by region as Tigure belo which is the cleanest place to live in terms of air quality? 4000 Asia Pacific Africa Middle East Europe & Eurasia S. & Cent. America | North America 3500 - 3000 - 2500 2000 - 1500 1000 - S00 1988 1993 1998 2003 2008 2013
by region as Tigure belo which is the cleanest place to live in terms of air quality? 4000 Asia Pacific Africa Middle East Europe & Eurasia S. & Cent. America | North America 3500 - 3000 - 2500 2000 - 1500 1000 - S00 1988 1993 1998 2003 2008 2013
Chapter26: Biodiesel
Section: Chapter Questions
Problem 3Q
Related questions
Question
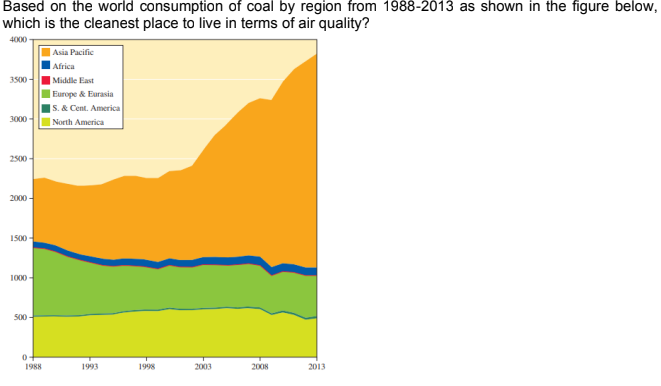
Transcribed Image Text:Based on the world consumption of coal by region from 1988-2013 as shown in the figure below,
which is the cleanest place to live in terms of air quality?
4000
| Asia Pacific
Africa
3500-
|Middle East
| Europe & Eurasia
|5. & Cent. America
| North America
3000 -
2500 -
2000 -
1500 -
1000 -
500 -
1988
1993
1998
2003
2008
2013
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning
