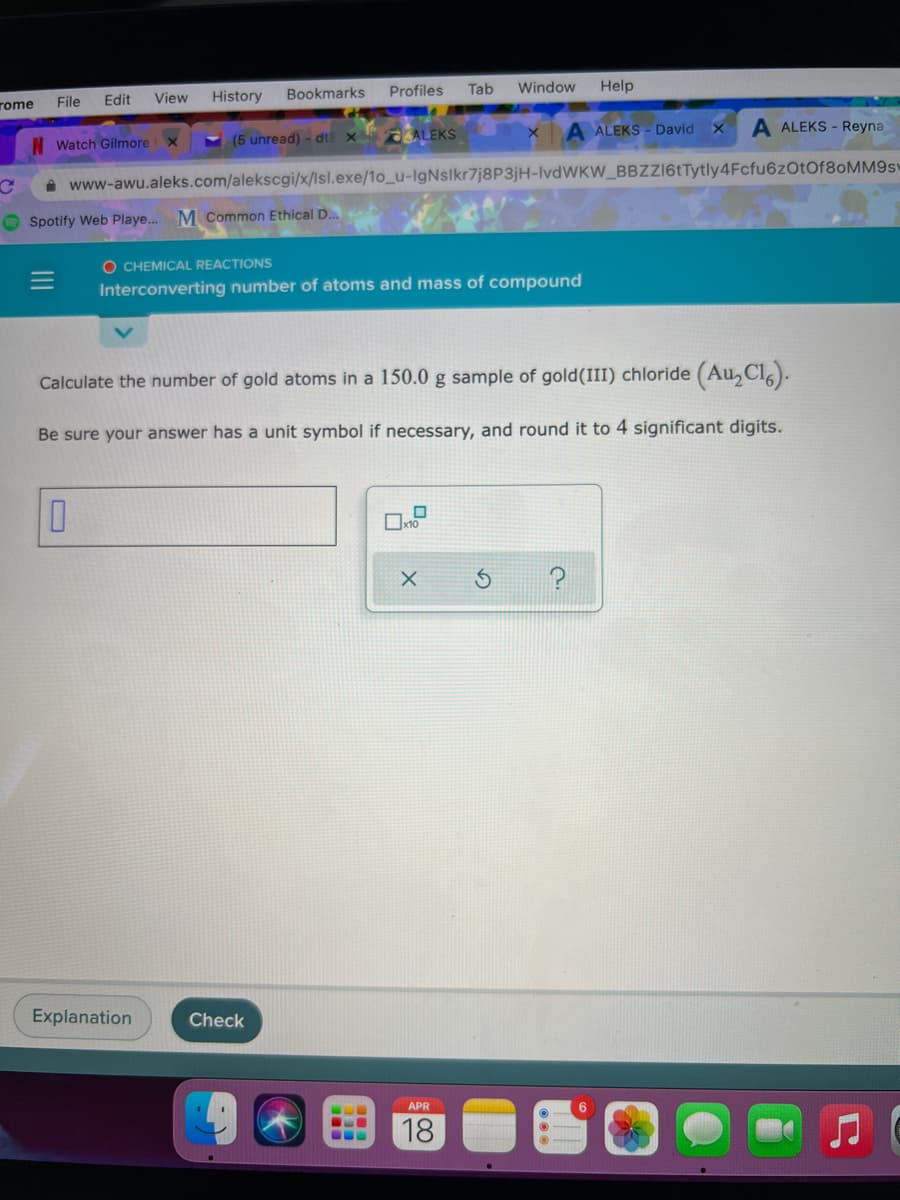
Calculate the number of gold atoms in a 150.0 g sample of gold(III) chloride (Au,Cl,). Be sure your answer has a unit symbol if necessary, and round it to 4 significant digits.
Calculate the number of gold atoms in a 150.0 g sample of gold(III) chloride (Au,Cl,). Be sure your answer has a unit symbol if necessary, and round it to 4 significant digits.
Chapter28: Atomic Spectroscopy
Section: Chapter Questions
Problem 28.4QAP
Related questions
Question
100%
Transcribed Image Text:rome
File
Edit
View
History
Bookmarks
Profiles
Tab
Window
Help
N Watch Gilmore x
(5 unread) - dtE X
AALEKS
A ALEKS - David
A ALEKS - Reyna
A www-awu.aleks.com/alekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-IVDWKW_BBZZI6tTytly4Fcfu6zOtOf8oMM9sv
O Spotify Web Playe..
M Common Ethical D..
O CHEMICAL REACTIONS
Interconverting number of atoms and mass of compound
Calculate the number of gold atoms in a 150.0 g sample of gold(III) chloride (Au,Cl).
Be sure your answer has a unit symbol if necessary, and round it to 4 significant digits.
Explanation
Check
APR
18
II
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

