Briefly explain the picture of nitrogen cycle with this 4 major processes, - Nitrogen fixation - Decay (ammonification) - Nitrifiication - Denitrification
Briefly explain the picture of nitrogen cycle with this 4 major processes, - Nitrogen fixation - Decay (ammonification) - Nitrifiication - Denitrification
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter23: Potentiometry
Section: Chapter Questions
Problem 23.12QAP: What arc the advantages of microfabricated ISEs? Describe typical applications of this type of...
Related questions
Question
Briefly explain the picture of nitrogen cycle with this 4 major processes,
- Nitrogen fixation
- Decay (ammonification)
- Nitrifiication
- Denitrification
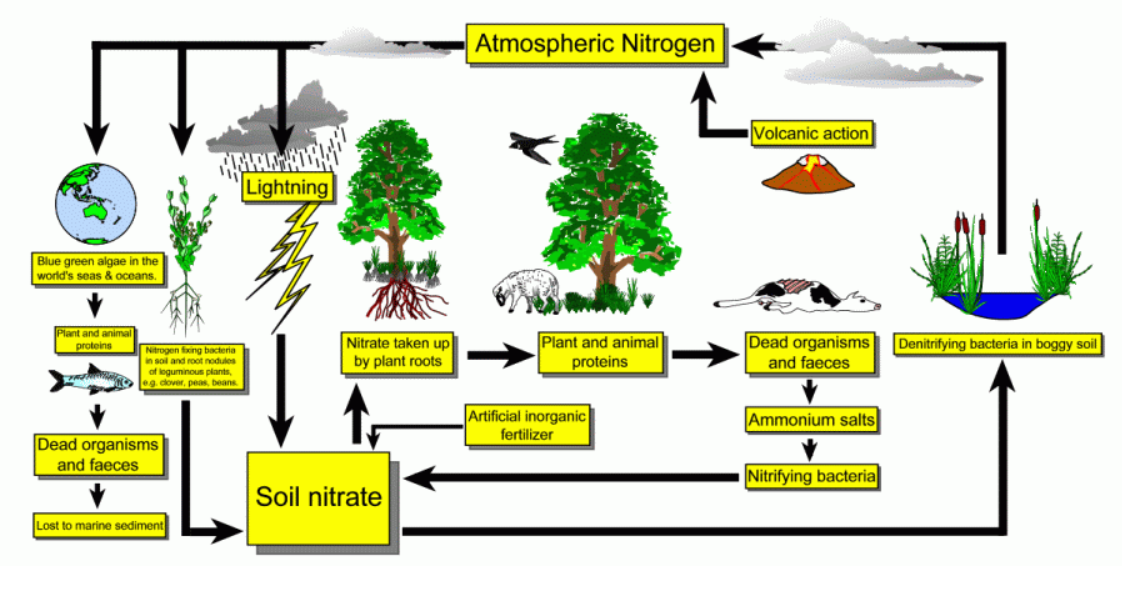
Transcribed Image Text:Atmospheric Nitrogen
Volcanic action
Lightning
Blue green algae in the
world's seas & oceans.
Plant and animal
Nitrate taken up
by plant roots
Plant and animal
proteins
Dead organisms
and faeces
proteins
Denitrifying bacteria in boggy soil
Nitrogen fing bacteria
in sol and root nodules
of leguminous pkants,
NO eg. dover, peas, beans.
Artificial inorganic
fertilizer
Ammonium salts
Dead organisms
and faeces
Nitrifying bacteria
Soil nitrate
Lost to marine sediment
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning