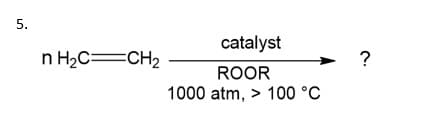
Q: Determine whether the following (forward) reactions favor high or low pressures? 1. 2SO2(g) + O2(g)…
A: Five questions based on Le-Chatlier principle, which are to be accomplished.
Q: 85. for the following reaction at 25°C and 1 atm: XeF,(g) → XeF,(s) + F,(g) AH¡ (kJ/mol) S° (J/K…
A: Given, And,
Q: reaction 1 reversed: 2CO2 + 3H20→C2H; OH + 302 Express your answer numerically in kilojoules per…
A:
Q: DBU faster reaction Br DBU slower reaction Br
A:
Q: Which of the following describes the effect of raising the temperature of an exothermic reaction…
A: According to the collision and activation energy theory of reaction rate, we know that the rate of a…
Q: What is the catalyst (if any) in the following? Group of answer choices NO2 NO3 NO There is no…
A: Fourth option. There is no catalyst.
Q: occur at the same rate. the reaction is no longer reversible.
A: Rate of chemical reaction is the change in the concentration in the given time interval.
Q: N(g) 0,(g) 2NO(g) AH = 180.5k) A Not enough information B The products will increase C No change D…
A:
Q: Q2. Explain the effect of catalyst in rate of the following reaction N2 + 3H, (Fe)catalyst 2NH3
A: Catalyst is a substance which can increase the rate of a reaction without itself getting consumed in…
Q: A reaction at -5.0 °C evolves 747. mmol of sulfur hexafluoride gas. Calculate the volume of sulfur…
A:
Q: Consider the following reaction: CH3COOH + H2O CH3COO- + H3O+ The position of equilibrium lies to…
A:
Q: Consider the following reaction ZAL (S) + 6HB. ZAIB13 (a9) + 3 Hz (g)
A: 2Al (s) + 6HBr(aq) -----> 2AlBr3(aq) + 3H2(g)
Q: Predict Majar produes H2 Lindlar's Catalyst Hz Pt O Na, NHz (D ー33°C
A: In the given above reactions, reduction is taking place. Gain of electrons by any species is called…
Q: H3C OH NaBr
A: Since you have posted multiple questions, we'll solve only the first question for you. If you want…
Q: Calculate log10 Keg for the reaction 5Br¯(aq) + 3B12(aq) + 6 0H (aq) Bro '3(aq) + ЗH20 Given :…
A: The log10Keq of the reaction is to be calculated 14.00 Normal 0…
Q: 6 Given H2SO4 reaction goes to completion and [SO4 ^2-]= 5 M, what is [H*]
A: ->H2SO4 is a strong acid which can completely dissociate into H+ and SO42-. Given-> [SO42-] =…
Q: At constant pressure for which of the reactions shown below should ΔH° be greater than ΔE° ? I. 2…
A: For a chemical reaction, the relation between change in enthalpy and change in internal energy is…
Q: Brazilians are quite familiar with fueling their automobiles with ethanol, a fermentation product…
A: Standard molar enthalpy of combustion of liquid ethanol is calculated as follows
Q: Most reactions are more rapid at low temperatures than at high temperatures. TRUE OR FALSE?
A: Rate of reaction is dependent on temperature.
Q: Arrange them separately in increasing order w.r.t SN¡ reaction: Br Br NH2
A: Nucleophilic Substitution reactions are those reactions in which one nucleophile replaces another.…
Q: In this reaction does equilibrium favor the products or reactants? C6H5COOH + NaOH → (C6H5COONa) +…
A: Acidity of a solution depends of pKa. If pKa is less then solution is more acidic amd if pKa has…
Q: reaction conditions. a) OH CH3 H3C ČI
A: Here we have to convert chlorobenzene to to 4-chloro-3,5-dimethylphenol by multistep reactions.
Q: The value of Keg for the following reaction is 0.16: A (g) + B (g) C (g) + D (g) The value of Keg at…
A:
Q: Calculate delta Go at 355 K for each of the reactions: a) CO+2H2=CH3OH b)N2+O2=2NO…
A: We know at 298 K ∆G° and ∆H° values are Compound ∆G°(KJ/mol) ∆H°(KJ/mol) CO -137.3 -110 H2 0…
Q: 1) Fill in the blanks below with appropriate reaction conditions or reaction products. H2, Pd HBr…
A: Some of the major classes of organic reaction are addition reaction, elimination reaction and…
Q: Но но 2 STEPS CO2M. OH Pro + CI CO2ME 80% yield Pro NH
A: The given reaction occurs in presence of base. Step 1: Production of amine from ammonium salt in…
Q: All chemical reactions are very slow. Select one: True False
A: All chemical reactions are very slow. False Molecular reactions may be slow or ionic reaction are…
Q: Organic Reaction Work on the reactions below complete with the reaction mechanism.
A:
Q: Ethanol (C2H,OH) can be made from the fermentation of crops and has been used as a fuel additive to…
A:
Q: 0. What is P2 If P1 = 200 mmHg, T1 = -73 celsius, T2 = 127 celsius. K = C + 273 The answer is 400…
A: Gay-Lussac’s Law: According to Gay-Lussac’s law, if we increase the temperature of a definite amount…
Q: CH) +2 Oze) CO2 +2 H,On 74 COe +2 H20 CH4) + 20z 2 C,HIo + 25 0z) → 16 CO + 18 H20 2 CH,OH +3 0z) →…
A: Endothermic reaction - Temperature of products lower than reactants. ∆H = positive. General…
Q: Which set of conditions will be required for this conversion? ? A H2, Lindlar's catalyst C 2eqNa,…
A: An unsaturated hydrocarbon (alkene or alkyne) does not react with hydrogen as such but in the…
Q: If a reaction has an equilibrium constant just greater than 1, what type of reaction is it?…
A:
Q: For the formation of C¿H;OH(1) AF2°C -41,770 cal. Find AF at 25°C %3D for the reaction 2 C(s) + 3…
A: Free energy of the reaction is given by ∆F =∆F° + RTlnQ Where Q = Reaction Quotient And Q =…
Q: Positive Negative Zero Indeterminate Converting octane to decane Heating liquid oxygen from 20 °C to…
A: Entropy change will be positive if a process involves increase in randomness. Randomness increase…
Q: For the following equilibrium reaction, explain the effect of adding more H2S to the syster 2 H2S(g)…
A:
Q: hat is the major Of the reaction 1) NaBH4, EtOН ОН 2) Н2О OH HO, HO
A: Interpretation: We have to tell what will be the product for the following :
Q: Br ? Step 1 Conditions? Step 2 Conditions?
A: There are many mechanisms in organic chemistry such as nucleophilic substitution, nucleophilic…
Q: Consider the following reversible reaction at equilibrium: C6H12O6(aq) + 6 O2(g) 6 CO2(g) + 6…
A: According to the le chatelier's principle, the reaction shifts in the endothermic direction when the…
Q: Using values from Appendix C, calculate the value of ▲H° for each of the following reactions: (d) 4…
A: Recall the reaction-- 4NH3(g) + O2(g) ---> 2N2H4(g) + 2H2O(l) ∆H°=?
Q: Et Et Me. LEt Et Me and Me H. H Me
A:
Q: H2C-O-Ĉ-R H2C-OH HO-CH ОН R-C-о-сн R,-CH2·OH R1-CH2-0 Catalyst 3 H2Ć-OH Нас-о-с-R
A:
Q: Keq of a reaction is 4 x 10Λ-7 at equilibrium:* the rate of the reverse reaction is much…
A: The Keq of a reaction is 4× 10-7 at equilibrium.
Q: 2 SO2 (g) + O2 (g)=2 SO3 (g) AH = 144 kJ Which of the below stresses will cause the total moles of…
A: Given that : The given equilibrium reaction is : 2SO2(g) + O2(g) ⇌ 2SO3(g) ∆H = 144 kJ…
Q: kindly answer all questions correctly with detailed explanation step by step for upvotes otherwise 5…
A: Given: Initial temperature = 25oC Final temperature = 720C Equilibrium moles of Br2 = 0.7 moles…
Q: 871 = (014 mulay CHE : not A CLA✓ + C-OH →→→ ? id 5 priesk In that is the Product Form POT 8 710…
A:
Q: At a particular temperature, Kc = 6.0 × 102 for 2NO(g) + 2H2(g) ⇌ N2(g) + 2H2O(g) Calculate Kc for…
A:
Q: CO₂Et ANO2 Pd catalyst Et N, heat
A: Note: According to our guidelines we are supposed to answer only first question. Kindly repost other…
Q: In the given reaction HO ConcH2S04, (P) The product (P) is
A: The given reaction is the dehydration of alcohol to form alkene as the product.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Anda how about the standart potencial? E = Eº - 0,0591/n log kpsDetermine DG0 for the reaction:2CO(g) + 2H2(g) ---> CO2(g) + CH4(g)DH0 = -247.3 kJ/mol DS0 = -256.5 J/mol KThe oxidation of glucose to CO2 and water is a major source of energyin aerobic organisms. It is a reaction favored mainly by a large negativeenthalpy change. C6H12O6(s) + 6O2(g) → 6CO2(g) + 6H2O(1) ΔH ° = -2816 kJ/mol ΔS ° = +181 J/mol • K(a) At 37 °C, what is the value for ΔG° ?(b) In the overall reaction of aerobic metabolism of glucose, 32 moles ofATP are produced from ADP for every mole of glucose oxidized. Calculatethe standard state free energy change for the overall reaction whenglucose oxidation is coupled to the formation of ATP at 37 °C.(c) What is the efficiency of the process in terms of the percentage of theavailable free energy change captured in ATP?
- The value of delta G at 281.0oC for the formation of phosphorous trichloride from its constituent elements, P2 (g) + 3CI2 (g) ---> 2PCI3 (g) is _____ kJ/mol. At 25.0oC for this reaction delta H is -720.5kJ/mol, delta G is -642.9 kJ/mol, and delta S is -263.7 J/K.During the industrial synthesis of methanol (CH3OH), carbon monoxide (CO) reactswith hydrogen gas. For methanol, ΔH= -100.4 kJ/mol. a) Is it preferable to maintain this system at high or low pressure? Explain.At 25°c ,the thermodynamic equilibrium constant for reaction 3C2H2(g) converts into C6H6(l) is K=1.00 ×1088 and ITS ∆Hrxn=-631 KJ/mol .. CALCULATE ENTROPY CHANGE ∆Srxn in J/mol.k??
- For the reaction C(s) + CO2 (g) -> 2CO (g) determine kp and kc at 1000K and at 1100kFor the reaction 2 C6H5COOH(s) + 15 O2(g) → 14 CO2(g) + 6 H2O(g), ΔrU⦵ = −772.7 kJ mol−1 at 298 K. Calculate ΔrH⦵The value of Delta G at 261.0oC for the formation of phosphorous trichloride from its constituent elements, P2 (g) +3Cl2 (g) -> 2PCI3 (g) is ___ kJ/mol. at 25.0oC for this reaction, delta H is -720.kJ/mol, Delta G is -642.9kJ/mol and delta s is -263.7 J/K
- Calculate the Delta Hrxn for the following reaction: C(s)+O2(g)--> CO2(g) DeltaHrxn=-393.5 kJ H2(g)+1/2O2(g)--> H2O (l) DeltaHrxn=-285.8 kJ C5H12(g)+8O2 (g)--> 5CO2(g) +6H2O(l) DeltaHrxn=-3536 kJ 10C(s)+12H2(g)-->2C5H12(g) DeltaHrxn=??????just to verify the answer rto 1 is increasing and 2 is increasing>??Calculate ΔG0reaction in kJ/mol at 298.15 K givenFe2O3(s)+3H2(g)→2Fe(s)+3H2O(l)ΔG^0reaction=ΣnΔG0Products−ΣnΔG0Reactants Substance ΔG0f (kJ/mol) ΔH0f(kJ/mol) S0298K (J/mol K) Fe2O3(s)Fe2O3(s) -742.2 -824.2 87.40 H2(g)H2(g) 0 0 130.7 Fe(s)Fe(s) 0 0 27.3 H2O(l)H2O(l) -237.1 -285.83 70.0 answer choices: a) 30.9 b)175.2 c)none of these d)158.7 e)-622.58



