C) Kc= D) Kc = %3D [НзО*2 [H3O*j2 noitbson odt oi (8 CH K 180 90000 KE 3) The reaction below has a K, value of 3.3 x 10-5. What Svriw bas TD is the value of Kp for this reaction 3) 98.0 at 700 K? =Osau9d mundiliups 1e ai li (A 2 SO3(g) = 2 SO2(g) + O2(g) B) (OMg baogroa pecsree0-1 AS.O=0sauroed ainsibasn banwol O 8E.0 =0 9auood atorbong biswo ( a0 92 d abubong bwot ( 4) 4) IE Ko=20 x 1033 at 25°C, for the following reaction: H2(g) Cl2(g) = 2 HCl(g), then find Kp at the
C) Kc= D) Kc = %3D [НзО*2 [H3O*j2 noitbson odt oi (8 CH K 180 90000 KE 3) The reaction below has a K, value of 3.3 x 10-5. What Svriw bas TD is the value of Kp for this reaction 3) 98.0 at 700 K? =Osau9d mundiliups 1e ai li (A 2 SO3(g) = 2 SO2(g) + O2(g) B) (OMg baogroa pecsree0-1 AS.O=0sauroed ainsibasn banwol O 8E.0 =0 9auood atorbong biswo ( a0 92 d abubong bwot ( 4) 4) IE Ko=20 x 1033 at 25°C, for the following reaction: H2(g) Cl2(g) = 2 HCl(g), then find Kp at the
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter16: Solubility And Complex Ion Equilibria
Section: Chapter Questions
Problem 72E: A solution is prepared by adding 0.10 mole of Ni(NH3)6 Cl2 to 0.50 L of 3.0 M NH3. Calculate...
Related questions
Question
What is the value of kp?
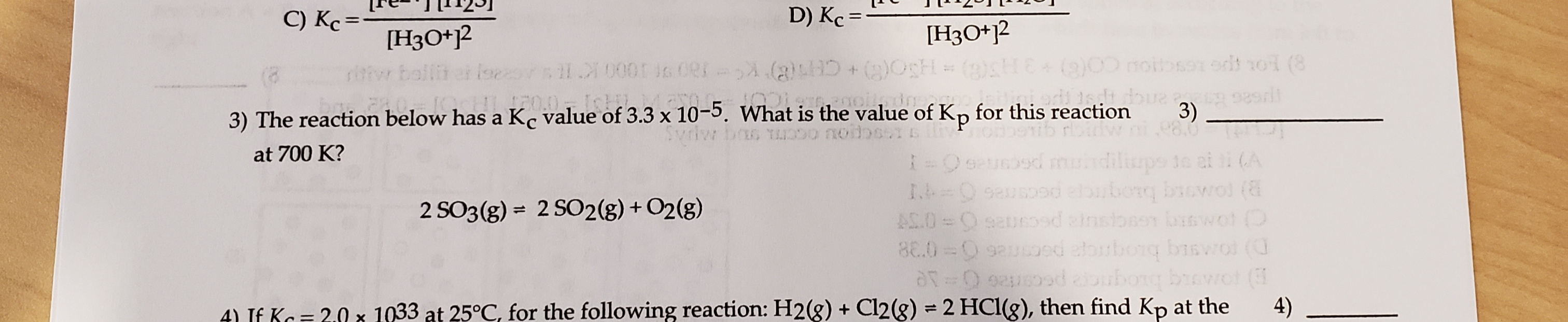
Transcribed Image Text:C) Kc=
D) Kc =
%3D
[НзО*2
[H3O*j2
noitbson odt oi (8
CH K 180 90000 KE
3) The reaction below has a K, value of 3.3 x 10-5. What
Svriw bas TD
is the value of Kp for this reaction
3)
98.0
at 700 K?
=Osau9d mundiliups 1e ai li (A
2 SO3(g) = 2 SO2(g) + O2(g)
B) (OMg baogroa pecsree0-1
AS.O=0sauroed ainsibasn banwol O
8E.0 =0 9auood atorbong biswo (
a0 92 d abubong bwot (
4)
4) IE Ko=20 x 1033 at 25°C, for the following reaction: H2(g) Cl2(g) = 2 HCl(g), then find Kp at the
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning