Q: The Ka of propanoic acid (C₂H₂COOH) is 1.34 x 10-5. Calculate the pH of the solution and the…
A: We have to find out pH and equilibrium concentration of C2H5COOH.
Q: The decomposition of ethylene oxide (CH₂)2O(g) → CH4(g) + CO(g) is a first order reaction with a…
A:
Q: A sample consisting of 3.0 mol of perfect gas molecules with Cvm. 3R/2 which is initially at and 299…
A: A sample of 3.0 mol of a prefect gas is initially present at a temperature of 299 K and have a…
Q: The sun supplies energy at a rate of about 1.0 kilowatt per square meter of surface area (1 watt =…
A:
Q: Consider the following carbocation: L Which of the following carbocations would result from its…
A: Answer :
Q: 8. Indicate the relationship between each pair of molecules: enantiomers, diastereomers, or not…
A:
Q: For each pair of compounds listed, check the box next to the one with the higher boiling point.…
A: Given that - There are different groups of elements and compounds, which have different boiling…
Q: Part A A certain reaction has activation energy of 670 k3 mol and a frequency factor of A-180-10¹2…
A: The question is based on the concept of chemical kinetics. we need to calculate rate constant of the…
Q: (a) H₂O2 molecular mass conversion factor (b) C6H12 molecular mass conversion factor (c) SO₂Cl₂…
A: H2O2,C6H12, SO2Cl2
Q: What is the expected product of the following reaction sequence? O (A) A 00 O OH SOCI₂ py (B) NH₂…
A: The concept used in this question is taken from general Organic Chemistry. The correct answer of…
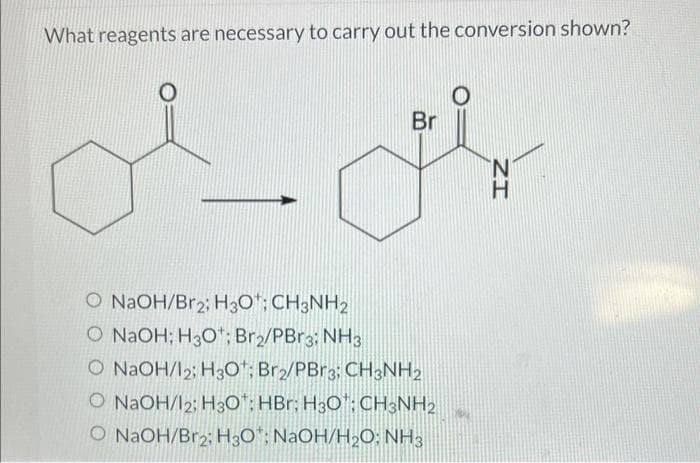
Q: Identify the reagent(s) you could use to accomplish the following transformation. Select all that…
A: This question belongs to basic organic reaction.
Q: Consider the rate law, (Look at picture) The balanced equation corresponding to this rate law is:…
A:
Q: What alkenes are formed when each alcohol is dehydrated with TSOH? Label the major product when a…
A:
Q: For each compound in the table below, decide whether there would be any hydrogen-bonding force…
A:
Q: IDENTIFY THE FF. BOND POLARITY AND d. SF e. SiF MOLECULAR GEOMETRY POLARITY OF MOLECULE
A: A bond formed between atoms with different electronegativity is polar
Q: The freezing point depression of 1% chloramphenicol is 0.06 deg Celsius. How many milligrams of…
A:
Q: What is the pH of a 0.660 M solution of NaCN (Ka of HCN is 4.9 × 10-¹⁰)?
A: Given [NaCN] = 0.660 M Kb = 4.9*10-10 NaCN ⇌ Na++ CN- pH = -log (H+) pOH = -log (HO-)
Q: Consider the titration of 100.0 mL of 0.200 M acetic acid ( K₂ = 1.8 x 10-5) by 0.100 M KOH.…
A: According Bartleby instructions if there is more than 3 subparts, I have to solve maximum 3…
Q: (c) What is the pKa of the acid? (only confused on part c, answers for a and b are given)
A: We have been given a weak acid which we are titrating with a strong base sodium hydroxide. we need…
Q: A 16.59 gram sample of chromium is heated in the presence of excess fluorine. A metal fluoride is…
A:
Q: What is the IUPAC name of the expected major product formed when 5-chloro-1-methylcyclohexene is…
A:
Q: state which ones are rotational Raman active and explain why (a) H2O (b) CO2 (c) NH3 (d) N2O…
A: Rotational Raman active molecules are those that have a polarizability that changes during rotation,…
Q: When 1 mole of N₂ (g) reacts with H₂(g) to form NH3 (g) according to the following equation, 92.2 kJ…
A: Answer : Given that, the reaction is N2(g) + H2(g) → NH3(g) Mole of N2 = 1 mol. Energy evolved =…
Q: Rank the samples of gas described table below der of increasing average speed of the atoms or…
A:
Q: In your own words, explain why water is known as the "universal solvent." Answer in complete…
A: The question is based on the concept of solubility. solubility follows a basic principle "like…
Q: What is the coordination number of the metal atom in the [Cd(EDTA)]- complex? X 5 +
A:
Q: What mass of natural gas (CH4) must you burn to emit 263 kJ of heat? Express the mass in grams to…
A:
Q: 9.39) The line bond structure of 4-bromo-3-hydroxypentanal is shown befow. The carbons are numbered…
A:
Q: Propose a structure for the HNMR data below and come up with a table that denotes the different…
A: The coupling constant J which is usually measured in hertz is a measure of the interaction between a…
Q: wires heat sample Water Stirrer nud Sample buming Steel sample outside chamber In an experiment, a…
A:
Q: 1. How many unpaired electrons are in the boron atom? This atom is A. Paramagnetic B. Diamagnetic 2.…
A:
Q: Suppose you measure out 0.0132 moles of ammonium sulfate on the balance. If the weigh boat…
A:
Q: Significance of your results: Titanium is also commonly anodized, but does not need to be dyed to…
A: First reaction : Ti2+ aq + 2 e- ↔ Ti s…
Q: iv. Circle the letter of any compound (shown below) that does not meet the structural requirements…
A: Aldol Condensation can be defined as an organic reaction in which enolate ion reacts with a carbonyl…
Q: Arrange the following ions in order of increasing ionic radius: oxide ion, fluoride ion, sodium ion,…
A: Given ions: Oxide ion, fluoride ion, sodium ion and aluminum ion.
Q: Equal volumes of the following pairs of solutions are mixed. Which pair will produce a buffer…
A: Buffer solution is the solution which resist the change of pH on addition of small amount of acid or…
Q: Which of the following solid salts is more soluble in 1.0 M H O a. CaF2 O b. CaCl2 O C. KNO3 O d.…
A: The question is based On the concept of solubility. we need to identify the salts which are more…
Q: Identify reagents that can be used to achieve the following transformation: The transformation above…
A:
Q: Write the complete electron configuration for the common monatomic ion formed by the element sulfur,…
A: Question is based on the concept of electronic configuration. we have to write the common electronic…
Q: 3. Calculate the volume of 5.9 x 1023 molecules of propane gas trapped in a container at a pressure…
A: Given, Molecules of propane gas = 5.9 x 1023 Temperature = 269 °F Pressure = 253.3 kPa
Q: The production capacity for acrylonitrile (C3H3N) in the United States is over 2 billion pounds per…
A: A limiting reagent is the species that is used up first in the chemical reaction. It decides the…
Q: At 25 °C, the equilibrium partial pressures for the reaction 2A(g)+B(g) ----C(g)+2D(g) were found to…
A: Since, Equilibrium constant or Kp is the ratio of the equilibrium partial pressure of product over…
Q: An iron–sulfur protein in Complex III donates an electron to cytochrome c1. The reduction…
A: FeS (ox) + e– → FeS (red) Ɛ°' = 0.280 V cyt c1 (Fe3+) + e– → cyt c1 (Fe2+) Ɛ°' = 0.215V
Q: How many joules of work must be done to inflate a balloon to a volume of 3.3 L if atmospheric…
A:
Q: Label the functional groups and identify the correct compound based on the IR spectrum. It is hard…
A: The question is based on the concept of IR spectroscopy. We need to identify the compound.
Q: Considering only ions with charges of +1, +2, -1 and -2, or neutral atoms, give the symbols for 4…
A: We know that the Isoelectronic species are those species which have same/equal number of electrons…
Q: ) a) Explain why pyrrole is aromatic. Draw an orbital energy diagram (arrows on lines) that…
A: We need to explain/answer (a) why pyrrole is aromatic and draw the labelled orbital diagram. (b)…
Q: + 2 NO₂ Ne CO₂ room tenp 50 °C OH NO NO
A: This is an example of crossed aldol condensation wherein at RT is is forming aldol while at 50 deg C…
Q: A sample of copper absorbs 55.2 kJ of heat, resulting in a temperature rise of 95.0°C, determine the…
A:
Q: How many moles are in 0.065g of NaOH. How many ions are in 0.75g of Na_(2)CO_(3).
A:
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

- Provide the reagents necessary to carry out the conversion shown. 1. H2O; 2. KMnO4 1. O3; 2. DMS; 3. Na2Cr2O7/H2SO4/H2O 1. NaOH, Br2; 2. DMS; 3. H2O/heat 1. O3; 2. DMS; 3. KMnO4 1. DMS; 2. H2O2/heatChoose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) Br2, FeBr3 Na/NH3, -33 degrees C NBS, light KMnO4, H3O+ Mg metal, ether KOH, EtOH, heatExplain how the reaction of (CH3)2CHCH(Cl)CH3 with H2O yields two substitutionproducts, (CH3)2CHCH(OH)CH3 and (CH3)2C(OH)CH2CH3
- reactions and products for C9H8O4 C9H8O4 + O2 --> C9H8O4 + H2O --> C9H8O4 + HCl --> C9H8O4 + OH- --> C9H8O4 + Na --> C9H8O4 + F -->Choose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) KMnO4, H3O+ CH3Cl, AlCl3 HNO3, H2SO4 Cl2, FeCl3 fuming sulfuric acidWhich of the following reagent best accomplish this transformation below? a. BH3, THF, H2O2 b. NaNH2, NH3 c. H2SO4, H2O, HgSO4 d. H2, Lindlar
- What steps are needed to prepare phenylacetylene, C6H5C = CH, from each compound: (a) C6H5CH2CHBr2; (b) C6H5CHBrCH3; (c) C6H5CH2CH2OH?Rank (1-4) from most reactive to least reactive (in decreasing reactivity in an elimination reaction with NaOH in DMF at 100 degrees celsius)Supply the missing reagent in the following reaction: (see attachment) A. Lindlar, H2, mCPBA B. O3 or KMnO4 C. fused KOH D. NH3 E. NaNH2
- The following questions ask you to choose which of the pair reactions shown has been correctly identified as faster. Choose all correct answers In the SN2 displacement by iodide of CH3Cl or of CH3OTs, CH3OTs reacts faster. In the SN2 displacement by acetate of bromoethane or bromocyclohexane, bromocyclohexane reacts faster. In the SN2 displacement by CH3CH2O- or by CN- on 2-bromopropane, CN- reacts faster. In the SN2 displacement by HC≡C- on bromomethane in benzene or in acetonitrile, reaction in acetonitrile goes faster.Draw the four resonance structures formed during bromination of methoxybenzene, CH3OC6H5, with Br2/FeBr3 to give para-bromomethoxybenzene and then circle the most stable resonance form.What reagent(s) is needed to complete the transformation below? Options: BH3, then NaOH/H2O2 HBr with peroxides (ROOR) Br2 in H2O Br2 in HOCH3

