH HO H2SO4 C
A:
Q: name the molecular Shape around each Central atom that has an arrow pointing at it.
A: Given a molecule with pointing central atoms:
Q: Predict the product of the oxidation of the given aldehyde. Include all hydrogen atoms. Modify the…
A: The reactant has an aldehyde functional group and this functional group is oxidized into carboxylic…
Q: Consider the following reaction between calcium oxide and carbon dioxide: CaO (s) + CO2 (g)…
A: The objective of this question is to calculate the percent yield of the reaction between calcium…
Q: we now have: Step 1: Fast, reversible HA = H+ + A- Step 2: Fast, reversible X + H+ = HX+ Step 3:…
A: The objective of the question is to determine whether the given rate law contains an intermediate.…
Q: The compound shown below is the product of a Claisen condensation. Draw structural formulas for the…
A: The objective of this question is to draw the enolate and ester involved in the formation of a given…
Q: Calculate the pH of a buffer that is 0.280M in NaHCO3 and 0.330M in Na, CO. Express your answer to…
A: The objective of this question is to calculate the pH of a buffer solution and a mixed solution. The…
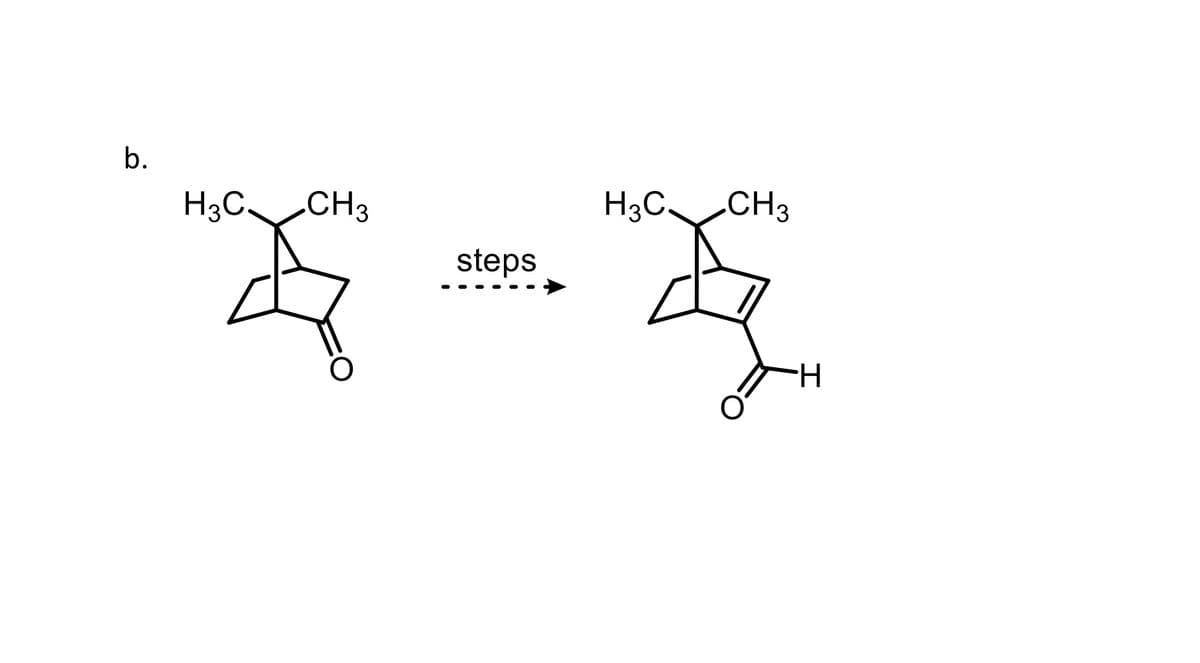
Q: Devise a multistep synthetic route for the following transformation. You may only use three-carbon…
A: The given synthesis scheme is shown below.We have to provide a synthesis pathway.
Q: Give the IUPAC name for the following compound:
A: The IUPAC rules for naming alkenes are given as follows:Find out the longest chain that contains…
Q: which one is kinetically favoured and which is thermodynamically flavoured? explain your answer
A: Alkene C is kinetically favored and Alkene B is thermodynamically favored.Explanation:Compound A has…
Q: following redox reactions as either galvanic or electroly a) Br2(1) + Sn(s) → 2Br (aq) + Sn²+(aq):…
A:
Q: Draw the major product (constitutional isomer) formed when the reactant shown is hydrogenated with…
A: The objective of the question is to find the major product formed from the reaction in which the…
Q: Gve the organic product(s) formed at the end of the following sequence or reactions: Zn(Hg) +…
A: Option CExplanation:Step 1: Step 2:hope you got your answer. Step 3: Step 4:
Q: According to the Experiment #8 PowerPoint provided in the Experiment #8 module on your lab section's…
A: The emission spectrum is weak once everything is setup.Need to chose the correct option.
Q: Draw the major product of this reaction. Include stereochemistry if applicable. Use a dash or wedge…
A: The reactions in which an atom or a group of atoms are replaced by another atom or group of atoms…
Q: Give a mechanism for the formation of the minor product methylenecyclohexane from dehydration of…
A: Alcohols are organic compounds that have OH functional groups. Alcohol behaves a nucleophile due to…
Q: A metal object has a mass of 19.67 g. When it was placed in a graduated cylinder containing 20.0 mL…
A: the given metal is Iron Iron 7.87 g/mL error is zero Thank you Let me know if you have any…
Q: -NH pyrrole Х benzaldehyde + NiCl2 DBU, Microwave irradiation 6 min
A: The objective of this question is to predict the reaction mechanism involved in the synthesis of…
Q: How can you tell if they are in equilibrium based on k?
A: 1st and 4th are equilibrium2nd and 3rd are not at equilibrium Explanation:Step 1To determine if a…
Step by step
Solved in 3 steps with 3 images


