Q: after studying the chemical reactions of many compounds made of carbon, August discovers a ______…
A: Carbon can made different compounds i.e. it made carbon-carbon single bond, carbon-carbon double…
Q: 5. Isoelectronic ions are the species that have the same number of electrons. Part a. What +2 ion is…
A: Isoelectronic species: Isoelectronic species can be define as the atoms or ions having the same…
Q: back carbon to provide the Newman projection in the most stable conformation. H3C HII!!!! Н (H3C) 3C…
A: This question belongs to stereochemistry.
Q: In the laboratory, a general chemistry student measured the pH of a 0.392 M aqueous solution of…
A:
Q: Consider the following balanced chemical equation: H₂O2 (aq) + 31 (aq) + 2 H+ (aq) → 13¯ (aq) + 2…
A:
Q: A chemist mixes 81.4 g of benzene with 49.9 g of heptane and 10.0 g of 2-ethyltoluene. Calculate the…
A: Mass percent is the percent ratio of mass of element to the total mass of the compound.
Q: What is the value for x in the equation below? e(1.24x) = 412.8(9.07+9.21)
A:
Q: A common anesthesia gas is Halothane, with the formula C2HClBrF3 and the structure is shown in the…
A: ideal gas law, PV = nRT where P = pressure, V = volume, n = mole of gas, R = Universal gas constant…
Q: See Figure 2-2. Match each atom to its hybridization. C-1 is : C-2 is: C-3 is : Nis O is
A:
Q: 4. Which glassware in the lab can be used to precisely measure volume of a liquid? Explain
A: Liquid takes the volume of the container in which it is kept.
Q: After 72.0 min, 34.0% of a compound has decomposed. What is the half-life of this reaction assuming…
A: Given -> Time(t) = 72.0 min -> 34.0% compound decomposed .
Q: how do you figure out what to expect IR to show about these two molecules?
A: Main application of IR spectroscopy is to identification of functional group present in the…
Q: 4. Identify the isomers below as structural, cis-trans, or enantiomers. 5.Is this molecule a…
A:
Q: 2-bromo-2-ethoxypropane ii) You did a reaction where you wren't sure whether you had a pure sample…
A: We have find out the answer.
Q: Question 4 A gas has a pressure of 431 torr when the temperature is 14 °C. The temperature changes…
A: Given, Initially, Pressure (P1) = 431 torr Temperature (T1) = 14°C = (14 + 273.15)K = 287.15 K…
Q: 34) The product formed by the reaction of toluene with chlorine in the presence of sunlight is: (a)…
A: 34) Given, The product formed by the reaction of toluene with chlorine in the presence of sunlight…
Q: ctrolyte, write out an ionic equation as NaCl(s)→ Na* (aq) + Cl(aq) f. CaCl₂ (soluble salt) strong…
A:
Q: 3. For each of the following compounds, categorize it as a strong, weak, or non-electrolyte. Give…
A: The substances which dissociate into ions (cations and anions) when they dissolve in a polar solvent…
Q: Arrhenius Equation A(g) + B(g) → C(g) The rate law for the above reaction is: -d[A]/dt k[A][B] The…
A: In this question we have to calculate the activation en erg(Ea), pre exponential factor (A) and the…
Q: Write an IUPAC name for the following alkane/cycloalkane:
A: According to IUPAC Nomenclature, Name of alicyclic compounds are obtained by adding prefix cyclo to…
Q: C. Br F b. 사
A: We will answer using IUPAC nomenclature of organic molecules.
Q: You measure the following dimensions of a rectangular metal block of metal A: length =11.0 cm; width…
A:
Q: Small amounts of oxygen gas can be produced in the laboratory by heating potassium chlorate, KClO3.…
A:
Q: 2- Give the common names for the following Alkenes: Ethene, Propene, 2-Methyl propene, and 2-Methyl,…
A: Alkene contains carbon-carbon double bonds. The common name of an alkene can be given on the basis…
Q: A 5.0 g sample of NaCl is dissolved in enough water to give 500 ml of solution. Wat is the Na+ ion…
A:
Q: order to compare them to one another.) • Fluid A: m = 2.060 kg, V = 2000 mL • Fluid B: m = 6.729 x…
A: The fluids will be layred according to the density. The fluid of the highest density will layred on…
Q: Information: A student performs an experiment with three unknown liquids. The following measurements…
A: In the previous problem the densities of each fluid were calculated as follows: density of fluid A=…
Q: When 17.6 g of a metal is placed in a graduated cylinder container 10.0 ml of water, the water level…
A: Given, mass of metal = 17.6 g Initially water level mark = 10.0 mL Final water level mark = 12.2 mL…
Q: Green plants use light from the Sun to drive photosynthesis. Photosynthesis is a chemical reaction…
A: Balanced chemical equation for the reaction of water and carbon dioxide to produce glucose and…
Q: I don't understand why there's a carbon in the middle of NH2
A: To answer: why there's a carbon in the middle of NH2
Q: Identify the reagent(s) you could use to accomplish the following transformation. Select all that…
A: The given reactions are examples of substitution reaction. Substitution reactions are those in which…
Q: Draw several molecules of methanamide and show the intermolecular hydrogen bonding that account for…
A: In this question we have to draw the structure of methanamide which shows H bonding between…
Q: Fill in the Blank; Amino acids with ionisable side chains have 1) high 2) low 3) none 4) two 5)…
A: We have been given that amino acid have ionisable side chain.We have been asked to fill the blank in…
Q: UTE Problem 15 of 17 Solve the equation for m. 2m² - 3m 4= 0 m = M 6 ± √b² 4ac 2a CONTINUE
A:
Q: A chemist adds 480.0 mL of a 0.00368 mmol/L copper(II) fluoride (CuF₂) solution to a reaction flask.…
A: We know- Molarity=Number of molesVolume of solution in Land Number of moles=Mass of soluteMolar mass…
Q: 4. If an object has a density of 4.05 g/cm³, what is its density in units of kg/m³? a. 4.05 x 10¹…
A:
Q: 36) The major product formed in the following reaction is: NOH H*
A: The above reaction is an example of Beckmann rearrangement; the mechanism of reaction is as follows
Q: Using the following information, compute density (= mass/volume) to the correct number of…
A: Given mass = 1.375 ± 0.007 g volume = 1.12 ± 0.10 mL
Q: Calculate the percent ionization of a 0.441 M solution of hydrocyanic acid. (Assume that K₁ (HCN) =…
A:
Q: What type(s) of bonding would be expected for each of the following materials: (Write on your…
A: Types of bonds are a)Ionic bonds b) Covalent bonds c) Metallic bonds d) van der Waals forces of…
Q: If you mix 1.15 g Na₂C₂O (FM 134.00) with 0.60 g KMnO4 (FM 158.03) and excess aqueous acid, which…
A: The limiting reactant is the one that is completely consumed before the other reactant is consumed.…
Q: Activity 2.2. Using noble gas cores, write the shorthand electron configuration of the following: a.…
A: Using noble gas cores, write the shorthand electron configuration of the following: a. Zn b. Ba c.…
Q: Calculate the amount of energy necessary to transform 9.75 mL of water from 21.0 degrees Celcius to…
A:
Q: 4. Write the (ground state) electronic configuration for each of the following. a. antimony b. lead…
A: we have to determine the electron configuration of the given atoms and ions
Q: A solution of DNA has a concentration of 10 nanograms/ml. The experiment calls for 8.2 microliters…
A: Given concentration of DNA solution is 10 nanograms/mL.
Q: 3. Consider the following pairs of compounds. (CLA) a. Br₂ and 12- Pick the one with the higher…
A: We are given a pair of compounds in the question. We need to identify the one with higher vapour…
Q: Which statements about vapor pressure below are true? Select all that apply. A. Water in a 150 mL…
A: Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour…
Q: Sort the following scenarios based on whether they result in positive or negative heat (+heat or…
A: Answer: Whenever system ( be it object or chemical reaction) loses the heat then heat change will be…
Q: Which structure below is NOT a resonance structure of the compound shown in the box? O ( B 00 A All…
A:
Q: Structure 4: Draw the structure with a postively charged oxygen. H₂C X CH, H₂C CH3
A:
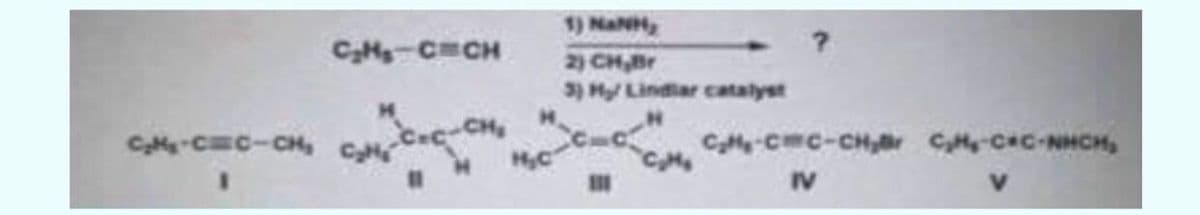
Step by step
Solved in 3 steps with 2 images

- -Name the correct mechanism via arrow pushing -name each mechanism -name each minor and major products (1S,2R)-1-iodo-2-methylcyclohexane + sodium cyanide/ DMF-->Please complete reactions in clear handwritten of all subpartsOrganotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?
- 2A---> 2B + C (Kp1) 2D---> 2E + C (Kp2) Now find the corresponding Kp3= f(Kp1, Kp2) = ?for this reaction: D + B----> A + E elaborate throughly with every step and annotationOrganotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Name compound and include stereochemistry if needed
- idk how to solve part b of this problem.-Name the correct mechanism via arrow pushing -name each mechanism -name each minor and major products (1R,2S)-1-iodo-2-methylcyclohexane + ethanol/DMSO/stirred 3 daysAlcohols are important for organic synthesis, especially in situations involving alkenes. The alcohol might be the desired product, or the OH group might be transformed into another functional group via halogenation, oxidation, or perhaps conversion to a sulfonic ester derivative. Formation of an alcohol from an alkene is particularly powerful because conditions can be chosen to produce either the Markovnikov or non-Markovnikov product from an unsymmetrical alkene. Using your reaction roadmap as a guide, show how to convert 4-methyl-1-pentene into 5-methylhexanenitrile. You must use 4-methyl-1-pentene and sodium cyanide as the source of all carbon atoms in the target molecule. Show all reagents needed and all molecules synthesized along the way.
- Fill in necessary products reactants or reagants of these reactions. Please note the existence of enantionmers in some cases.1. What is final product? 2. What are the 4 reactions & steps in mechanism (show product and mechanism steps)? 3. It is kinetic or thermodynamic?The reaction of an organic acid with an alcohol, inorganic solvent, to produce an ester and water is commonlydone in the pharmaceutical industry. This reactionis catalyzed by strong acid (usually H2SO4). A simpleexample is the reaction of acetic acid with ethyl alcoholto produce ethyl acetate and water: CH3COOH(solv) + CH3CH2OH(solv) ⇌ CH3COOCH2CH3(solv) + H2O(solv) where “(solv)” indicates that all reactants and products arein solution but not an aqueous solution. The equilibriumconstant for this reaction at 55 °C is 6.68. A pharmaceuticalchemist makes up 15.0 L of a solution that is initially0.275 M in acetic acid and 3.85 M in ethanol. At equilibrium,how many grams of ethyl acetate are formed?

