Q: Zn(s) + 2HCl(aq) → ZnCl2(aq) + H2(g) What mass of Zn(s) reacts to produce 1.00 L of gas at 1.00 atm…
A: Zn(s) + 2HCl(aq) → ZnCl2(aq) + H2(g)Notice the 1:1 mole ratio that exists between zinc and hydrogen…
Q: Calculate the number of molecules in 11g of CO2
A: The number of molecules or atoms present in the one mole of the substance is equal to the Avogadro…
Q: Draw the structure of benzophenone.
A:
Q: 5. Draw the mechanism of the following reaction (students need to draw arrows, intermediate, and…
A: Alkene give addition reaction with halogens.The product formation depends on the medium of…
Q: Draw the structure(s) of the product(s) of the following Diels-Alder reaction. Stereochemistry must…
A: This is an example of a Diels-alder reaction in which a conjugated diene and a dienophile undergo…
Q: For the following reaction, 0.390 moles of iron are mixed with 0.267 moles of hydrochloric acid.…
A: Limiting reagent: HCl Amount of iron(II) chloride= 0.1335 molesExplanation:Reaction involves:- Iron…
Q: 1. Show that the partition function for a harmonic oscillator in the x direction only is given by:…
A: All parts are solved below.Please give helpful rating if it is useful.Explanation:
Q: (b) Cr₂S Element Oxidation Number Cri S (b) Cr₂S3 Element Cr C ง Oxidation Number i S i E SUPPOR
A:
Q: Acetic acid reacts with NaOH. At equilibrium the [CH3COOH] = 0.536 and the [CH3COO-] = 0.208 Ka =…
A: The objective of this question is to calculate the pH of a solution of acetic acid (CH3COOH) and…
Q: Which of the following stereoisomers will be formed in this reaction? Choose all that apply. H₂/Pd B
A: The addition reaction in which H2 is molecule is added at C=C bond to form alkane is called…
Q: Hydrazine, N₂H4, reacts with oxygen to form nitrogen gas and water. - N₂H(aq) + O2(g) → N2(g) +…
A: We will first calculate the mass of nitrogen produced by using the ideal gas eqation.Then we will…
Q: Draw the product of the following epoxide reaction, including the stereochemistry at any stereogenic…
A:
Q: Thermodynamics
A: For diamond heat capacity is unusually small as compared to elements.The graph is shown in images ,…
Q: Designate the Brønsted-Lowry acid and the Brønsted-Lowry base on the left side of each equation, and…
A: Brønsted–Lowry theoryAccording to Brønsted–Lowry theory, acid is a substance which donates an H+ ion…
Q: Consider the following compound. Part 1 of 2 H
A: The objective of the question is to predict the structure of diene for the following molecule given.
Q: In the illustration: The product that forms is 8 The illustration to the left represents mixture of…
A:
Q: Draw the major product of this reaction. Include stereochemistry if applicable. Ignore byproducts.…
A: The objective of the question is to find the major product of the reaction in which substituted…
Q: Draw a major resonance contributor of this enolate anion. Include all lone pairs in your structure.…
A: Final answer is given in explanation please see from there.Explanation:Approach to solving the…
Q: Only typed solution. Thank you!
A: Then the concentration of B at equilibrium :=3.24×10−3MExplanation:Step 1. The given chemical…
Q: A recent Chemistry & Engineering News issue, that was also sent out as a Canvas the editor…
A: In the given question, both compound A and compound B are different from each other.Both compounds…
Q: + OH H ...J Due the limitations of my graphic software: -the O atom does not show two lone pairs…
A: Both the given questions are about the substitution reaction ie; halide is substituted by ethanol or…
Q: For the following reaction, 10.6 grams of sodium are allowed to react with 5.70 grams of water…
A: Answer:Maximum amount of NaOH = 12.7 gLimiting reagent: H2O Amount of Excess reagent remain after…
Q: Consider the structure given below. Give correct detailed Solution with explanation needed, don't…
A: We can easily predict the number of bonding, non-bonding and antibonding orbital of cyclic systems…
Q: ? ? N ? OH NH2
A: Given the following transformation, propose a series of reactions to complete the synthesis.
Q: Consider the following reactants: Br Would elimination take place at a significant rate between…
A: The objective of this reaction is to draw the major product for the reaction given.
Q: 5. The hydrolysis of ethyl ethanoate is a reversible reaction. The equation for the equilibrium is…
A: f)i)According to the equation, every mole of CH3COOH produced yields one mole of C2H5OH, hence the…
Q: Draw the major product(s) of the following reaction. CH3 Give detailed mechanism Solution with…
A: This is the addition reaction of olefin,at first protonation occurs at the double bond and form a…
Q: QUESTION 5 What is the major organic product obtained from the following reaction? OH SOCI₂ pyridine…
A:
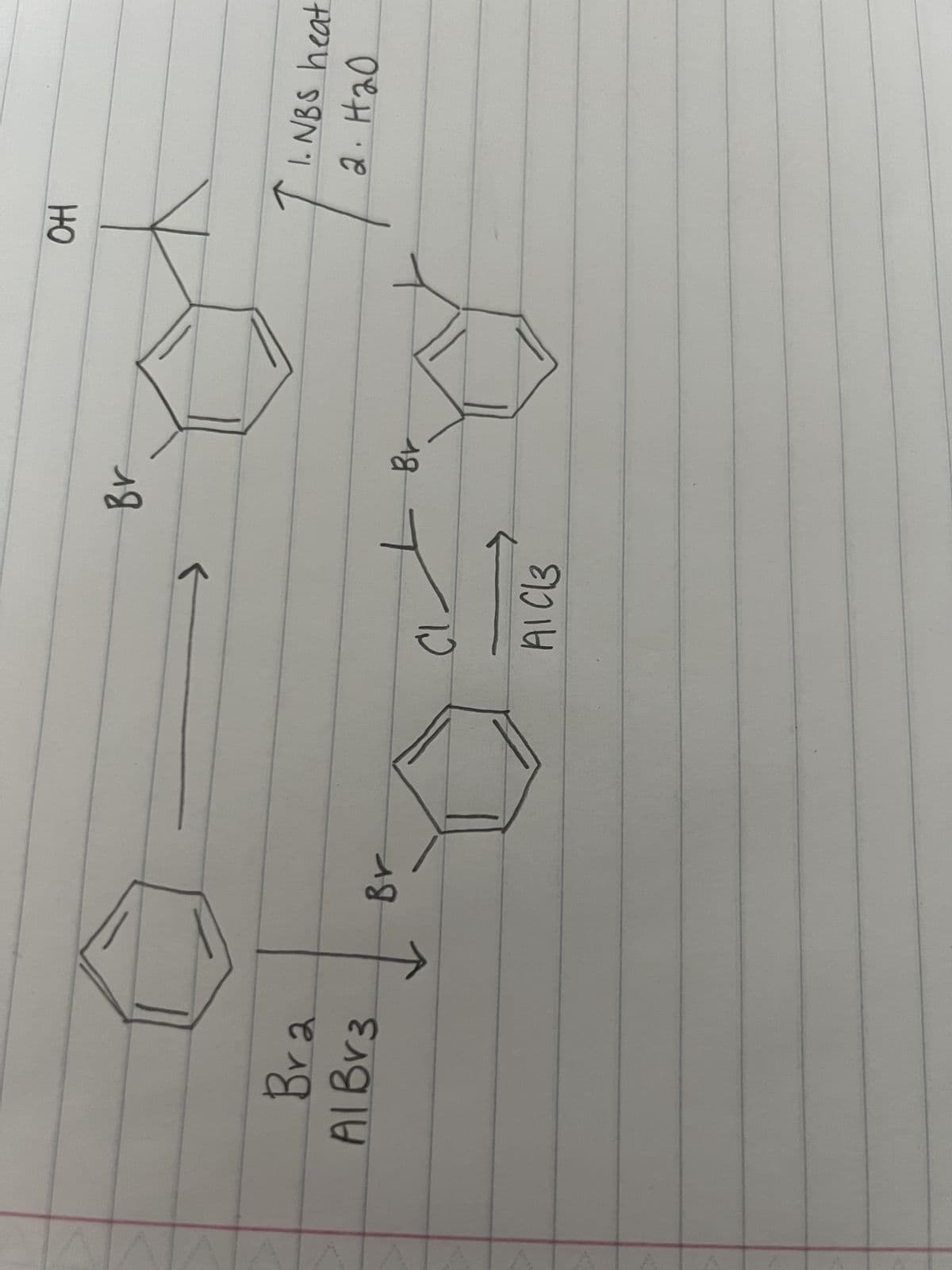
Q: For the reaction determine A) Why the reaction is performed with a distillation setup instead of a…
A: A) The reaction between t-butyl alcohol and hydrochloric acid (HCl) is typically performed with a…
Q: Give the IUPAC name for the following compound.
A: See solution in image Explanation:
Q: Complete this sentence: The mechanism of the reaction below is Хоты Nal 2 and the rate of this…
A: SN1 and SN2 are the uni- molecular and biomolecular substitution reactionsrespectively.In SN1…
Q: How many ozone molecules (O3) are in 1.00 m3 of air in the stratosphere where T = 250 K and the…
A: ozone molecules (O3) volume = 1.00 m3 T = 250 Kozone partial pressure is 1.0 × 10–3 atmcalculate…
Q: we use the algebra method to find the pka values of 3.954 and the graphical method to find the pKa…
A: Conclusion:The graphical method is preferred for its direct measurement of pH changes and reliable…
Q: Which is the MAJOR product of the following reaction? Br D Select one: D CH3OH, Na IV
A: In this reaction the base generated by the reaction between Na and MeOH is NaOMe which a strong…
Q: 4 Part A Which of the two acid solutions is more concentrated? Acid (b) is more concentrated. Acid…
A: The titration curve for two weak acids is given in the question.Both are titrated with 0.100 NaOH
Q: h 3aOH b. c. d. a. 2 a. 1. PnCH PPO 2 N ΠΕ a 1. PhCH, PPhy/ NaOH 2. H₂O+ b. NH₂ SOCI ملی NaCN NH…
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: differentiate between 2-hexanone and 3-hexanone on the basis of mass spectra
A: We have to differentiate between 2-hexanone and 3-hexanone using mass spectrometry.
Q: d. Why is the ẞ-epoxide not formed on mCPBA epoxidation although the ẞ-epoxide is predicted based on…
A: Although β-epoxide production is predicted based on OH-directed epoxidation, it is not present in…
Q: Add curved arrows to the reactant side of the SN2 reaction shown. Select Draw Rings More Erase H H-…
A: SN2 Mechanism:The reaction between methyl bromide (CH3Br) and hydroxide ion (OH-) to yield methanol…
Q: Which of the following is used as a component of the preservative formaldehyde? O & 80 H-C-H O…
A: The question is based on concept of organic chemistry.Formaldehyde is used as a preservative.we need…
Q: n the laboratory, a general chemistry student measured the pH of a 0.393 M aqueous solution of…
A: The acid dissociation constant Ka is the equilibrium constant, which indicates the acidic strength…
Q: [Review Topics] [References] Draw structural formulas for an aldehyde or ketone and alkyl (or aryl)…
A:
Q: The oxidation number of Fe in K3Fe(CN)6 is
A: We need to find oxidation number of iron(Fe) The compound given is Potassium Ferricyanide…
Q: ** Draw the major organic substitution product or products for (2R,3S)-2-bromo-3-methylpentane…
A: The bimolecular nucleophilic substitution reaction is a concerted reaction that occurs in a single…
Q: 1 How can one distinguish experimentally if the ortho ester shown below hydrolyzes in acidic water…
A: One ortho ester is given.
Q: Consider the following data on some weak acids and weak bases: base acid K₁ name formula name…
A: SolutionpH valuepH0.1 M KClO10.2640.1 M NH4Cl5.1320.1 M KBr7.0030.1 M…
Q: Solid magnesium sulfite is slowly added to 125 mL of a 0.482 M sodium hydroxide solution until the…
A: We will first calculate millimoles of both NaOH and magnesium ion. And after that we will find the…
Q: This amino acid is
A: Amino acids are organic compounds that contain both amino and carboxyl functional group. They are…
Q: For many purposes we can treat ammonia (NH3) as an ideal gas at temperatures above its boiling point…
A: Information about the question
Q: After running various experiments, you determine that the mechanism for the following reaction is…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Does this synthesis look right?
Step by step
Solved in 1 steps with 1 images

- ?⟶2? [A] (M) 1.33 × 10−2 2.66 × 10−2 3.99 × 10−2 Rate (L mol−1 h−1) 3.80 × 10−7 1.52 × 10−6 3.42 × 10−6Please don't provide handwritten soluion .......Nitrogen ΔHof (kJ/mol) ΔGof (kJ/mol) So (J/mol K) N2 (g) 0 0 191.6 N (g) 472.7 455.6 153.3 NH3 (g) -46.1 -16.5 192.5 NH3 (aq) -80.0 -27.0 111.0 NH4+ (aq) -132.0 -79.0 113.0 NO (g) 90.3 86.6 210.8 NOCl (g) 51.7 66.1 261.8 NO2 (g) 33.2 51.3 240.1 N2O (g) 82.1 104.2 219.9 N2O4 (g) 9.2 97.9 304.3 N2O4 (l) -20.0 97.0 209.0 N2O5 (s) -42.0 134.0 178.0 N2H4 (l) 50.6 149.3 121.2 N2H3CH3 (l) 54.0 180.0 166.0 HNO3 (aq) -207.4 -111.3 146.4 HNO3 (l) -174.1 -80.7 155.6 HNO3 (g) -135.1 -74.7 266.4 NH4ClO4 (s) -295.0 -89.0 186.0 NH4Cl (s) -314.4 -202.9 94.6 NH4Cl (aq) -299.7 -210.5 169.9 NH4NO3 (s) -365.6 -183.9 151.1 NH4NO3 (aq) -339.9 -190.6 259.8 Oxygen ΔHof (kJ/mol) ΔGof (kJ/mol) So (J/mol K) O2 (g) 0 0 205.1 O (g) 249.2 231.7 161.1 O3 (g)…
- Calcium ΔHof (kJ/mol) ΔGof (kJ/mol) So (J/mol K) Ca (s) 0 0 41.4 Ca (g) 178.2 144.3 158.9 Ca2+ (g) 1925.9 CaC2 (s) -59.8 -64.9 70.0 CaCO3 (s, calcite) -1206.9 -1128.8 92.9 CaCl2 (s) -795.8 -748.1 104.6 CaF2 (s) -1219.6 -1167.3 68.9 CaH2 (s) -186.2 -147.2 42.0 CaO (s) -635.1 -604.0 39.8 CaS (s) -482.4 -477.4 56.5 Ca(OH)2 (s) -986.1 -898.5 83.4 Ca(OH)2 (aq) -1002.8 -868.1 -74.5 Ca3(PO4)2 (s) -4126.0 -3890.0 241.0 CaSO4 (s) -1434.1 -1321.8 106.7 CaSiO3 (s) -1630.0 -1550.0 84.0 Carbon ΔHof (kJ/mol) ΔGof (kJ/mol) So (J/mol K) C (s, graphite) 0 0 5.7 C (s, diamond) 1.9 2.9 2.4 C (g) 716.7 671.3 158.1 CCl4 (l) -135.4 -65.2 216.4 CCl4 (g) -102.9 -60.6 309.9 CHCl3 (l) -134.5 -73.7 201.7 CHCl3 (g) -103.1 -70.3 295.7 CH4 (g) -74.8 -50.7 186.3 CH3OH (g)…Calculate the ΔG°rxn using the following information. Mg(s) + N2O(g) → MgO(s) + N2(g) ΔG°f (kJ/mol) 103.7 −569.32SO2 (g) + O2 (g) ------> 2SO3 (g) ΔS° = _____J/K
- How do you cancel the units to get Δ‡S J/K*mol? If A has units of dm3/mol*s and KbT/h leaves me a remaining unit of 1/sPlease don't provide handwritten soluitonHow can I round this number? Example: Cp = qmetal = -365.4 cal = 0.0501 cal/g°C m∆T (109.7g)(-66.5°C) help me with this Cp = qmetal = -77.0 cal = 0.9803921cal/g°C m∆T (109.7g)(-66.5°C)



