Q: Arrange the following sets of organic compounds according to increasing solubility in water I.…
A: If the aliphatic carbon chain length of alcohol is longer then the solubility in water like polar…
Q: Determine whether each of the following compounds is soluble or insoluble: Drag the appropriate…
A: "Like dissolves like" is the thumb rule for dissolution of solute in solvent. Covalent compounds are…
Q: For each of the two horizontal series below, rank each molecule from highest (1) to lowest…
A: All known compounds can be dissolve in certain solvents whereas they remain insoluble in other…
Q: Rank the attached compounds in order of increasing water solubility ?
A: Solubility of any molecule increases with increase in the extent of hydration. In the given problem,…
Q: _1. Which of the following compounds would you expect to have the highest percent hydration at…
A: Given,
Q: Which of the following molecules is the MOST water soluble? H2N. HO, H. Но C
A: Concept : compounds having more number of highly electronegative atom like Nitrogen and oxygen with…
Q: Compounds solubility, in waterBenedict's Test lodoform Test Tollens' test CH3-CH2-CHO ç =0 CH2
A: Since you have asked a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Rank the following compounds in order of increasing water solubility. The first compound in your…
A: The solubility of a compound depends on the nature of the solute and the solvent. If the nature of…
Q: How does the boxed functional group affect this drug's solubility? .CI it increases water solubility…
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question…
Q: Rank the following compounds in each set from most soluble to least soluble in water:
A: Solubility: The general rule that in solubility is like dissolve like, in other words, polar…
Q: NH2 NH2 NH2 a.
A:
Q: In which of the following solvent the substance CH3NH2 will be most soluble? CH3COCH3 A. CH3CH2OH В.…
A: Solubility of methyl amine.
Q: From each pair of compounds, select the one that is more soluble in water.
A: From the given pair of compounds, the one that is more soluble in water is to be selected- CH2Cl2 or…
Q: Sort these carboxylic acids based on their solubility in water. Drag each item to the appropriate…
A: Carboxylic acids are generally soluble in water upto 5-carbons because of formation of hydrogen…
Q: Which of the following compounds is completely SOLUBLE in water? O Cyclohexane O pentadecane O 2,…
A: There are different type of compounds which have different solubilities in different solvents…
Q: Lauric acid will more likely dissolve in: a. H2O b. CCl4 c. CH3OH d. octanol
A: Lauric acid is a 12-carbon saturated fatty acid. The salts and esters of lauric acid are know as…
Q: 2-butanone has low solubility from butanal. * O True O False
A: Aldehyde and ketones are moderately soluble provided it contains less number of carbon. With…
Q: Which of the following would be MOST soluble in water? All of these would have about the same…
A: Solubility is defined as the ability of a solute to dissolve in a particular solvent. Here, the…
Q: What will be the solubility of the sugar if dissolved in 60 degrees celsius water?
A: If a solute is a solid or liquid, increasing the temperature increases its solubility. For example,…
Q: Arrange the given structures in INCREASING WATER SOLUBILITY. 1 - Least Water Soluble and 5 - most…
A:
Q: Based on molecular polarity, which of the following would be soluble in methanol CH3OH pentane…
A: As saying goes like dissolved like , as methanol is polar in nature as it contains -OH group .
Q: :Hydrates are defined as compounds that have had their water molecules removed. compounds with water…
A: Hydrate term is related to water.
Q: Benedict's Test Compound Solubility Iodoform Tollens" in water Test test CH3-CH2-CHO C = 0 + CH +…
A: All Hydrogen bond containing compounds are soluble in water , because water is a polar Hydrogen bond…
Q: In the previous problem, you compared the water solubility of the following molecules. H2N. HO, H.…
A: Intermolecular forces refer to the type of interactions which are present in between atoms or…
Q: Arrange the organic compounds from most soluble in water to least soluble in water. Most soluble in…
A: The solubility of a compound depends on the nature of the solute and the solvent. If the nature of…
Q: compound given are a common organic solvent. From the pair of compounds, select the solvent with…
A: Interpreting given different compounds:
Q: Which of the molecules shown below would be expected to have the highest water solubility A CH3CH₂OH…
A: The solubility of a compound increases as its molecular mass decreases. Solubility also depends upon…
Q: From each pair of compounds, select the one that is more soluble in water.
A: Water is the polar molecule due to the presence of electronegative element oxygen which attract…
Q: Is cyclohexane soluble in water? HCl? NaOH? NaHCO3?
A: formula of cyclohexane is C6H12
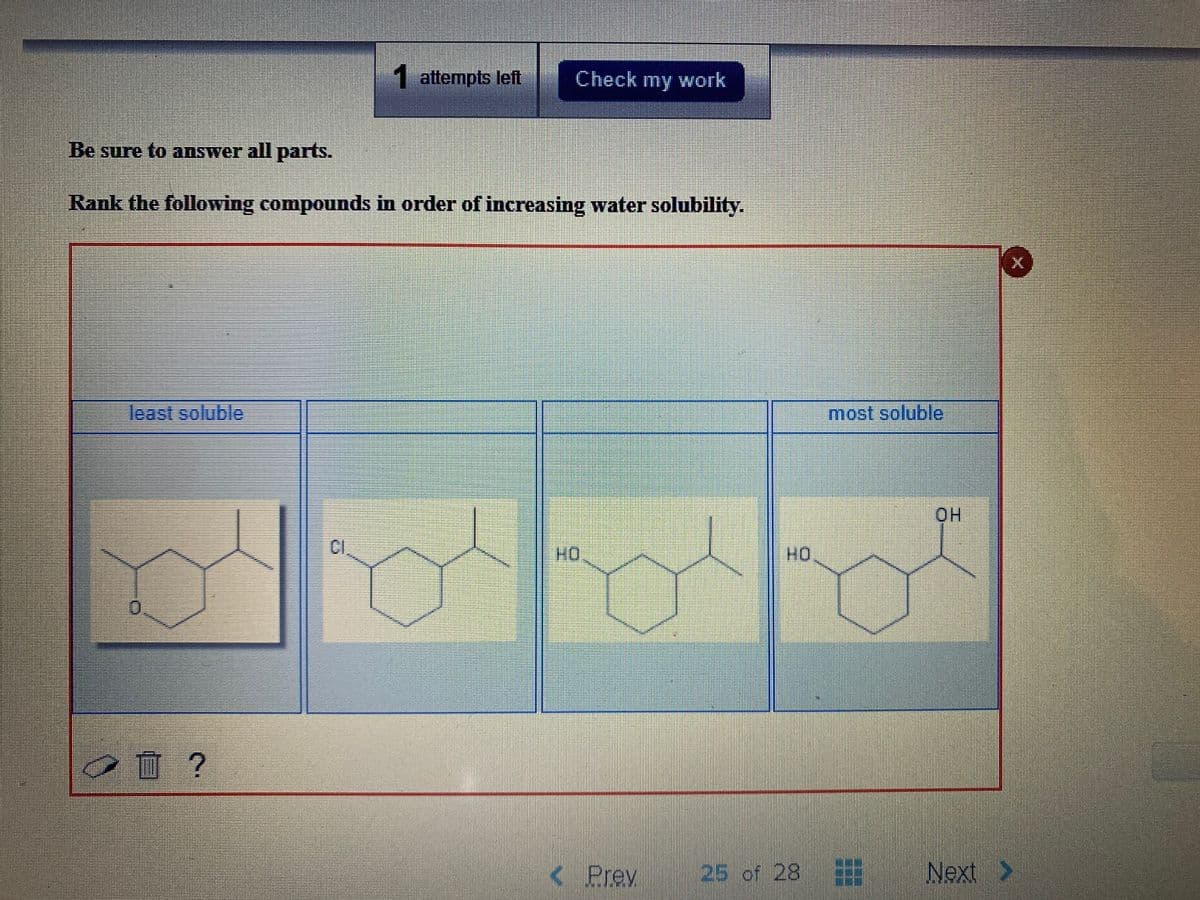
Q: Rank the following compounds in order of increasing water solubility.
A: Since in case of molecule 1 i.e alcohol we will have very high number of H bonds between it and…
Q: Select one: HO, HO OH
A: Applying concept of hydrolysis of alcohol with the help of hydrogen bonding and chain length.
Q: Thymol is very slightly soluble in water and very soluble in 1.0 M NaOH. Explain. CH3 ОН CH- CH3…
A:
Q: Which one of the following is least soluble in water? О снзон O CH3CH2CH2OH O CH3CH2OH O…
A: Solubility in the water is depends upon the interaction with the water molecule. Water is the polar…
Q: Rank the following compounds according to increasing solubility in water. 1 CH-CH-CH-CH; II…
A: Organic compounds are mostly made up of carbon, hydrogen, oxygen, nitrogen atoms. Organic compounds…
Q: Rank the following compounds in order of increasing solubility in MeOH (polar):
A:
Q: Rank the following compounds in decreasing order of water solubility (highest to lowest). see the…
A: Solubility of solute in a solvent follows the rule of like dissolves like. This means that polar…
Q: Thymol is very slightly soluble in water and very soluble in 1.0 M NaOH. Explain.
A:
Q: Which of the following is relatively soluble in a non polar solution but is soluble in water HCOOH…
A: The non-polar solution is that which has no separation of charge so no ions will be formed or the…
Q: compound given in this problem is a common organic solvent. From pair of compound , select the…
A: Water is a polar molecule. The hydrogen and oxygen atoms are present in the same molecule. So,…
Q: CH, CH3 H;C HO H. A B NH, H;C *CH3 H;C CH3 H;C `CH3 D E F
A: Naphthalene is a white crystalline solid which has the property to sublime. The chemical formula is…
Q: Arrange the following compounds in the order of increasing solubility in water (least first).…
A:
Q: From each pair of compounds, select the one that is more soluble in water.
A:
Q: Based on your answers to Questions 7, 8, and 9, which ranks these three compounds in order of…
A: In this question, we will arranged the all three compound in increasing order of their solubility in…
Q: compound given in this problem is a common organic solvent. From pair of compound , select the…
A: The solubility of a compound in the solvent depends on the like dissolve-like principle. This means…
Q: Predict which member of each pair will be more soluble in water. Explain the reasonsfor your…
A: The solubility depends on the extent of H bonding, higher, the hydrogen bonding, higher will be the…
Q: Rank the following compounds according to water solubility. Put the most soluble compound at the top…
A: More polar and less hindered groups are considered to have more solubility
Q: Which
A: In the given question we have to choose the correct option among following compound which is most…
Q: Thymol is very slightly soluble in water and very soluble in 1.0 M NaOH. Explain.
A:
Q: Predict which member of each pair will be more soluble in water. Explain the reasonsfor your…
A: To predict which of below is more soluble in water. heptan-1-ol or 4-methylphenol
Q: Which of the following is least soluble in water? A. CH3OH B. CH3CH2CH2OH C. CH3CH2OH D.…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 4 images

- Objective: To determine the percent NaCl (w/v) of a water sample 5.00 mL sample diluted to 50 mL solution 0.1000 M AgNO3 Volume of AgNO3 trial 1=15.30 mL trial 2=14.90 mL trial 3=15.10 ml1-A bottle of Na2EDTA.2H2O is labeled with an assay of 100.5%, what is this implied?What is the result of heating to 100 ºC?Show that mmol of aspirin recovered divided by mmol of limiting reagent x 100% gives the same % yield as calculated above
- What are the volume (uL), moles, and/or weight (mg) of each reagent and solvent in:50 mg of the “Bromo” starting material (1 equivalent), acid catalyst (if any, 0.1 eq) and in 0.25 M of solvent?Melting point: 98-105 Percent yield: 60% did you successfully make pure product? Explain.how do you find mmole of tetraborate and [B4O5(OH)4^2-] for trial 1?
- In a few sentences summarize your results for odor, solubility, acidity and the FeCl3 test for the knowns. Ethanol 2-Propanol t-Butanol Cyclohexanol PhenolDescribe the preparation of 2L of a 0.4 M HCl starting with a concentrated HCl solution (28% w/w, specific gravity = 1.15) -36.5 gm/molHello, I do not understand this quiestion or how to do this problem. Can you help me so I can follow along. 7. 5.00 mL of stock solution is diluted to 25.00 mL, peoducing solution ALPHA. 10.00 mL of solution ALPHA is diluted to 25.00 mL resulting in solutin BETA. 10.00 mL of solutionBETA is then diluted 25.00 mL producing solution Gamma. a. Determine the dilution factor in produciing solution Alpha from the stock solution. b. Determine the dilution factor in producing solution Beta from the solution Alpha c. Determie the dilution factor in producing solution Gamma from the stock solution Beta. d. Determine the overall serial dilution factor in producing solution Gamma form the stock solution.
- A. Compute the amount of reagents which will be used for the preparation of the following reagents. Show your computation clearly and neatly. a. 2 L of 5% (NH4)2C2O4 e. 1.5 L of 0.04 M KMnO4b. 500 mL of 0.1% (NH4)2C2O4 f. 3 L of 1 M H2SO4c. 1 L of 6 M NH4OH g. 500 mL of 0.1 M HCld. 2 L of 0.1 M NaOH B. Describe the preparation and storage of each reagents.Give good solution of all 3 subparts plzEach of the student statements below is wrong. Explain why they are not correct. (a) All my compound dissolved right away in the small amount of solvent I added at room temperature, and I didn’t need to heat it at all. This means I’m going to get lots of pure compound out. (b) I did a recrystallization of naphthalene and my percent recovery was very high (99%), so I must have pure product. (c) When you’ve dissolved all your compound in hot solvent, and you’re in a hurry, it’s ok to just place it straight into the ice bath.

