Br -CH3 Но. NH2 1 What will be the appearance of the benzene signals in the 'H NMR spectrum of compound 1? O simple triplet O two doublets a complex multiplet one singlet one doublet 2)
Br -CH3 Но. NH2 1 What will be the appearance of the benzene signals in the 'H NMR spectrum of compound 1? O simple triplet O two doublets a complex multiplet one singlet one doublet 2)
Chapter13: Structure Determination: Nuclear Magnetic Resonance Spectroscopy
Section13.SE: Something Extra
Problem 65GP: 3-Methyl-2-butanol has five signals in its 13C NMR spectrum at 17.90, 18.15, 20.00, 35.05, and 72.75...
Related questions
Question
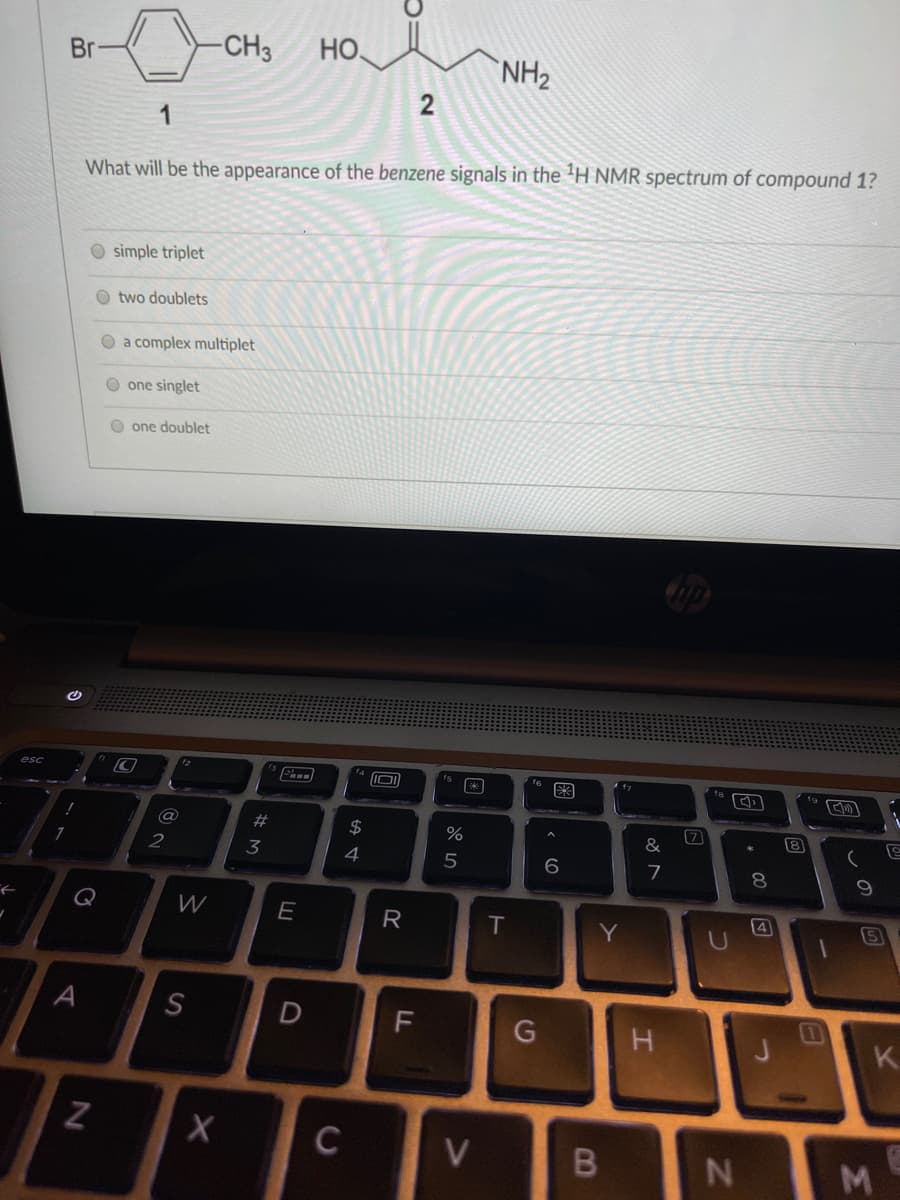
Transcribed Image Text:-CH3
Но.
NH2
Br
2
1
What will be the appearance of the benzene signals in the 'H NMR spectrum of compound 1?
O simple triplet
two doublets
O a complex multiplet
one singlet
O one doublet
esc
17
@
23
24
%
8
2
3
4
6.
8.
Q
E
R
Y
14
5
A
S
F
K
C
Σ
同
图
Expert Solution
Step 1
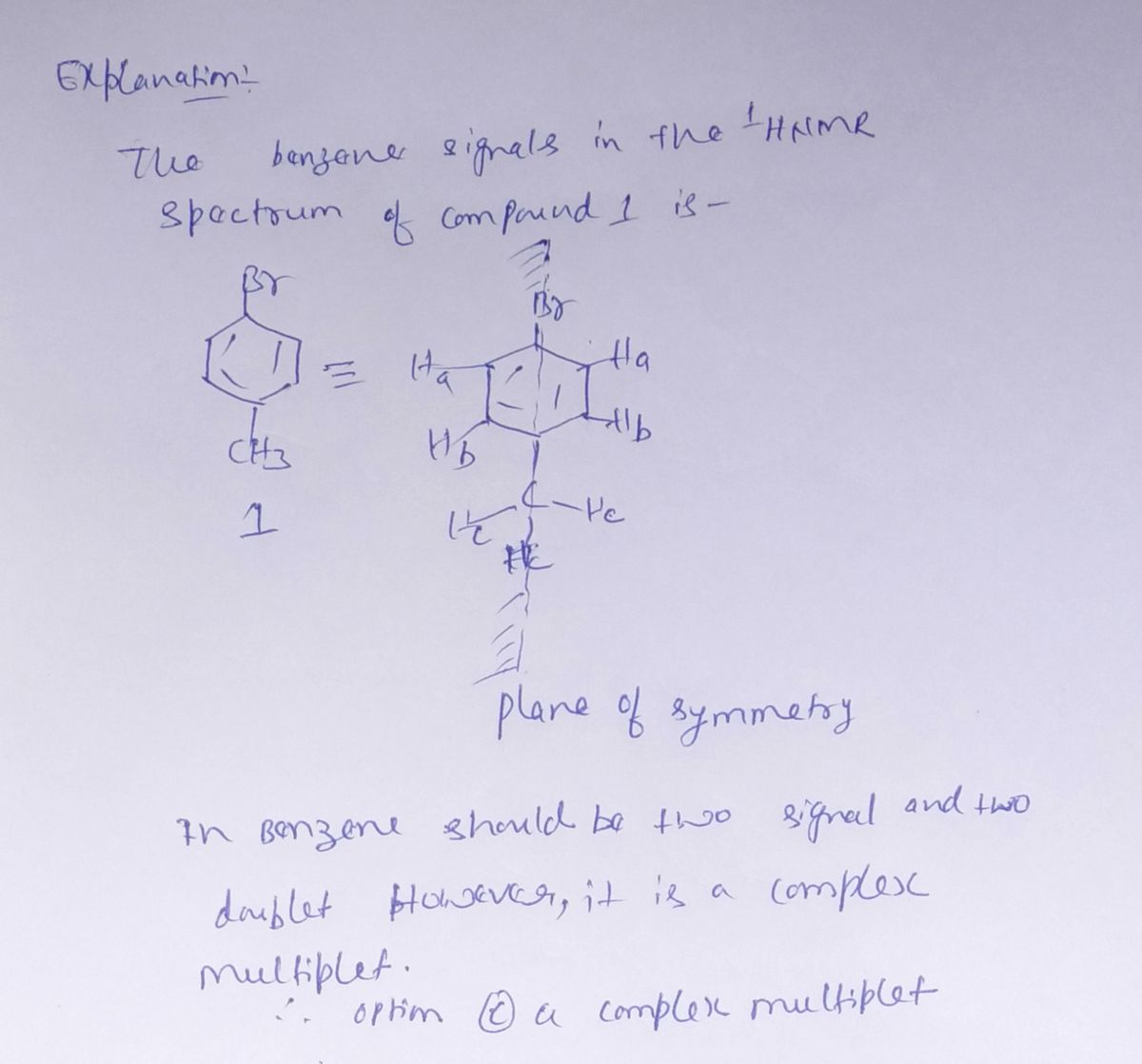
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

