Q: name both c and d H₂C C. H₂C d. CH₂ CH₂1 CH 3 CH3 CH 3 CH₂ CH₂ \ CH3
A: Number of C-atoms. Name 1 meth 2 eth 3 Prop 4 But 5 Pent 6 Hex 7 Hept If all C-C bond…
Q: label each pair of structures below as Constitutional isomers (C), diastereomers (D), Enantiorners…
A: To answer the given questions, one must know the definitions of constitutional isomers, enantiomers…
Q: 0.641 g of a semi-synthetic alkaloid was dissolved in 25 ml of 1% w/v acetic acid and was analysed…
A: Ans 1. To calculate the level of impurity in % w/w, we need to first calculate the amount of…
Q: on 30 What is the pH in the titration of 15.0 mL of 0.40 M acetic acid (CH3COOH, K₂ = 1.8 x 10-5)…
A: Given data- Volume of acetic acid = 15.0 mL Concentration of acetic acid = 0.40 M Ka of acetic…
Q: A sample of pure water is saturated with PbCl2. In this saturated solution, which of the following…
A:
Q: The conjugate base of 19.27 forms the complex [COL3] which has mer- and fac-isomers. (a) Draw the…
A: The mer-isomer has the three ligands arranged around the metal ion in a trigonal meridional fashion…
Q: Macmillan Learning Organic Chemistry Maxwell Draw the two major organic products for the reaction:…
A:
Q: H H H H S ZIH О H Н I- H I Н H н H-S н +0-I HIN I І Н I Т
A: Acidity is defined as the ability of a molecule to release protons. if a compound easily donate…
Q: 2. a) b) c) d) Complete the following reactions: Br НО OH + OH да ОН + + жон OH OH OH H₂SO4 Д H₂SO4…
A: Acid react with alcohol to give ester
Q: Identify the major product(s) for the following reaction. 01 Oll O III I and II Oll and Ill 42- HCI…
A: This is 1,2 addition which occur at low temperature 0• C This is 1,4 addition which occur at high…
Q: Identify the following substance as aromatic, anti-aromatic, or non-aromatic. N -N CH3 A) aromatic…
A:
Q: What are the equilibrium concentrations of 250mg of acetic acid, HC₂H3O₂ in 150ml solution? (acid is…
A: Question Based on the concept of chemical equilibrium . we need to calculate equilibrium…
Q: For many purposes we can treat ammonia (NH₂) as an ideal gas at temperatures above its boiling point…
A: Given T1 = -23°C = 273.15 - 23 = 250.15 K T2 = ? Let P1 be x So, P2 = 3x
Q: What is meant by highly concentrated solution?
A: The meaning of highly concentrated solution is given below.
Q: T 112. 5. If 245mL of oxygen is produced at STP, how many grams of hydrogen peroxide, H₂O₂ were…
A:
Q: 1) Draw below eclipsed and staggered conformations of ethane in Newman projection. Eclipsed Build a…
A: In a Newman projection, the molecule is viewed along the axis of a specific carbon-carbon bond. The…
Q: Carbon disulfide is prepared by heating sulfur and charcoal. The chemical equation is S₂(g) + C(s) =…
A:
Q: The original concentration of a solution is 200 mg/dL. The solution was diluted 1:5. Then one part…
A:
Q: If you place 1.0 g of the complex in a 62.5 mL flask at 125 °C, what is the total pressure in the…
A: Given that: Mass of the complex = 1.0 g Volume of flask = 62.5 mL Temperature = 125oC
Q: Consider the following data on some weak acids and weak bases: acid name formula acetic acid HCH,CO,…
A: The question is based on the concept of acidity. we need to arrange the given salts in order of…
Q: For the reaction: N2O4 (g) ---> 2NO2 (g) If a reaction vessel initially contains an N2O4…
A:
Q: 20.36 (a) Which of the following complexes would you expect to suffer from a Jahn-Teller distortion:…
A:
Q: Consider the following neutralization reaction between a strong acid and a strong base (incomplete…
A: The balanced equation for the reaction between HCl and Ca(OH)2 is 2 HCl (aq) + Ca(OH)2 (aq)…
Q: Calculate the pH of a solution containing 0.010 M HCOOH (Ka = 1.90 × 10-4) and 0.061 M HCI. A. 0.39…
A:
Q: What is the pH of an aqueous 0.25 M NaBrO solution? Ka(HBrO) = 2.0 × 10-9
A:
Q: A chemical engineer is studying the following reaction: N₂(g) + 3H₂(g) → 2 NH3(g) At the temperature…
A: The direction in which the reaction proceeds to reach equilibrium can be predicted by determining…
Q: Consider the compound beryllium fluoride. Draw the Lewis-Dot Structure for each ion and show how…
A: This question belongs to Chemical Bonding.
Q: The equilibrium H₂(g) + 12(g) 2HI(g) has a Kc = 49 at a certain temperature. If 1.14 moles of H₂…
A:
Q: Write the correct formulas for the following compounds 1. Phosphorus tribromide 2. Chlorous acid 3.…
A: Since, Naming the compound,first write name of cation then after anion. Thus,
Q: The line drawings for the cis-trans isomers of a compound are given below. Omitting the cis-trans…
A: IUPAC nomenclature is used for naming the organic compound as recommended by international union of…
Q: ow many grams of Mg(OH)2 is necessary to prepare 500mL of 0.5M Mg (OH)2 solution? FW of Mg(OH)2 =58…
A: Given, Volume of solution = 500 mL = 500 mL × ( 1 L ÷ 1000 mL ) = 0.500 L molarity of Mg(OH)2 =…
Q: the number of observed unpaired electrons (stated after the formula): 12+ (c) [Cr(en)3] (4); (d)…
A: This question belongs to Coordination chemistry. As per Bartley Q&A guidelines I can answer…
Q: Calculate the pH of the resulting solution when 20.0 mL of 0. 150 M NaOH is added to 50.0 mL of…
A:
Q: The formation constant of a complex ion [M(NH3)4]²+ (aq) is 4.7E8. If a solution is prepared by…
A: The chemical reaction representing the formation of the complex is M2+(aq) + 4NH3(aq) ⇔[M(NH3)4]2+…
Q: For the reaction below, Kc = 0.60 at 550 K. The reaction starts with a 0.050 M concentration of…
A: Given : Kc = 0.60 Initial Concentration of A2B2 = 0.050 M Temperature = 550 K
Q: Gaseous ethane (CH₂CH3) reacts with gaseous oxygen gas (0₂) to produce gaseous carbon dioxide (CO₂)…
A: Given : Mass of ethane reacted = 0.90 gm Mass of oxygen gas reacted = 5.6 gm Mass of carbon…
Q: Assume the volume of the filter flask is 250.0mL, you added a total 35mL of solution so the initial…
A: states that the volume of a gas is directly proportional to its temperature at constant pressure…
Q: A certain substance X has a normal freezing point of 8.1 "C and a molal freezing point depression…
A:
Q: 650 mL of a liquid is heated from 25.0 oC to 100.0 oC. The density of the liquid is 2.00 g/mL. The…
A:
Q: A) B) 1) 9- 2) H2O2, NaOH فه C) D) you
A:
Q: c. Draw a potential energy diagram for the formation of NO₂(g): N₂(g) +20₂(g) → 2NO₂(g). A Hreaction…
A: We will Draw a potential energy diagram for the formation of NO2(g): N2(g) + 202(g) → 2NO2(g),…
Q: 8.3) The chemical equation for the reaction of nitrogen with hydrogen to produce ammonía is shown…
A: When the reaction begives, there will be only reactants. As the reaction progresses, reactants start…
Q: Consider the potential energy diagram in the 350 kJ 250 kJ 200 kJ 100 kJ Photo What is the…
A: Given, Average energy of reactants A, Ereactants = 450 kJ Maximum amount of energy required to cross…
Q: CO 40: Predict and draw the major and minor products for the reactions of alkynes, including…
A: Answer : The major products of the given reaction are listed below :
Q: 6) For the reaction shown below, predict whether the reactants or the products are predominant at…
A: For this reaction Keq =4.9*10-10
Q: Draw the structure(s) of the major organic product(s) of the following reaction. Dilute aqueous HCI…
A:
Q: If 180g of NaCL is added to 200 mL of solution, what is the resulting concentration?
A:
Q: 2CO(g) + O₂(g) → 2CO₂(g) 9.0 L of O2 react with excess CO at STP. How many moles of CO2 form during…
A: To solve this problem we have to calculate the mole of CO2 produced .
Q: How many atoms of vanadium are in 1.28 grams of vanadium?
A: Given : Mass of vanadium = 1.28 gm Also, molar mass of vanadium = 50.94 g/mol
Q: Identify each of the following as acidic, basic, or neutral: 1. a solution where the H3O+…
A: Here, we have to identify the given solutions as acidic, basic, or neutral.
Please show the mechanisms and major product for the reactions below
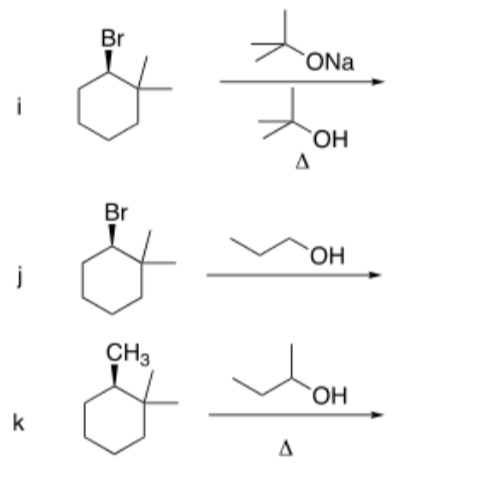
Step by step
Solved in 6 steps with 2 images


