Q: What concentration of potassium hydroxide is needed to give an aqueous solution with a pH of 11.270?…
A:
Q: A chemist measures the enthalpy change AH during the following reaction: P4(s) + 6 Cl₂(g) 4 PC13(g)…
A: The enthalpy change is a state function and the Hess law states that the change of enthalpy in a…
Q: Draw all products of the following reaction, then upload all drawings to the question at the end. I…
A: Given,The reaction:
Q: Determine if the mechanisms are SN1 or SN2. Then show the arrow pushing mechanism and predict the…
A: The given reaction is shown below.We have to predict the type of the reaction.Also, we have to give…
Q: Draw the structure(s) of the major organic product(s) of the following reaction. HO NaCN aqueous…
A: In the given reaction the reactant contains an ketone and alcohol functional group. The ketone gives…
Q: Air is a mixture of several gases. The 10 most abundant of these gases are listed here along with…
A: Answer:Mole fraction of a gas in a gaseous mixture will be equal to the ratio of number of moles of…
Q: Calculate the moles of Sodium Phosphate and Calcium Chloride in the six different trials.
A: In the given question we have to calculate the moles of sodium phosphate and calcium chloride.moles…
Q: 8) What is the product of this reaction sequence? Starting vii ii H 1: НО. Reagents OH /Acid…
A: This is multi step synthesis based problem
Q: When this reaction occurs H₂O + NH3 -> -OH + NH4+ Which of the following statements are correct?…
A: The given Bronsted-Lowry acid base reaction is 1.NH3 + H2O NH4+ + OH-Identify the Bronsted-Lowry…
Q: The compound shown below is the product of a Claisen condensation. Draw structural formulas for the…
A: The given product is shown below.We have to give the structural formula of the starting reactant of…
Q: Hydrolysis Time (min) 25.00 20.00 15.00 10.00 5.00 0.00 0 °C Experiment 1: Temperature 22°C 37°C…
A: The question is based on the concept of experimental chemistry.We need to interpret the bar chart…
Q: 8. A 24-hr urine specimen was diluted to exactly 2.000 L. After buffering to pH 10.0, a 50.00 mL…
A: The objective of the question is to calculate the amount of calcium and magnesium ions.The titration…
Q: Which type of product is formed from the reaction? Aldehyde [0]
A: The product formed when an aldehyde undergoes oxidation depends on the specific conditions and…
Q: 7. Galactosamine is another monosaccharide molecule, which plays crucial functions in some hormones.…
A: Given is structure of Galactosamine.It is monosaccharide.
Q: Draw the major product of this reaction. Ignore inorganic byproducts. O 1. Ag2O, NH4OH 2.…
A: Given is organic reaction.In which reactant is benzaldehyde.This is oxidation reaction.
Q: How many different products are formed when the following compound is treated with HNO, and H₂SO4?…
A: Given is organic reaction. This is nitration reaction. Nitration is a type of electrophilic aromatic…
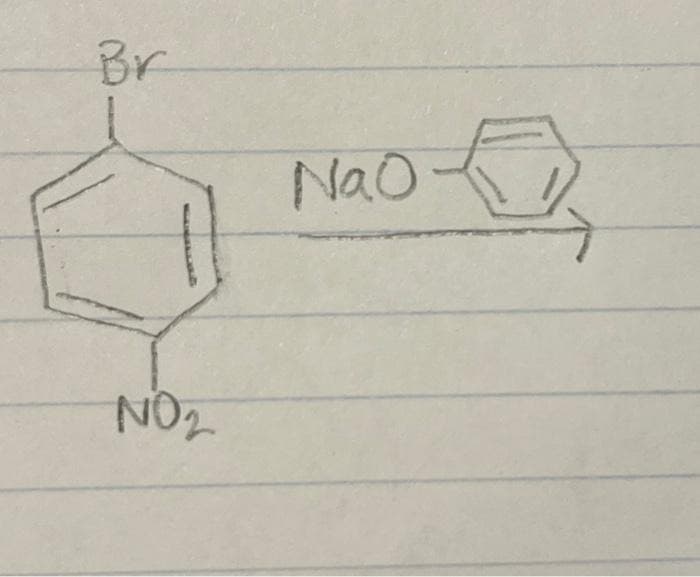
Q: Br -NO₂ Spell out the full name of the compound.
A: Answer:For systematic naming of the organic compounds IUPAC nomenclature system is used where IUPAC…
Q: Complete the mechanism of the following step by adding the missing curved arrow(s). Edit Drawing…
A: The question is based on the concept of organic synthesis. We need to synthesize the product using…
Q: Draw the product of this reaction. Ignore inorganic byproducts. KOH CHCI 3
A:
Q: if the bubble in the tip of the buret is not removed prior to performing the HCl titration will the…
A: A question is presented regarding the analysis of the effect of an air bubble in the burette tip on…
Q: Predict the Product. Predict the major organic product(s) or reactant(s) for the following…
A: To predict the major organic product(s) or reactant(s) for the following reactions:
Q: Draw curved arrows to show the movement of electrons in the step of the mechanism shown below.…
A: The Claisen condensation is an organic reaction that forms a carbon–carbon bond between two esters…
Q: 2. For each of the following molecules, draw all possible products of an allylic bromination. .d
A: For allylic bromination the N-bromo succinamide and carbon tetra chloride (CCl4) is used in the…
Q: Question 9 of 17 What mass of KOH would need to be dissolved in 500.0 mL of water to produce a…
A:
Q: How many kWh are required to produce 5.00 kg of Mg from the electrolysis of molten MgCl₂ if the…
A: We have to calculate the total amount of kWh that is required.
Q: Classify each of the molecules below. CH3 CH3-C-OH CH3 1° alcohol 20 alcohol 3° alcohol 4° alcohol…
A: Alcohol is the functional group -OH , which can be classified as 1° , 2° , 3° alcohols.
Q: (Incorrect) At a certain temperature the pH of a 0.030 M KOH aqueous solution is 12.39. Calculate…
A: Given,Molarity of KOH = 0.030 MpH of the solution = 12.39
Q: For the reaction 2 C6H5COOH(s) + 15 O₂(g) AUrxn = -772.7 kJ/mol at 298K. Calculate AHrxn. 14 CO₂(g)…
A: The objective of the question is to calculate the for the given chemical reaction.
Q: HBr a. Propose a reasonable mechanism, using curved arrow notation, for the formation of all…
A: We have to predict the major products at different temperatures.
Q: For each compound below, identify which position(s) is/are most likely to undergo an electrophilic…
A: In the given molecule two substituents are present out of that the methyl is electron donating or…
Q: Predict the MAJOR product for the following reaction. 1. B 2 AICH Brz FeBry CI ہ ہو تم کانه C
A: When the benzene reacts with an alkyl halide in the presence of a Lewis acid catalyst alkyl benzene…
Q: If 43.0 mL of 0.200 M HCI solution is needed to neutralize a solution of Ca(OH)2, how many grams of…
A: Given,Molarity of HCl = 0.200 MVolume of HCl = 43.0 mL
Q: Interconverting pH and hydronium ion concentration Each row of the table below describes an aqueous…
A: The pH of a solution can be calculated using the formula:pH = - log[H3]
Q: 3. Devise two different methods for creating the following alcohols. Start with any alkene. Your…
A: The question is based on organic reactions. We need to identify the product and explain its…
Q: Given the following values, calculate the mass of sodium oleate produced in the experiment. Please…
A: The provided calculation is part of a chemical analysis, and it involves determining the mass of…
Q: a. How many molecular orbitals will be formed by combination of the 28 and 2p atomic orbitals in…
A:
Q: The joining of two amino acids via a peptide bond (the process of protein synthesis) has a positive…
A: If the free energy change (∆G) for a reaction is negative then the reaction will be spontaneous and…
Q: Predict the major product of the following reaction. heat ?
A:
Q: Connections to biology 6. Idose is a monosaccharide (i.e. sugar) molecule. The structure shown on…
A: The conformational isomers are formed by the rotation of a carbon-carbon single bond in a compound.…
Q: Predict the substitution products of the following reaction and pay attention to stereochemistry.…
A: The question is based on organic reactions. We need to identify the product and explain its…
Q: Choose the situation below that would result in a AHsolution near 0 A B OC OD DE O When IAH solvent!…
A: Question is based on the concept of solutions. We need to identify the condition in which enthalpy…
Q: The reaction below is a base-catalyzed aldol reaction. O || CH₂-C-H CH3-C-H Ethanal (Acetaldehyde) H…
A: The aldol reaction is an organic reaction in which two carbonyl compounds reacts in presence of…
Q: 40 mL of a 35% v/v acetic acid solution is needed. In the stock room is a bottle of 50% v/v acetic…
A: The concentration of the stock acetic acid solution = The volume of the required acetic acid…
Q: OH Ph Pd" (Cat.) -Ph
A: Given:Pd( II) acts as a catalyst.Palladium( II) (Pd( II)) is a commonly used catalyst in various…
Q: Br Predict the ¹H NMR chemical shift of the circled proton(s) in the structure below. H₂ C. CH3 A)…
A: Given is organic compound. The name of given compound is 1-bromobutane.In 1HNMR number of peaks is…
Q: 1. Circle the structure or structures that represent the enantiomer of the following molecule 8.00m…
A: A pair of substances which are non-super imposable mirror images are called enantiomers. or two…
Q: Draw the tautomer of this aldehyde. Include all lone pairs. Ignore inorganic byproducts. H3O*…
A: Information of question
Q: How many moles of propylene (C3H) are in 25.0 g of the substance?
A:
Q: How many peaks would be observed for each of the circled protons in the compounds below? Н 11 O 1-7;…
A: Given are organic compounds. We calculate multiplicity or peaks by n+1 rule, where n is number of…
Q: Complete the table below. Round each of your entries to 2 significant digits. You may assume the…
A: An acid is a species which donates protons while a base accepts protons. An acid after donating…
Draw the product
Please answer fast I give you upvote
Step by step
Solved in 3 steps with 3 images


