Q: The reaction of hydrogen sulfide(g) with oxygen(g) to form water(l) and sulfur dioxide(g) proceeds…
A: The given reaction is, 2H2S (g)+ 3O2 (g) → 2H2O (l) + 2SO2 (g) Given that: mass of H2S = 7.60 g…
Q: 7. Consider the combustion of methanol: pg292 2CH3OH(I) + 302(g) 2CO2(g) + 4H2O(I) Calculate the AH…
A:
Q: Consider the problem below: (Equation 1) 5 CO(g) + 5 H2O(1) · C5H10(1) + 5 O2(g) AH = 890.6 kJ --->…
A:
Q: Based on the reactions: 2NO2 (g) + 2NO (g) + O2 (9) – – > 2N½O4 (g) ; AH, = -228.54 kJ %3D 4NO (g) +…
A:
Q: ΔH for the reaction IF5 (g) → IF3 (g) + F2 (g) is __________ kJ, given the data below. IF (g) + F2…
A: Given reactions, IF (g) + F2 (g) → IF3 (g) ; ΔH = -390 kJ ----1 IF (g) + 2F2 (g)→ IF5 (g)…
Q: 3Fe2O3(s) + H2(g)---->2Fe3O4(s) + H2O(g) When 55.9 g Fe2O3(s) reacts with sufficient H2(g), 0.700…
A: 55.9 gm of Fe2O3 evolve = 0.7 KJ energy For this reaction, 3 moles of Fe2O3 required. So, in 3 moles…
Q: The reaction of hydrogen(g) with oxygen(g) to form water(g) proceeds as follows: 2H2(g) +…
A:
Q: H3C. NBS CHS KMNOJOH- OH- (acid work up) (heat) (acid work up) AICI3 (heat) AICI,
A:
Q: Given the following data 2 CIF(g) + O2 (g) → Cl2 O(g) + F20(g) 2 CIF3 (g) + 202(g) → Cl2 0(g) +…
A:
Q: Calculate AH f co(s) from the following information: CO2(9) AH = -365KJ %3D Given: C(s) + O2(g) COs)…
A:
Q: The combustion reaction of octane is represented by the following equations: 2C8 H18 (1) + 25 O2 (g)…
A: The enthalpy change is a state function and the Hess law states that the change of enthalpy in a…
Q: What will be the final temperature of a mixture made from 25.0 g of water at 15.08C, 45.0 g of water…
A: When two or more substances(liquids in this situation) at different temperatures are mixed, then…
Q: 1. Calculate AH for the combustion of methane (CH4) Original equation: CH4 + 202 → CO2 + 2 H2O…
A: Enthalpy change of the reaction is equal to the sum of enthalpies of the given reactions.
Q: 2AF(g)+ 0,(g)→ A,0(g) + F0(g) 2AF,(g)+20,(g)→ Cl,0(g) +3F,0(g) 2F,(g) + 0,(9)-2F,0(g) AH = 167.4kJ…
A: Interpretation - To determine the enthalpy change (∆H ) for the final reaction and also draw…
Q: A 100.0 mL sample of 0.200 M aqueous hydrochloric acid is added to 100.0 ml of 0.200 M aqueous…
A: ∆H=-qn where q=heat of reaction,n=moles of Hclbut…
Q: Complete the following equations: CH;CHCICOOH + NH2CH2CH3 ----100°C----> CH;CH,CH;COOH + NH3…
A: We know that, when a carboxylic acid undergoes reaction with a primary amine at 100 oC to produce a…
Q: From the following enthalpies of reacion, calculate the AH for the reaction in kJ of CF4(g) with…
A:
Q: Given the following data 2 CIF(g) + O2(g) Cl2 O(g) + F20(g) 2 CIF, (9) + 202 (g) C, O(g) + 3F20(g)…
A: Interpretation: From the given reaction, the ∆H of the given equation is to be calculated.
Q: Problem attached
A: The balanced reaction taking place is given as, => 4 NH3 (g) + 5 O2 (g) -----> 4 NO (g) + 6…
Q: 150 ml of .350M HCl was mixed with 150 mL of .350 M NaOH in a calorimeter of negligible heat…
A:
Q: Hydrogen sulfide, H2S is a foul-smelling gas which burns toform sulfur dioxide 2H2S(g)+…
A: H2S is hydrogen sulphide and in this reaction moles of Hydrogen sulphide is present
Q: Phereseadula has to determine the specific heat capacity of milk. He uses a constant volume…
A: given values specific heat = Cp heat capacity = C = 758 J/K initial temp = Ti =22.5 oC final…
Q: For the following reaction the enthalpy of the reaction is SHr-35.5 kJ 2A-38- 5C if 8 moles of A…
A: The balanced reaction taking place is given as, => 2 A + 3 B → 5 C…
Q: Given the following data: Solution Conc. (M) Volume (ml) Tiitial ("C) Ttnal NaOH 0.750 50.00 19.84…
A: The detailed solution of your question given below-- Since ,we know that in this process the…
Q: Syngas can be burned directly or converted to methanol. Calculate AH° for the reaction CO(g) + 2H2…
A: The standard enthalpy of formation is the enthalpy change of reaction in standard conditions when…
Q: Determine AG rxn in kJ for 3X(g) + 2Y(g) = Z(g) + 3M(g) given the following information. Substance X…
A:
Q: From the following enthalpies of reacion, calculate the AH for the reaction in kJ of C2H4(g) with…
A:
Q: 3 Fez03 + Co → 2FE304 + CO2 Given the following information: Fe203 + 3 CO → 2 Fe t3 CO2 AH = -5.7KJ…
A:
Q: If the needed reaction is 4C --> D and the given reaction is D --> 4 C with ΔH = 134.7 kJ,…
A: If the needed reaction is 4C → D and the given reaction is D → 4 C with ΔH = 134.7 kJ, then the ΔH…
Q: HCI(aq) + Ba(OH)2(aq) --> Bacl2(aq) + 2 H20(1) AH= -118 kJ Calculate the heat when 114.0mL of 0.50OM…
A: a) Moles of HCl can be calculated as., 0.500 M HCl = 0.500 moles/ 1 L we have, 114 mL, which is…
Q: Considering the following reaction: 2CO(g) + O2(g) --> 2CO2(g) AH° = -258 kJ What is the AH° for the…
A: The enthalpy of the reaction is considered as the state function that is it depends only on the…
Q: Calculate the AH° for the reaction: 2C2H6 (g) + 7 O2 (g) 4CO2 (g) + 6H2O (g) Using the following…
A: Target equation is : 2C2H6(g) + 7O2(g) = 4CO2(g) + 6H2O(g) Applying Laplace-Lavoisier law and…
Q: Calculate AG° (in kJ) for the reaction below at 36°C. 2 SO2(g) + O2(g) 2 SO3(g) Enter your result as…
A: Given 2SO2(g) + O2(g) -----> 2SO3(g) Temperature = 36°C = 309°K
Q: Given the following data 2 CIF (g) + O2 (9) → Cl2 O(g) + F20(g) 2 CIF3 (g) + 202(9) → Cl2 O(g) +…
A: ClF(g) + F2(g) --> ClF3(g)
Q: Diborane (B2H6) is a propellant. combustion reaction of diborane is as follows: B2H6(s) + 302(g) –…
A: Given, mass of diborane cmbusted = 198 g molar mass of diborane (B2H6) = 27.66 g/mol we know,…
Q: The following reaction is being studied: MX (s) ⟶ M+ (aq) + X-(aq) ∆Hrxn = ??? kJ/mol MX To find…
A: Given that : Initial temperature of DI water = 22˚C Final temperature of solution = 24.5 ˚C Also,…
Q: ith the given data: Initial temperature = 30oC; Final temperature = 50oC Initial volume of water =…
A: #(a): Volume of 70% isopropyl alcohol burnt, V = 200 mL - 195 mL = 5 mL Volume of isopropyl…
Q: 2. Calculate AH for the reaction: CH4 (g) + 4 Cl2 (g) CCI4 (g) +4 HCI (g) Use the following…
A: We have to calculate ∆Hrxn for the reaction CH4(g) + 4Cl2(g) → CCl4(g) + 4HCl(g).
Q: The reaction of ammonia(g) with oxygen(g) to form nitrogen monoxide(g) and water(g) proceeds as…
A: 4NH3(g) + 5O2(g) --> 4N2O(g) + 6H2O(g) Molecular Mass of NH3 = 17 g mol-1 therefore, Energy…
Q: Given that H2(g)+F2(g)⟶2HF(g) Δ?∘rxn=−546.6 kJ 2H2(g)+O2(g)⟶2H2O(l)…
A:
Q: Calculate AHrxn for the following reaction: C(s) +H20(g)→CO(g) +H2 (g) Use the following reactions…
A:
Q: Determine the value of AH°xn for the target equation by using the three reactions provided. Target…
A: The given data is as follows: Target equation: 2A + D → F A + B → C ∆H1=267 kJ…
Q: Nitromethane, CH3NO2, can be used as a fuel. When the liquid is burned, the (unbalanced) reaction is…
A: The balanced reaction will be written as: 4CH3NO2 (l) + 3O2(g) → 4CO2 (g) + 2N2 (g) + 6H2O (g) These…
Q: Calculate AH for the reaction: N0 g) +O (g)NO2 (g)- 203 (a)3 02 (a) O2 (0) 20 ( NO 9) + O3 (g) - AH…
A:
Q: Given the following data: 2 C6H6(l) + 15 O2(g) → 12 CO2(g) + 6 H2O(l) ΔG0= -6399 kJ…
A: Finding ∆G°
Q: I2(g) + Cl2(g) ⇌ 2 ICl(g) Calculate ΔGrxn in kJ at 25°C from the following ΔG°f values. ΔG°f (I2) =…
A: Given information: ΔG°f (I2) = 62.42 kJ/mol ΔG°f (Cl2) = 0 kJ/mol ΔG°f (ICl) = -5.52 kJ/mol We have…
Q: Given the following thermochemical reactions: AH° = -818.3 kJ N2H4() + 2H2O2(g) → N2(g) + 4H20()…
A:
Q: When 1 mole of P4O10(s) reacts with H2O(l) to form H3PO4(aq) according to the following equation,…
A: Given data: 1P4O10(s) + 6H2O(l) -------->> 4H3PO4(aq) + 453kJ Exothermic reaction: A reaction…
Q: I2(g) + Cl2(g) ⇌ 2 ICl(g) Calculate ΔGrxn in kJ at 25°C with the conditions PICl = 2.55 atm, PI2 =…
A: Since ΔGrxn = ΔG°rxn + RT ln Q where Q = reaction quotient given by Q = PICI2 / [ PI2 X PCl2 ]…
Q: Zeroed out mass of calorimeter and themometer Got a 50 mL graduated cylinder. Measured 25 mL of…
A: Heat of dissolution can be calculated from the temperature change occurred in the water after…
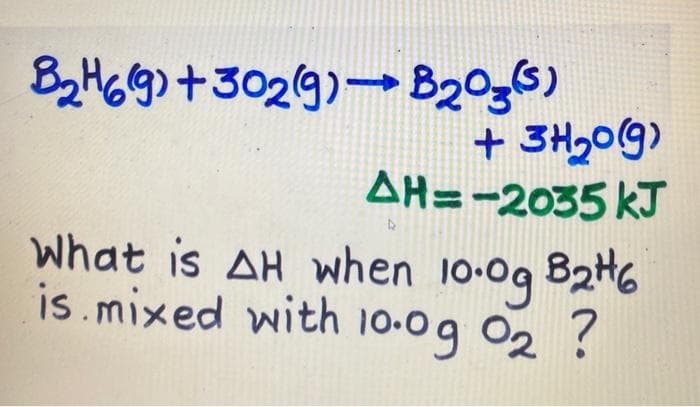
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Explain why it is problematic to include a constraint such as the following in an LP model for a blending problem: Total octane in gasoline 1 blend $ divided by Barrels of gasoline 1 blended daily which is greater thanor equal to 10Oceanic uptake of carbon dioxide is thus described:CO2 (g) + H2O ⇔ H2CO3, K = [H2CO3]/PCO2 = 3 x 10-2 M atm-1 H2CO3 ⇔ HCO3- + H+, K = [HCO3-][H+]/[H2CO3] = 9 x 10-7 moles/LHCO3- ⇔ CO32 - + H+, K = [CO32 -][H+]/[HCO3-] = 7 x 10-10 moles/LCharge balance equation:[H+] = [OH-] + [HCO3-] + 2[CO32 ] If the CO2 concentration in the atmosphere is 300 ppm, what is the pH of the ocean?1) Commercial fuming Sulphuric acid (Oleum-H2S2O6) is 99.9% solution. Please convert it into molarity.2) Find out the Volume (dm3) of product (gas) at RTP when 0.58 M, 150 mL NaOH (aq.) reacts with 350 mL, 0.25 NH4Cl.
- Calculate deltaH1 Please provide 3 sig figs, units are KJ/mol, please only give number value in answer. Mg(s) + 1/2O2(g) --> MgO(s) deltaH1 = ? Mg(s) + 2H+(aq) --> Mg2+(aq) + H2(g) deltaH2 = +10.1 kJ/mol MgO(s) + 2H+(aq) --> Mg2+(aq) + H2O(l) deltaH3 = -5.00 kJ/mol 1/2O2(g) + H2(g) --> H2O(l) deltaH4 = -286.0 KJ/molWhich of the following is a spontaneous reaction.? a. Rxn with ΔH =- 10Kj/mol ΔS= -5J/mol T= 300K b. NaCl +H20 -> NaOH + HCl 25C c. H20(l) -> H2O(s) Temp: 25C d. Dissolution of 100g of solid sugar in 100 mL ice tea. Consider following reaction: HgO (s) -> Hg(l) + ½ O2 (g) Delta H = +90.7 kj/mol. What quantity of heat in kj/mol is required to produce one mole HgO? Write your answer without units. Given the following data 2ClF(g) + O2(g) --> Cl2O(g) + F2O (g) Delta H= 167.4 kJ I 2ClF3(g) + 2O2(g) --> Cl2O(g) + 3F2 O (g) Delta H= 341.4 kJ II 2 F2(g) + O2(g) ---> 2F2O (g) Delta H= -43.4 kJ III Calculate the delta H in kJ for below reaction: ClF(g) + F2(g) ---> ClF3(g)6 I- + BrO3- + 6 H+ ----- 3 I2 + Br- + 3 H2O Rate= k[I-]m[BrO3-]n[H+]p rate= (∆[I2])/(3*∆time) [I-] 0.010M [S2O3-2] 0.0010M H2O [BrO3-] 0.04M HCl 0.10M Starch Indicator (sec.) Volume, ml Volume, ml Volume, ml Volume, ml Volume, ml 1 10 10 20 10 10 1-3 drops 301.90 2 20 10 10 10 10 1-3 drops 213.28 3 10 10 10 20 10 1-3 drops 214.92 4 10 10 10 10 20 1-3 drops 86.45 Temperature of Reaction: _______20 C________ 1. Using the dilution equation (M1V1 = M2V2) calculate the concentration of the I-, BrO3- and the H+ (HCl) in the final mixture just prior to any reaction. 2. Calculate the rate for each experiment as explained in the background information. 3. Using the information above determine the rate law (m, n and p) for the reaction. Show all calculations/reasoning in your notebook. m_____________ n_________________ p…
- 1. What is the mEq of silver nitrate? 170 0.0170 0.170 17.0 2. How much (mL) silver nitrate is required for the standardization? 4.995 49.950 49.590 45.990 * MW of silver nitrate = 170 g/mol * Please take note that Im looking for the silver nitrateBeaker 0.00200 M Fe(NO3)3, mL 0.00200 M NaSCN, mL total volume, mL 1 3.000 2.000 10.00 2 3.000 3.000 10.00 3 3.000 4.000 10.00 4 3.000 5.000 10.00 5 (blank) 3.000 0.000 10.00 In Solutions 1-4 you are adding successively larger volumes of 0.00200M SCN- to the Fe3+ solution and diluting to 10.00 ml. Calculate the final diluted molarity of SCN- in solution #1 Your answer should have 3 sig figs =Pls solve this problem and I upvote. Do not reject if it's hard for you skip it so one of the expert can do it. Thank you. I pray for your wellbeing
- When drinking water is disinfected with chlorine, an undesired byproduct, chloroform (CHCl3), may form. Suppose a 70-kg person drinks 2 L of water every day for 70 years with a chloroform concentration of 0.08 mg/L (the drinking water standard). Potency factor for chloroform = 6.1 x 10-3 (mg/kg-day)-1 The upper-bound cancer risk for this individual. a. 0.545 b. 5.45x10-6 c. 5.45x10-4 d. 5.45x10-10 e. 14x10-6 If a city with 300,000 people in it also drinks the same amount water described in the above question, how many extra cancers per year would be expected? Assume the standard 70-year lifetime. a. 0.163 b. 1.63x10-4 c. 1.63x10-3 d. 1.63x10-6 e. 6x10-2From this given value of Kc: 2H2S (g) <-------> 2H2 + S2 Kc=2.3 X 10-4 at 1404K What is Kc for: H2 + 1/2S2 <--------> H2Spneumonia is characterized by fever (a body temperature) does the patient have a body temperturr that indicates that he has pneumonia? justify your answer with suitable caculations.





