Calculate the total binding energy in kJ per mole nuclide and in kJ/mol nucleons for the following nuclides, using the data given below. Total binding energy Nuclide kJ/mol nuclide kJ/mol nucleons (a) He (b) Mg (c) Ca 20 Particle or atom Mass (u) proton 1.00728 neutron 1.00866 electron 0.০0055 He 4.00260 23.98504 41.95862 1 u- 1.66054x10 27 ka
Calculate the total binding energy in kJ per mole nuclide and in kJ/mol nucleons for the following nuclides, using the data given below. Total binding energy Nuclide kJ/mol nuclide kJ/mol nucleons (a) He (b) Mg (c) Ca 20 Particle or atom Mass (u) proton 1.00728 neutron 1.00866 electron 0.০0055 He 4.00260 23.98504 41.95862 1 u- 1.66054x10 27 ka
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter18: Nuclear Chemistry
Section: Chapter Questions
Problem 27QRT
Related questions
Question
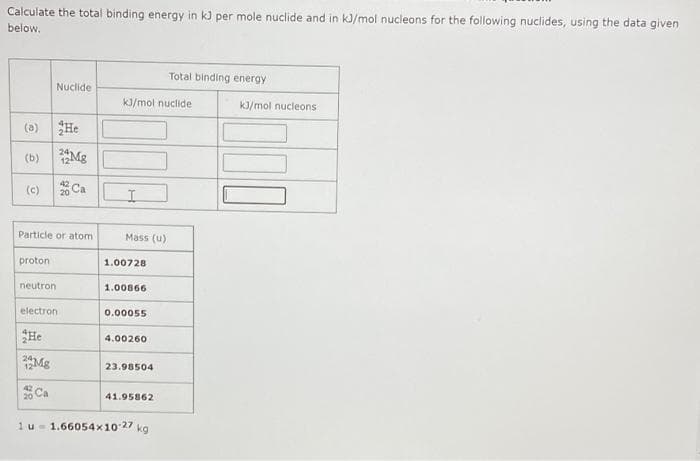
Transcribed Image Text:Calculate the total binding energy in kJ per mole nuclide and in kJ/mol nucleons for the following nuclides, using the data given
below.
Total binding energy
Nuclide
kJ/mol nuclide
kJ/mol nucleons
(a) He
(b)
Mg
(c)
Ca
Particle or atom
Mass (u)
proton
1.00728
neutron
1.00866
electron
0.০0055
He
4.00260
Mg
23.98504
20 Ca
41.95862
1u- 1.66054x10 27 kg
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning