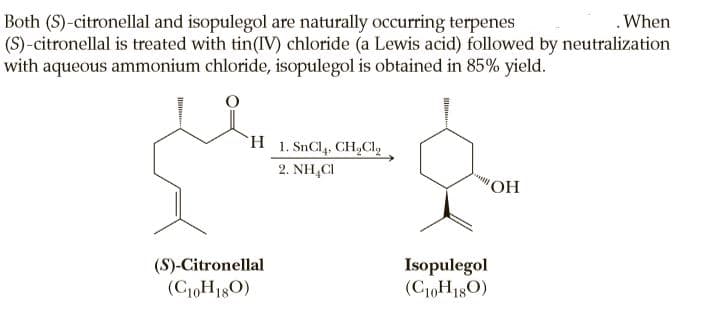
Both (S)-citronellal and isopulegol are naturally occurring terpenes (S)-citronellal is treated with tin(IV) chloride (a Lewis acid) followed by neutralization with aqueous ammonium chloride, isopulegol is obtained in 85% yield. .When H 1. SnCl4, CHCI, 2. NH,CI (S)-Citronellal (C10H180) Isopulegol (C10H180)
Q: Draw a structural formula for an alkene with the indicated molecular formula that gives the compound…
A:
Q: 2. An ether is reacted with H2SO4 at 140°C. What is the likely outcome? an alkene will be formed O a…
A: Given, an ether is reacted with H2SO4 at 140 °C
Q: Q5.) Two isomeric aromatic compounds X and Y have the following percentage composition by lmass! C =…
A:
Q: Propose two possible structures for a hydrocarbon having an exact mass of 96.0939 that forms…
A: NOTE : There will be 3 different structure are possible for a hydrocarbon having an exact mass of…
Q: The structure shown below is that of one of the geometric isomers (the E- isomer) of the…
A: In the given question a molecule is given and it is asked that why this is assigned as E-isomer.…
Q: What does alkyl halides gives on heating with dry Ag2O ?
A:
Q: How many of the carbon-carbon double bonds in lycopene have the possibility for cis,trans isomerism?…
A: It is a term referred to those compounds which contain at least one or more asymmetric/chiral…
Q: When C6H14 is reacted with Br2(g) in the presence of ultraviolet light, four different…
A: According to our guidelines sir/madam, We are allowed to give the answers to the first three…
Q: Name the nitrile(s) with formula C6H1N that contain two me I branches on the same carbon of the main…
A: another name of cyanides is nitrile For this compound ,According to IUPAC naming rule CN group…
Q: 3. Write the structure of the alkene X, that forms the compound below, when treated with ozone (O3)…
A:
Q: 5. (a) Compound A with molecular formula C,Hs is produced when benzene reacts with chloromethane in…
A: Answer:- This question is answered by using the simple concept of chemical reaction of organic…
Q: For small-scale and consumer welding applications, many hardware stores sell cylinders of MAAP gas,…
A: SOLUTION: Step 1: The gas mixture of propyne and 1,2-propadiene could be prepared from a double…
Q: An unknown alcohol C6H14O gave a rapid Lucas Test. It could not be oxidized easily, but upon…
A: DBE of the alcohol = (C+1)-H/2 = (6+1)-14/2 = 0. Hence, it is an aliphatic alcohol. According to…
Q: Consider the structure of cyclohexyne, if it undergoes acetylide reaction with NaNH2 and 2 moles of…
A: Consider the structure of cyclohexyne, if it undergoes acetylide reaction with NaNH2 and 2 moles of…
Q: 5. An unknown hydrocarbon A with the formula C6H12 reacts with 1 molar equivalent of H2 over a…
A: The given unknown hydrocarbon A with formula C6H12reacts with 1 molar equivalent of H2 over a Pd…
Q: Compound A has the formula C gH 8. It reacts rapidly with KMNO 4 to give CO 2 and a carboxylic acid,…
A: C8H8 : Degree of Unsaturation (DOU) = (2C+2 - H )/ 2…
Q: An unknown alcohol with a molecular formula of C7H14O was oxidized to an aldehyde with HOCl. When an…
A: With Molecular formula C7H14O, there could many possibilities of alcohol. But as the question hints,…
Q: What is the major product of the reaction when the alkane reacts with Cl2 instead of with Br2?…
A: Given: The given alkane reacts with Cl2 instead of Br2.
Q: Compound A has the formula C8H8. It reacts rapidly with KMNO4 to give CO2 and a carboxylic acid, B…
A:
Q: When 3-ethylcyclohexene is reacted with one equivalent of bromine, the product would have bromine…
A: Alkene reacts with bromine to form dibromoalkane. The reaction proceeds by the bromonium ion…
Q: important in photosynthesis; allows plant to obtain energy from A. Analyte E sunlight B. Analyte C v…
A: 1) important for photosynthesis, allows plants to obtain energy from sunlight - analyte A Actually…
Q: Alkenes can be converted to alcohols by reaction with mercuric acetate to form a…
A:
Q: Give the product(s) formed when 1-butene reacts with the following: (a) HBr (b) dilute…
A: The reactant given is 1-butene. And the reagents given are, (a) HBr (b) dilute H2SO4 (c) Br2 in…
Q: Dimethyl ether (CH3OCH3) and ethanol (CH3CH2OH) are isomers, butCH3OCH3 has a pKa of 40 and CH3CH2OH…
A: The structures of two isomers are as follows- It is given that pKa of dimethyl ether is 40 and…
Q: 9. There are several possible forms of a trisubstituted cyclohexane with the formula C10H200. I have…
A: To identify the most and least commonly naturally occurring compound.
Q: Upon successful bromination of an alkene, what is the molecular weight increase of the compound?
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Benzene is one of the compounds used as octane enhancers in unleaded gasoline. It is manufactured by…
A: Equilibrium constant (Kc ) for above reaction can be given by Kc = [C6H6] / [C2H2]3
Q: Draw a structural formula for the cycloalkene with the molecular formula C6H10 that reacts with Cl2…
A: In a chemical reaction; the substance which involves in conversion is said to be reactant whereas…
Q: New hydrocarbon compounds K, L, M and N have been formed from an organic reactions in Dr. Meredith…
A:
Q: _CH=CH₂ _CH=CH₂ and CH3O 0₂N₁ A B Which will be true about the reaction of the two alkenes with HBr?
A: ->The reaction of alkene with HBr proceeds through formation of carbocation intermediate.Hence ,…
Q: 1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL…
A: Given, Density of 1,4-Pentadiene = 0.66gmL molar mass of 1,4-Pentadiene = 68.12gmol volume of…
Q: (S)-2-butanol reacts with potassium dichromate (K2CIO4) in aqueous sulfuric acid to give A(C,HgO).…
A: K2CrO4 is an oxidising agent. Ethyl magnesium bromide is grignard reagent. Here we have to find out…
Q: Why are sh Does the compound below undergo saponification reaction? Why? C-OCHCH, CH, nronoro an…
A: Saponification is the process in which formation of soap takes place. In this process, alkaline…
Q: An ether solution of cyclohexane-1,2-dicarboxylic acid (CDA), a compound with two carboxyl (COOH)…
A: The balanced chemical equation for the complete detprotonation is show below:
Q: What alkane, with molecular formula C7H16, forms seven monochlorinated products (disregarding…
A: Monochlorinated products are those where substitution of one chlorine in alkane product gives…
Q: An unknown hydrocarbon A with the formula C6H12 reacts with 1 molar equivalent of H2 over a…
A: Reduction of alkene gives alkane.
Q: Compounds A and B are isomers of the molecular formula CgH19Br. Both yield the same alkene C in an…
A: In this question, we want to determine the Compound A, B and Alkene-C You can see details Solution…
Q: Draw a structural formula for an alkene and a dichloroalkane with the given molecular formula that…
A:
Q: (a) Octane burns with oxygen gas. (b) 3-ethylpentan-2-ol is combined with an oxidizing agent, H2O2.…
A: a) Octane burns with oxygen gas. The equation for the given reaction is, 2C8H18 + 25O2 ---> 16CO2…
Q: The reaction of (Z)-3-Methyl-3-hexene with H2O/H2SO4 produces 3 – Methyl-3-hexanol (75 % yield) (a)…
A: (Z)-3-Methyl-3-hexene reacts with H2O/H2SO4 and undergoes acid-catalyzed hydration to produce…
Q: One of the following statements is incorrect in using Simmons-Smith reagent to prepare cyclopropane:…
A:
Q: 3CH2CH2CH2C(CH3)2 a this alcohol be synthesized selectively by hydrobor If yes, draw the structure…
A: In Hydroboration-Oxidation Reaction anti-markonikov rule follow. Anti-markonikov rule says that…
Q: ) Name the reactant (common and IUPAC) and show the structures for A and B 1) Excess CH,CH,MgBr А OH…
A:
Q: mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude…
A:
Q: Selact the compound which forms methanol on warming with dilute NAOH solution. -CH2CH3 A CH;CH2-…
A: Base hydrolysis of ester produces sodium salt of carboxylic acid and alcohol.
Q: 9. 1-Butanethiol, CH3CH2CHCH2SH, is the product from the reaction of 1-bromobutane and ???? Draw the…
A:
Q: Draw a structural formula for an alkene and a dichloroalkane with the given molecular formula that…
A: Alkene can be converted to alkyne by treating it with sodamide which removes two hydrogen atoms from…
Q: Alkenes can be converted to alcohols by reaction with mercuric acetate to form a…
A: In the oxymercuration reaction, the addition of Hg(CH3COO)2 and OH occur via the anti-addition…
Isopulegol is formed as a single stereoisomer. Account for the fact that only a single stereoisomer is formed
Step by step
Solved in 2 steps with 3 images

- 4. Compound A has the formula C 8H 8. It reacts rapidly with KMnO 4 to give CO 2 and a carboxylic acid, B (C 7H 6O 2), but reacts with only 1 molar equivalent of H 2 on catalytic hydrogenation over a palladium catalyst. On hydrogenation under conditions that reduce aromatic rings, 4, equivalents of H 2 are taken up and hydrocarbon C (C 8H 16) is produced. What are the structures of A, B, and C.A chemist allows some pure (2S,3R)-3-bromo-2,3-diphenylpentane to react with a solution of sodium ethoxide(NaOCH2CH3) in ethanol. The products are two alkenes: A (cis-trans mixture) and B, a single pure isomer. Under thesame conditions, the reaction of (2S,3S)-3-bromo-2,3-diphenylpentane gives two alkenes, A (cis-trans mixture) and C.Upon catalytic hydrogenation, all three of these alkenes (A, B, and C) give 2,3-diphenylpentane. Determine the structuresof A, B, and C; give equations for their formation; and explain the stereospecificity of these reactionsCembrene, C20H32, is a diterpenoid hydrocarbon isolated from pine resin. Cembrene has a UV absorption at 245 nm, but dihydrocembrene (C20H34), the product of hydrogenation with 1 equivalent of H2, has no UV absorption. On exhaustive hydrogenation, 4 equivalents of H2 react, and octahydrocembrene, C20H40, is produced. On ozonolysis of cembrene, followed by treatment of the ozonide with zinc, four carbonylcontaining products are obtained: Propose a structure for cembrene that is consistent with its formation from geranylgeranyl diphosphate.
- Nitromethane is reacted with ethyl prop-2-enoate with EtO-Na+, EtOH 3 equivalents to give the product X(C16). X then reacts with H2/Raney Ni to give the product Y(C14), which in turn reacts with Na to give Z (C12). Indicate which products X, Y and Z are.An unknown hydrocarbon A with the formula C6H12 reacts with 1 molar equivalent ofH2 over a palladium catalyst. Hydrocarbon A also reacts with OsO4 to give diol B. When oxidized with KMnO4 in acidic solution, A gives two fragments. One fragmentis propanoic acid, CH3CH2CO2H, and the other fragment is ketone C. What are thestructures of A, B, and C? Write all reactions and show your reasoning.A chemist allows some pure (2S,3R)-3-bromo-2,3-diphenylpentane to react with a solution of sodium ethoxide(NaOCH2 CH3) in ethanol. The products are two alkenes: A (cis-trans mixture) and B, a single pure isomer. Under the same conditions, the reaction of (2S,3S)-3-bromo-2,3-diphenylpentane gives two alkenes, A (cis-trans mixture) and C. Upon catalytic hydrogenation, all three of these alkenes (A, B, and C) give 2,3-diphenylpentane. Determine the structures of A, B, and C; give equations for their formation; and explain the stereospecificity of these reactions.
- Compound A (C11H23Br) is a secondary alkyl halide. On being heated with a solution of sodium ethoxide in ethanol, compound A yielded a mixture of two alkenes B and C, each having molecular formula C11H22. Catalytic hydrogenation of the major isomer B or the minor isomer C gave only 3,5-diethylheptane. Draw structures for compounds A, B, and C consistent with these observations.An unknown alcohol with a molecular formula of C7H14O was oxidized to an aldehyde with HOCl. When an acidic solution of the alcohol was distilled, two alkenes were obtained. The alkene formed in greater yield was determined to be 1-methylcyclohexene. The other alkene formed the original un-known alcohol when treated with BH3/THF followed by H2O2, HO-, and H2O. Identify the unknown alcohol.Compound AA, C6H14O, reacts very fast and immediately form a cloudy solutionwith Lucas reagent. Compound BB, an alkene is formed when compound AAreacted with concentrated sulfuric acid, H2SO4.Ozonolysis process of compoundBB will produce only propanone. Reduction of propanone with Nickel, Ni givescompound CC. Esterification of compound CC with propanoic acid producescompound DD. Draw the structural formula of compounds AA, BB, CC, and DD
- Oximene and myrcene, two hydrocarbons isolated from alfalfa that have the molecular formula C10H16, both yield 2,6- dimethyloctane when treated with H2 and a Pd catalyst. Ozonolysis of oximene forms (CH3)2C = O, CH2 = O, CH2(CHO)2, and CH3COCHO. Ozonolysis of myrcene yields (CH3)2C = O, CH2 = O, (two equiv), and HCOCH2CH2COCHO. Identify the structures of oximene and myrcene.1. Using Br2 in C2H4Br2 will result in HBr and ______. a. C2H3Cl3 b. C2H4Cl3 c. C2H2Cl3 d. none of the above 2. How many halogenation are posible in propane? a. 3 b. 8 c. 6 d. 10 3.Sulfonation of pentane will result in ________ and water. a. C5H11SO3H b. C5H12SO3H c. C5H14SO3H d. none of the above 4.Nitration of hexane will result in ________ and water. a. C6H13SO3H b. C6H15NO2 c. C6H13NO2 d. C6H14NO2 5.How many moles of O2 in heating a C12H26 (dodecane) a. 27 b. 37 c. 24 d. none of the aboveWrite out all the isomers of the compound with molecular formula C4H10O Select the normal/ primaryisomer and treat it with conc. H2SO4 and heat. Identify the reaction and give the product ‘A’ from it. Treatment of ‘A’ with HCl/H2O gives ‘B’ Treatment of ‘A’ with cold KMnO4/ -OH gives ‘C’ Treatment of ‘A’ with Hot KMnO4/-OH gives ‘D’ followed by acidification of the mixture to give ‘E’

