Q: If the density of NaCl solution is 1.02 g/ml, calculate the molarity of NaCl solution (neglecting th...
A: Given, Density of NaCl solution = 1.02 g/mL Density of the water = 1.00 g/mL Molarity of NaCl sol...
Q: エ
A:
Q: 2.69 Label the Lewis acid and Lewis base in each reaction. Use curved arrows to show the movement of...
A:
Q: Explain why industrial manufacturers of hydrogen may choose to use reformation of natural gas to pro...
A: Hydrogen production from natural gas or other hydrocarbons is achieved by partial oxidation. A fuel-...
Q: The names and chemical formulae of some chemical compounds are written in the first two columns of t...
A: MgSO4: It is an ionic compound and the major species present in the water will be : Mg2+, SO42- and ...
Q: Consider the equilibrium system described by the chemical reaction below, which has a value of Kc eq...
A: The given reaction is, CaCrO4(s) ⇌ Ca2+(aq) + CrO42-(aq)
Q: What additive may lower the electrical resistance in a plastics?
A: Additives are the chemicals which are added to the polymers to modify its various properties. There ...
Q: 1.51 How are the molecules or ions in each pair related? Classify them as resonance structures, isom...
A: Isomers are the molecules having same molecular formula but different structural formula.
Q: Highlight the functional groups in the following organic compounds and identify them.
A:
Q: What is the structure of the major organic product that would result from acid-catalyzed dehydration...
A: Interpretation: We have to draw the eliminated product for the given alcohol.
Q: A chemistry student weighs out 0.135 g of acetic acid (HCH,CO,) into a 250. mL volumetric flask and ...
A:
Q: Rank the following nuclides in terms of increasing half-life: radon-222 (k = 2.1E-9 ms-1), radium-22...
A: Interpretation : We have arrange these radioactive species in the order of their half - life.
Q: How many liters of 0.305M K3PO, solution are necessary to completely react with 187 mL of 0.0184 | M...
A: Formula used M1V1 (K3PO4) = M2V2 (NiCl2)
Q: You are employed as a coop student at the Drug and Alcohol Testing Association of Canada (DATAC) dev...
A: 2-Methyltetrahydrofuran (MeTHF) is a solvent produced from renewable raw materials by the hydrogenat...
Q: General Chemistry II Lab Manual, 2019 Revision 81 Questions: 1) 5.0 moles of NH3 and 2.0 moles of H2...
A: To calculate the Kc for the below reaction. 3H2g+N2g⇌2NH3g
Q: Name the following ionic compounds: (a) Li2O (b) FeCl3 (c) NaClO
A: Note: As per our guidelines we are supposed to answer first 3 parts. Please repost other 3 parts as ...
Q: 6. What product(s) would result from the oxidation of each of the following alcohols with, for examp...
A: KMNO4 is a strong oxidizing agent When KMNO4 react with alcohol produce ketone. Tertiary alcohols ar...
Q: (II) Write all the possible products of the reaction of A with Cl2/hv.
A:
Q: What is the experimental yield of CRUDE PRODUCT AND FINAL PRODUCT (grams and percentage) Experiment...
A: One mole of 2-Chloro toluene oxidized with KMnO4 to give 2-Chloro benzoic acid. Molecular weight of...
Q: Which of the following protein assays does not make use of the copper (I or II) ions in the determin...
A: The purpose of the protein assay is to determine the concentration of a protein out of different pro...
Q: A mixture of nitrogen and argon gas is compressed from a volume of 15.0L to a volume of 11.0L, while...
A: From thermodynamics, work done is calculated as: w = -PΔV Or, w = -P(Vf-Vi) where Vf and Vi are ...
Q: The pressure of a fixed amount of gas _________ when its volume increases and its temperature increa...
A: According to Boyle's law : For a fixed amount of an ideal gas kept at a fixed temperature, pressure ...
Q: Hydrazine, N, H,, reacts with oxygen to form nitrogen gas and water. N,H, (aq) + O,(g) → N,(g) + 2 H...
A: Percent yield :- It is calculated by using the formula Percent yield = [actual yield / theoretical ...
Q: A chemistry student weighs out 0.0932 g of acrylic acid (HCH,CHCO,) into a 250. mL volumetric flask ...
A:
Q: How are the hydrogen atoms classified?
A: Hydrogen atoms attached to a particular carbon centre, Depending upon the number of Hydrogen atom at...
Q: Find the pressure A rectangular tan the gasoline is 14 on the bottom. A 130 Ib wom an
A: 1. Given data set: Height (h) = 80 cm We need to calculate the pressure using the formula: P = h*g*d...
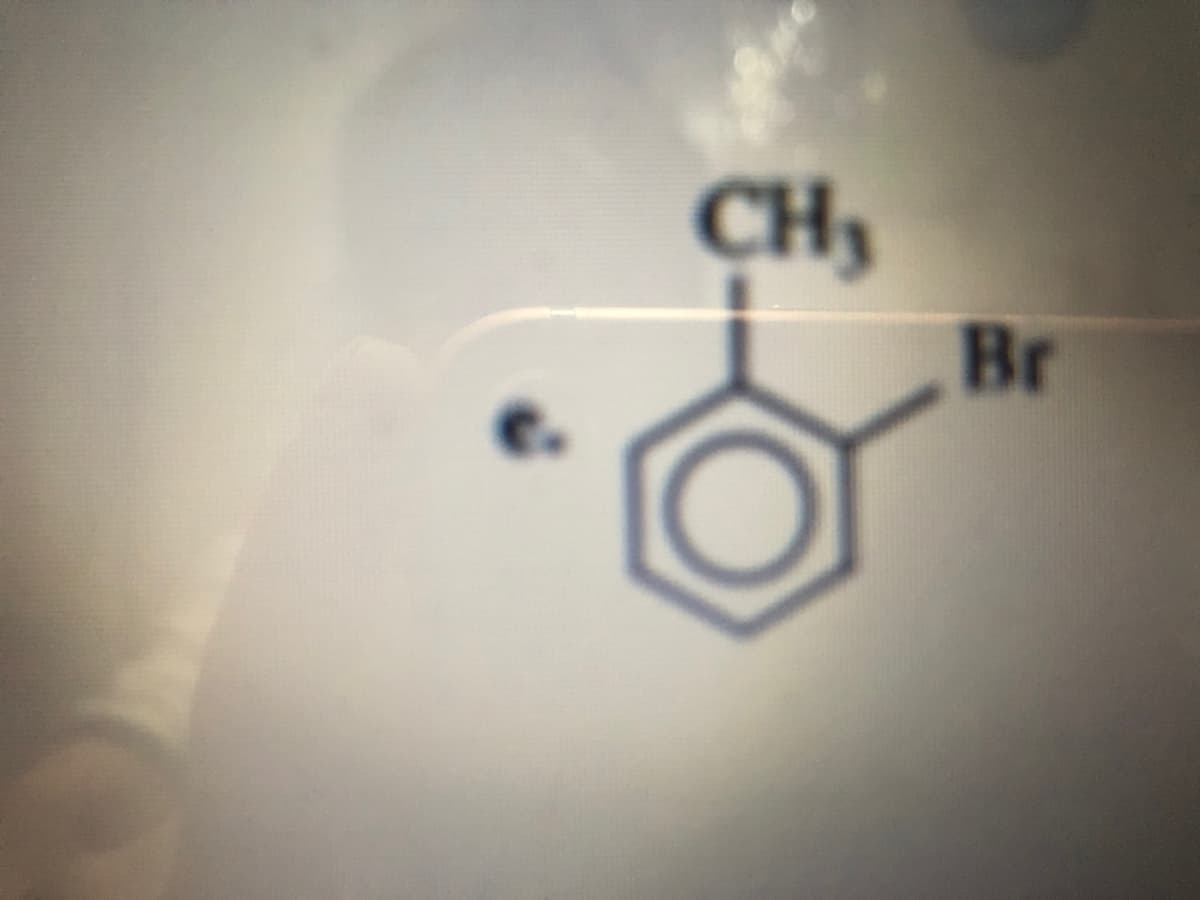
Q: Write the IUPAC name of the following molecule: *Lowercase letters only and DO NOT put space in betw...
A:
Q: common name & IUPAC
A: The IUPAC nomenclature is defined as the standardized name that is given to the organic compounds wh...
Q: ne wrong structure.
A: A permissible resonance structure has all the atoms with a complete octet.
Q: The ionization constant of nitrous acid is 4.50x10-4. Calculate the concentrations of H+(aq), NO2-(a...
A: Ionizaton constant is the ratio of concentration of product to reactant.Each concentration term is r...
Q: Explain how the concentration of the analyte is computed using the concentration of the analyte cons...
A: Titration is the method to determine the concentration of unknown substance.
Q: The combustion of octane, C,H18, proceeds according to the reaction shown. 2 C3H18(1) + 25 0,(g) – 1...
A: The balance chemical reaction : 2C8H18(L) + 25O2(g) ----> 16CO2(g) + 18H2O(L) Number of moles o...
Q: If the rate constant in a first-order degradation reaction of a drug product is 0.462/year, what is ...
A: Shelf life is the time required, for degradation of drug by 10%
Q: Write the IUPAC name of the following molecule: *Lowercase letters only and DO NOT put space in betw...
A: We are to write the IUPAC name for given structure.
Q: (a) Mn (b) Mw (c) PDI
A:
Q: number
A:
Q: A 1.85 g sample of an unknown gas at 47 °C and 1.00 atm is stored in a 2.85 L flask. What is the den...
A: Given: Mass, m =1.85 g Volume , V =2.85 L Pressure, P = 1.00 atm Temperature, T = 47 +273 = 320 K D...
Q: oxygen!) b)Using the data from the experiment, find the percent of nitrogen by mass in the oxide. c)...
A:
Q: Write the chemical formulas for the following compounds:
A: A chemical formula is defined as an expression which shows the elements present in a compound and al...
Q: 100. mL sample of 0.200M aq HCl acid is added to 100mLof 0.200M aq ammonia in a calorimeter.whose he...
A: Given that, Volume of 0.2 M HCl solution = 200 mL = 0.2 L Moles of HCl in the solution nHCl = 0.04 m...
Q: pecies is a valid resonance structure of A? Use curved arrows to show how A is converted to any . Wh...
A:
Q: Consider a neutralization reaction in which NaOH(aq) is mixed with H2SO4(aq) in a coffee cup calorim...
A: Given is coffee cup calorimeter, Mass m = 159.0 g ΔT = 7.70 °C We need to find heat of neutraliza...
Q: Clecause is R K Stereochemistry R & S ome tlechee 'S Br a) NH
A:
Q: What is the mass of the following? a. 25g iodine; density=4.93g/cm³ b. 1000 mL octane; density=0.089...
A:
Q: Unknown compound D,whose formula is C28H44O, is an alcohol that produces a compound whose formula is...
A: We will use the formula of unsaturation to complete this question .
Q: why is lupac for Hg2Cl2 Mercury (I) Chloride, instead of Mercury (II) Chloride?
A: Given, Molecules is Hg2Cl2 Why is IUPAC for Hg2Cl2 Mercury (I) Chloride, instead of Mercury (II) C...
Q: Which of the following conformers will obtain the highest strain energy? A В C
A:
Q: Are cis-2-hexene and trans-2-pentene stereoisomers?
A: Answer : cis-2-hexene and trans-2-pentene are not Stereoisomer. * Explanation : Isomers are the mol...
Q: natural gas
A:
Q: How would you separate your chocolate chip cookies? Which separation method would you use?
A: Separation method is a method in which two or more than two substances are separated with the help o...
Step by step
Solved in 2 steps with 1 images




