Q: 3. Two of the four structures below correspond to the 13C NMR spectra shown on this page. Indicate…
A: 13C NMR spectroscopy is useful to determine types of carbons, their splitting, signals etc hence we…
Q: Which is the correct order of basicity for the following compounds? 1 || NH O III > IV > II > I O II…
A: Basicity is defined as electron donation ability of a molecule. If it is able to donate electrons…
Q: T Ho NH₂ synthesis target Dar Starting Martrials
A: For the above given synthesis target and starting material. First we will break the synthesis target…
Q: ©Macmillan Learning Construct the molecular orbital diagram for H₂. 0.5 H Identify the bond order.…
A: Bond Order = 1/2 (number of electrons in the bonding molecular orbital - number of electrons in the…
Q: Draw the mechanism for the production of Nylon, identify the new linkage that has formed as a result…
A:
Q: Write the reaction for the sulfonation of benzene
A: Benzene react with oleum to form benzene sulphonic acid.
Q: The rate constant for a certain reaction triples when the temperature increases from 300 K to 319 K.…
A: The relationship between the rate constant (k) and temperature (T) is given by the Arrhenius…
Q: The anode reaction is: A voltaic cell is constructed from a standard Cd²+ |Cd half cell (E° red =…
A:
Q: A 1 liter solution contains 0.441 M hypochlorous acid and 0.331 M sodium hypochlorite. Addition of…
A: A buffer is a solution which resist any change in pH on adding a small amount of acid or base . it…
Q: Give the molecular geometry and number of electron groups for BrF5. Osquare planar, 6 electron…
A:
Q: The deep blue compound Cu(NH3)4SO4 is made by the reaction of copper(II) sulfate and ammonia. CuSO4…
A: The amount of product predicted by stoichiometry is called the theoretical yield, whereas the amount…
Q: Consider an electron moving along a-1-dimensional lattice with nearest-neighbor matrix elements V =…
A:
Q: Consider the dissolution of nitrous acid: HNO₂(aq) = H(aq) + NO₂ (aq). For this reaction under…
A: The given reaction is the dissolution of nitrous acid HNO2(aq) ⇌H+(aq) + NO2-(aq) The given data The…
Q: What is the predicted product of the reaction shown? NH₂ $ 8. 5 I HN HO IV NH- || Но H₂SO4 N V…
A:
Q: How many milliliters of 3.9 M HCl must be added to 3.7 L of 0.11 M K2HPO4 to prepare a pH = 7.40…
A: Answer: Buffer solution is a type of solution that resists the change in its pH on adding small…
Q: How much 5.80 M NaOH must be added to 490.0 mL of a buffer that is 0.0180 M acetic acid and 0.0275 M…
A: When NaOH is added to the buffer solution of acetic acid and sodium acetate, acetic acid will react…
Q: Method 2: Calculating AG from AH and Soº data. ● If you have AHP and So data, you can use Hess' Law…
A:
Q: Sive the major elimination product of the following compound. Br (CH₂), COH heat ?
A:
Q: [Review Topics] [References] Splitting of a signal in a proton NMR spectrum tells us the number of…
A: 1H NMR spectroscopy is useful for determination of types of hydrogens, their splitting, signals…
Q: .CO 5 F5 PUI % 5 F6 What is Standard Temperature and Pressure (STP)? A A) 298.15 K and 1.00 atm B)…
A:
Q: What are the major products of the following reactions? Include stereochemistry. a. 1. Hg(OAc)₂,…
A:
Q: 5. Consider the following reaction at some temperature: Kp = 9.00 Initially, 3.00 atm of H₂ and 3.00…
A: Reaction : H2(g) +F2(g) → 2HF (g) Kp=9.00
Q: A solution contains 0.454 M (CH3)2NH2Br and 0.267 M dimethylamine, (CH3)2NH. The pH of this solution…
A:
Q: Agt Pb²+ Hg₂²+ 3) Consider that a lab team has been performing the same Group I analysis as is…
A: 6 M HCI was first added. 6 M NH3 was added to the solid dissolving it without residue. K2CrO4 was…
Q: Part A Given the two reactions 1. H₂S(aq) HS (aq) + H+ (aq), K₁=9.82x10-8, and 2. HS (aq) = S²(aq) +…
A:
Q: A solution is made from 1.53 g of benzoic acid (C6H5COOH; Ka = 6.5 × 10–5 ) in water to a final…
A: Mass of benzoic acid (C6H5COOH) = 1.53 g Volume of the solution = 100 mL = 0.1 L Ka of benzoic acid…
Q: Write the equation in exponential fo log_(2)256=8 The equation in exponential form.
A: We have the logarithm represention log2256 = 8 We are to write this equation in exponential form.
Q: pka of butanoic acid is 4.82. Round your answer to 2 decimal places. Note for advanced students: you…
A:
Q: The reaction Q(g) + R(g) → Z(I) is shown to be exothermic. Which of the following is true concerning…
A: Reaction is spontaneous if ∆G = -ve & Reaction is non spontaneous if ∆G = +ve
Q: The first step of the mechanism is given in the sketch box below. Modify the given drawing of the…
A: The question belongs to basics in organic reation mechanism.
Q: The half-life of a first-order process is 1.39×10-2 seconds. What is the corresponding rate…
A:
Q: Explain the difference between an indicator endpoint for a titration analysis and the true…
A: Titration is a laboratory technique used to determine the concentration of a substance (the analyte)…
Q: Part A What is the half-life for this reaction at an initial concentration of 0.100 M? Express your…
A:
Q: Which of these indicators could be used in the titration of aspirin (pKa = 3.5) and which could be…
A: We have to find out suitable indicator. Given aspirin pKa = 3.5 quinine pKa = 8.05
Q: Use the References to access important values if needed for this question. Does a reaction occur…
A: Steps to write a net ionic equation and complete ionic equation for a molecular reaction :…
Q: Types of Chemical Reactions Practice Problems Chapter 10 Combination A + B AB K…
A: Incomplete reaction- (i) K + Cl2 (ii) Ca + O2 (iii) BaO + H2O We need to complete the…
Q: Which element is oxidized in the following reaction? Fe²+ + H+ + Cr₂O72- →→Fe³++Cr³++ H₂O
A: Oxidation is a chemical process that involves the loss of electrons or an increase in the oxidation…
Q: Draw the structures, including stereochemistry, of the major products (A) from the re- action…
A: Here the point is to introduce the mercury in the organic compound. The first step is the…
Q: If 25.5 g of NaOH is added to 0.550 L of 1.00 M Ni(NO₃)₂, how many grams of Ni(OH)₂ will be formed…
A:
Q: You want to design a galvanic cell using an iron (Fe) electrode as the cathode. Which of the…
A:
Q: What is the molarity of a solution that contains 2.38g of H2C2O4 x 2H2O in 200ml of solution?
A:
Q: Which of the choices is a L-hexose? O III Н- НО H H H ОН H НО ОН Н -ОН CH₂OH 1 Н CH2OH -0 -н -ОН -ОН…
A: If OH of the bottom chiral center is points to the right, the sugar is D If OH of the bottom chiral…
Q: Provide the MECHANISM for the following reaction. Do not add any new reagents. H3C- CH ₂ NaOEt EtOH
A: Carbonyl compounds having alpha-hydrogen in the presence of a base form enolate ion which can then…
Q: What is the oxidation number of nitrogen in the HNO2 molecule? O(A) O (B)-1 (C) +4 (D)-2 (E) +3
A: We have to find oxidation number of nitrogen whereas oxidation state of hydrogen and oxygen are…
Q: To measure the amount of calcium carbonate (CaCO3) in a seashell, an analytical chemist crushes a…
A: Given , Reaction :2HCl(aq) + CO32-(aq) → H2CO3(aq) + 2Cl-(aq)
Q: Calculate the standard cell potential for the galvanic cell, which has the overall balanced equation…
A:
Q: Draw the sugar below in its open-chain form: OH НО- CH₂OH он OH
A:
Q: What is the major product to the following reaction? H₂O H₂O*
A: Ketone reacts with water in presence of an acid catalyst to form a hydrate of ketone (geminal diol).…
Q: With the starting material provided, propose a synthesis pathway to prepare the diketone shown…
A: We have find out the answer. The given in reaction can be done by following four steps. 1) radical…
Q: Calculate the volume in milliliters of a 1.9M silver perchlorate solution that contains 50.0 g of…
A: First find out the number of moles for the given compound. Using the calculated moles and molarity,…
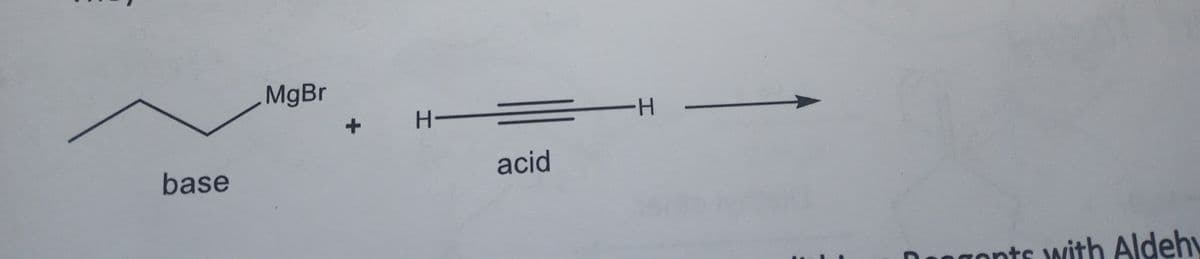
Predict the product(s) of the following organometallic reaction.
Step by step
Solved in 3 steps with 2 images

- Draw the product formed when phenylacetic acid (C6H5CH2COOH) istreated with each reagent. With some reagents, no reaction occurs. NaHCO3Thioglycolic acid, HSCH2CO2H, a substance used in depilatory agents (hair removers) has pKa = 3.42. What is the percent dissociation of thioglycolic acid in a buffer solution at pH = 3.0?Draw the product formed when phenylacetic acid (C6H5CH2COOH) istreated with following reagent. With some reagents, no reaction occurs. NH3 , Δ
- Which of the following is not a monodentate ligand ? Select one: a. OH- b. H2NCH2CH2NH2 c. H2O d. CN-Rank the following substituted anilines from most basic to least basic:Draw the products formed when p-methylaniline (p-CH3C6H4NH2) istreated with following reagent. Part (b), then CH3COCl, AlCl3
- Draw the structure corresponding to each name.a. 5-methylheptanoyl chlorideb. isopropyl propanoatec. acetic formic anhydrided. N-isobutyl-N-methylbutanamidee. 3-methylpentanenitrilef. o-cyanobenzoic acidg. sec-butyl 2-methylhexanoateh. N-ethylhexanamideDraw the products formed when p-methylaniline (p-CH3C6H4NH2) istreated with following reagent. excess CH3IThe cyano (-CN) group is considered a .... Ortho/Para Directing Activator Ortho/Para Directing Deactivator Meta Directing Deactivator

