Q: Which of the following is the most likely pH for a solution of calcium hydroxide and water?
A: The pH scale runs from 0-14. Neutral pH is 7.0. The higher the pH (greater than 7) the more…
Q: Consider the following solutions at 25C. dentity the major species present in each solution and…
A: Answer:- This question is answered by using the simple concept of calculation of pH of aqueous…
Q: What is the pH c
A:
Q: b Answered Ata particular tempex Question 20 of 23 What is the pH of a 0.340 M solution of NaCN (Ka…
A: HCN in aqueous solution break as HCN= H++ CN- Now at equilibrium let concentration of H+= conc. of…
Q: The hypochlorite ion, ClO-, acts as a weak base. (a) Is ClOa stronger or weaker base than…
A: (a) Since, the basic character is generated by the donation of lone pair electrons of N in…
Q: BNOH TSOH b) H,N BNOCOCI HO, toluene reflux base OH
A: BnOCOCl is Benzyl chloroformate and its structure is- It is commonly used as a amine protecting…
Q: The final part asks for pH
A:
Q: Four solutions of an acid dissolved in water are sketched below, as if under a microscope so…
A: According to the Arrhenius theory, the substance that gives hydrogen ion H+ in the aqueous solution…
Q: Provide a brief definition of pH
A: Acid is the substance that gives H+ ions in the solution while the base accepts H+ ions or gives…
Q: pH=-log[H+] solve for H+
A:
Q: Reverse the concentrations of the weak acid and conjugate base for 0.25 M HC2H3O2 and 0.030 M…
A: Case 1: Concentration of weak acid and conjugate base are as follows: [CH3COOH] = 0.25 M [CH3COONa]…
Q: A 20-year-old student with coffee addiction had an episode of extreme hyperventilation due to…
A:
Q: carcutate the pH of 55X10-8M KOH two different wayB A nommat general Chemistry approach that onty…
A: KOH --> K+ + OH- Concentration of KOH = 5.5x 10-8 M KOH (aq) --> K+ (aq)+ OH- (aq)…
Q: The pH scale measures the mumber of H- 1ons in a solution If there are a lot of H- ions, is the pH…
A: PH is defined as logarithm of concentration of H+ ion.
Q: what is the value of the temperature (in K)
A:
Q: 0.00026 moles of acetic acid is added to 2.5L of water. What is the pH of the solution?
A: Acetic acid also called Ethanoic acid(CH3COOH )is a weak acid and on ionization it does not fully…
Q: Four solutions of an acid dissolved in water are sketched below, as if under a microscope so…
A: According to the Arrhenius theory, the substance that gives hydrogen ion H+ in the aqueous solution…
Q: s the lowest pH?
A: Lower pKa value results in stronger acid whereas higher pKa value results in weaker acid.
Q: HCI AICI3 + Н Al+
A: Interpretation: The given reaction is to be balanced.
Q: For the following recipes derive the proton condition equations (without using the Tableau method).…
A: To derive the proton condition equation for: a) System with the receipe (NaHCO3)T = CT
Q: A 20-year-old student with a coffee addiction had an episode of extreme hyperventilation due to…
A: The given information is: pH = 7.53 Conc. of bicarbonate ion = 22mmol/L The formula to calculate…
Q: of decreasing acid strength u
A: Acid is proton donor while base is proton accepter.
Q: The hypochlorite ion, ClO-, acts as a weak base. (a) Is ClOa stronger or weaker base than…
A: Given that ClO- act as a weak base
Q: = highest percentage ionization?
A:
Q: -[ ] PhLi HONH,, HCI A C ELOH B D
A: The solution of the question is given below:
Q: e pH of
A:
Q: Determine the pH c
A:
Q: How do I deteremine if an acid is monoprotic, diprotic, or tripotic?
A: Monoprotic acid : Monoprotic acids are acids that can release only one proton per molecule and have…
Q: N(aq), was determined to have a p its hase ionization constant
A: According to the question, solution containing 4.2 g/L of the weak base pyridine was determined to…
Q: Dindicates concentrat on che pH of a solution, ha
A:
Q: you nave an oxalic acia concentration oT 0-2 M, wnat is the pH of the system? IT you nave a sodiu…
A: Solution- Oxalic acid is (COOH)2 its means Oxalic acid of form H2A. Therefore, in the chart, we…
Q: optional. Complete the reaction between a Brønsted-Lowry acid and base. Phases are HСІ + NH,
A:
Q: the pH c
A:
Q: Which of the following is/are neutral? a. tap water b. Solution with pH = 6.5 c. Solution with pH…
A:
Q: The PH of a Oil M agueas sólutiom of Naz( the sodium salt of 2s 10,70: what is thie ha for Hz?
A: Given: Concentration of NaZ solution = 0.15 M pH of NaZ solution = 10.70
Q: pH of a C
A:
Q: pH
A:
Q: H;SO, но Ph Нeat Ph Ph A B C
A: Requirement from question: FT-IR data Reaction mechanism: Compound FT-IR data C=C…
Q: A. One simple way to measure the [H] is to measure pH. If a 10.0mL solution of "acid rain" is…
A: In mixture of strong acid and weak acid pH depends on H+ ion concentration of strong acid.
Q: O o.IMNCI pH.- log Co-1) )o O0I MHCL pH. - log Co.0or] PH. 14.2.873 a) oIM CH CODH 1-8K10-. o le…
A: pH is a scale used to specify the acidity or basicity of an aqueous solution. The range goes from 0…
Q: A 0.130 M solution of a weak base has a pH of 11.22. Determine KbKb for the base.
A: The concentration of hydroxide ion can be calculated from given p H.
Q: Q6) 200 ml , o.0ol M NAOH is udded to 30oml, o.0005 M HCL. what is the PH of the Soluution ? Ans. :…
A: The balanced Chemical Reaction between HCl and NaOH is - HCl + NaOH ----> NaCl + H2O Here, one…
Q: , what is the pK, for the base?
A: Given: The Ka of the conjugate acid is 8.59×10-4
Q: How do yu tell the differnce betwee strong and weak electrolytes, bases, and acids?
A: Strong and weak electrolyte:
Q: What is the value of K.
A: A + B = C + D (reaction at equilibrium) Kc = [product]/[reactant] Kc = [C].[D]/[A].[B] Where…
Q: A)write expressions for pH and pOH of
A: (NH4) 2SO4 it is salt of weak base and strong acid , hence it under go hydrolysis…
Q: presence of an acid, su
A: Organic reactions are those in which organic reactant react to form organic products.
Q: pH of
A:
Kindly answer question c & d
Step by step
Solved in 2 steps with 2 images

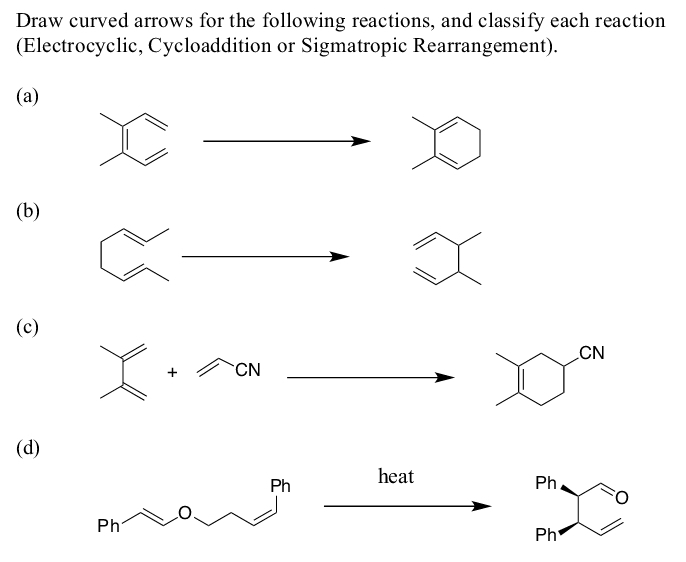
- Identify the pericyclic reactions in the followingreaction schemes. Give the complete reactionname and indicate the course of the reaction withthe aid of the arrow notation.a. Identify the mode of ring closure for the following electrocyclic reactions.b. Are the indicated hydrogens cis or trans?Identify the pericyclic reactions in the followingreaction schemes. Give the complete reaction name and indicate the course of the reaction with the aid of the arrow notation.
- When 1,2-dimethylcyclopentene undergoes hydroboration–oxidation, one diastereomerof the product predominates. Show why this addition is stereospecific, and predict thestereochemistry of the major product.Show the curved-arrow mechanism for the first step, and the structure of the cyclic intermediate formed, when cyclopentene in treated with KMnO4 . A Lewis structure for the permanganate ion is provided in the hint. Make sure to show all non‑bonding electron pairs and formal charges where necessary. Omit K+ .for the Cl2 addition reaction with 1,2dimethylcyclohexene, draw the most reasonable curved arrow mecanism. based on the reaction products, explain why a carbocation intermediate cannot be proposed
- The spirocyclic pentadiene derivative F shown below is converted stereospecificallyinto compound G on heating. The transformation involves two consecutive pericyclicreactions of the same type, and proceeds via compound H which is not isolated. Identify the type of pericyclic reaction occurring, and determine the structure ofcompound H.Suggest a detailed mechanism for the reaction below. Represent the product in the most stable conformation and also its stereochemistry (Identify stereogenic centers as R or S)1- Chloro-1,2-diphenyletane can undergo E2 elimination to give either cis- or trans-1,2-diphenylethylene (stilbene). Draw New man projections of the reactive conformations leading to both possible products, and suggest a reason why trans alkene is the major product?
- Classify each pericyclic reaction as an electrocyclic reaction,cycloaddition, or sigmatropic rearrangement. Indicate whether thestereochemistry is conrotatory, disrotatory, suprafacial, or antarafacial.Classify each pericyclic reaction as an electrocyclic reaction, cycloaddition, or sigmatropic rearrangement. Indicate whether the stereochemistry is conrotatory, disrotatory, suprafacial, or antarafacial.Suggest a detailed mechanism for the reaction below. Represent the product in the most stable conformation and also its stereochemistry.

