Caffeine has the following percent composition: carbon 49.48%, nydrogen 5.19%, oxygen 16.48% and nitrogen 28.85%. Its molecular weight is 194.19 g/mol. What is its empirical formula OND molecular formula
Caffeine has the following percent composition: carbon 49.48%, nydrogen 5.19%, oxygen 16.48% and nitrogen 28.85%. Its molecular weight is 194.19 g/mol. What is its empirical formula OND molecular formula
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter3: Stoichiometry
Section: Chapter Questions
Problem 3ALQ: True or false? The atom with the largest subscript in a formula is die atom with the largest percent...
Related questions
Question
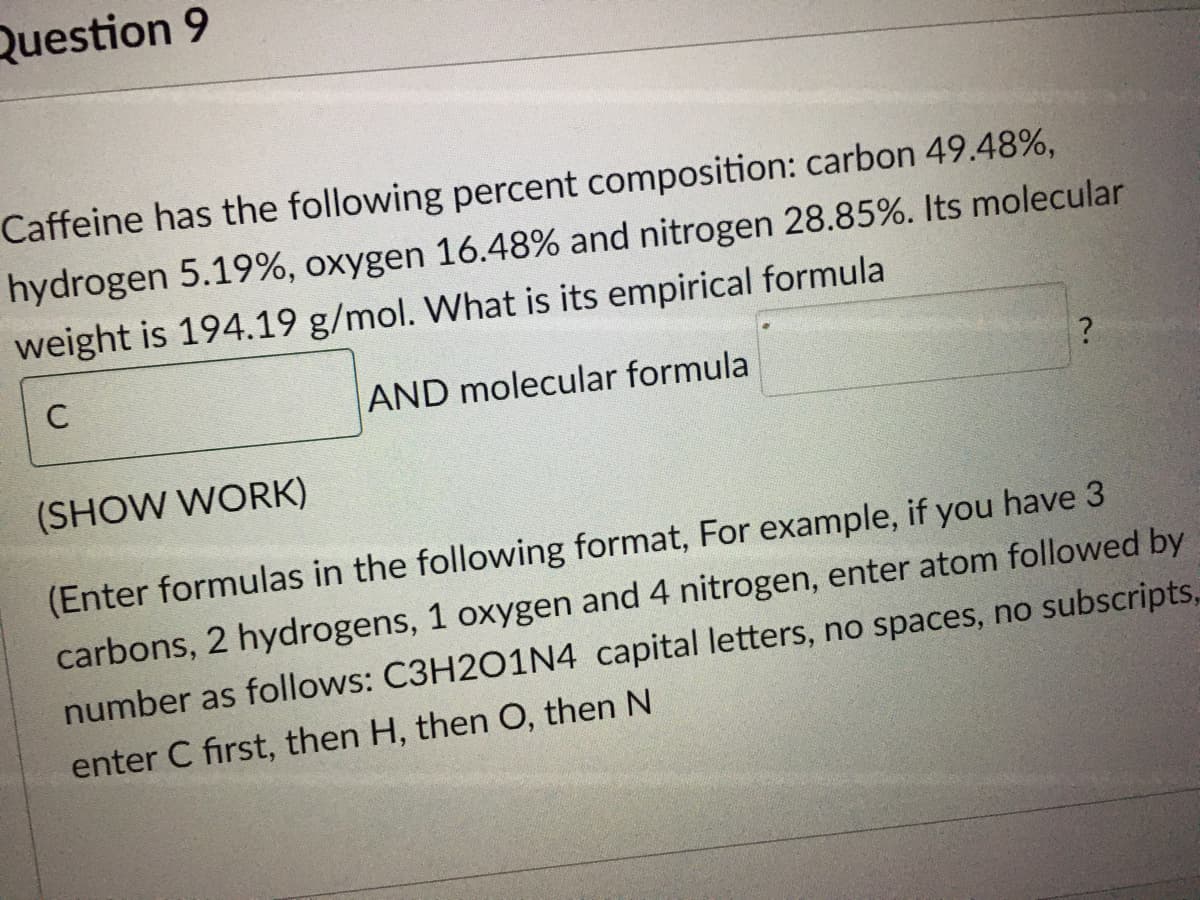
Transcribed Image Text:Question 9
Caffeine has the following percent composition: carbon 49.48%,
hydrogen 5.19%, oxygen 16.48% and nitrogen 28.85%. Its molecular
weight is 194.19 g/mol. What is its empirical formula
C
AND molecular formula
(SHOW WORK)
(Enter formulas in the following format, For example, if you have 3
carbons, 2 hydrogens, 1 oxygen and 4 nitrogen, enter atom followed by
number as follows: C3H2O1N4 capital letters, no spaces, no subscripts,
enter C first, then H, then O, then N
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.