Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.10QAP
Related questions
Question
Transcribed Image Text:A www-awu.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-liiHqRUDAYpH2DXijQEc443v2U4KEog-Tfi3mlfS3CecvGpd
G Google
O YouTube
Mail Araya, Denait...
M Inbox (1) - denaitar...
E Sign In - Oasis
Điscover Student L..
publi
O CHEMICAL REACTIONS
Balancing chemical equations with interfering coefficients
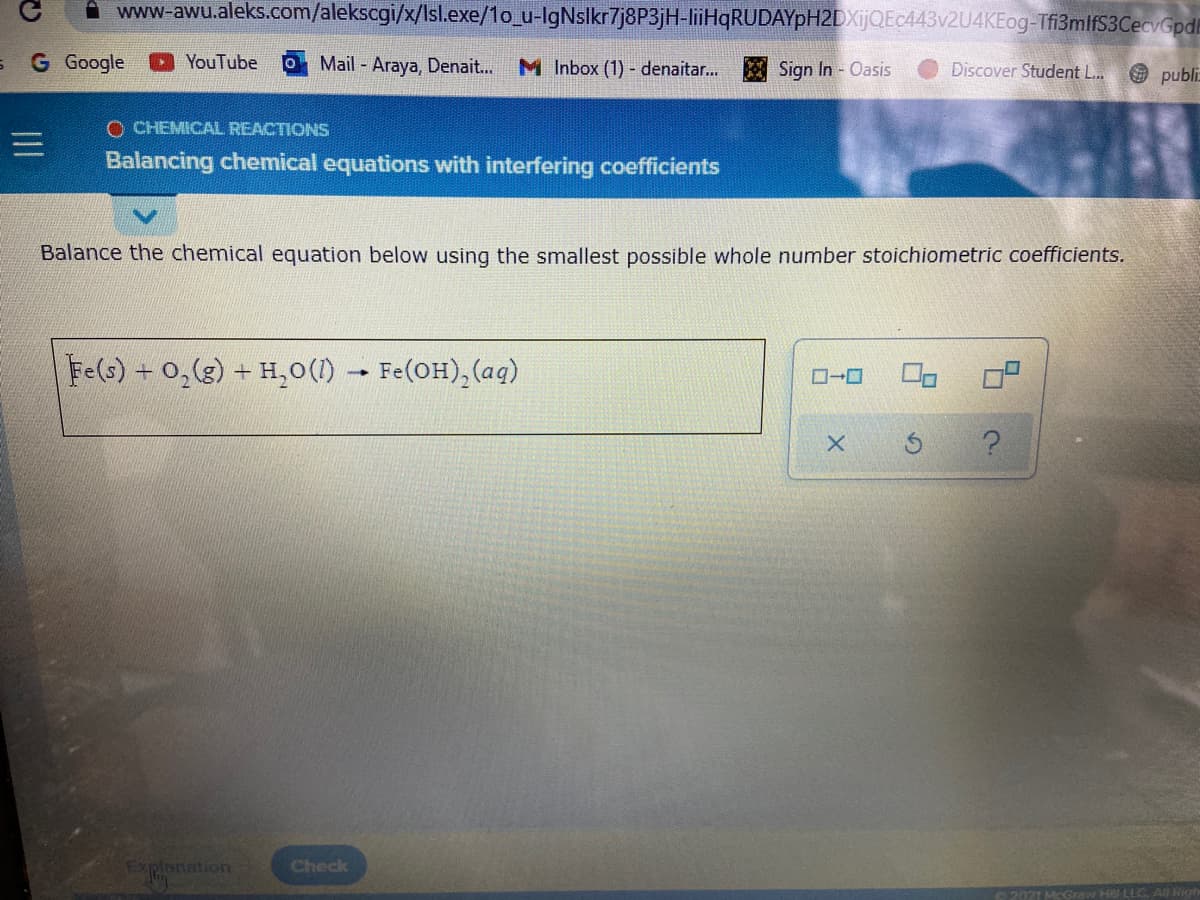
Balance the chemical equation below using the smallest possible whole number stoichiometric coefficients.
Fe(s) + 0,(g) + H,0(1) - Fe(OH),(aq)
Explanation
Check
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

