Q: Br CH,COONa DMF
A: SN2 reactionIt is a bimolecular nucleophilic substitution reaction in which the rate of reaction is…
Q: Silver sulfate dissolves in water according to the reaction Ag2SO4(s)⇌2Ag+(aq)+SO42−(aq) Kc=1.1×10−5…
A: Answer:Ionic compounds that have low solubility in water are called as sparingly soluble compounds.…
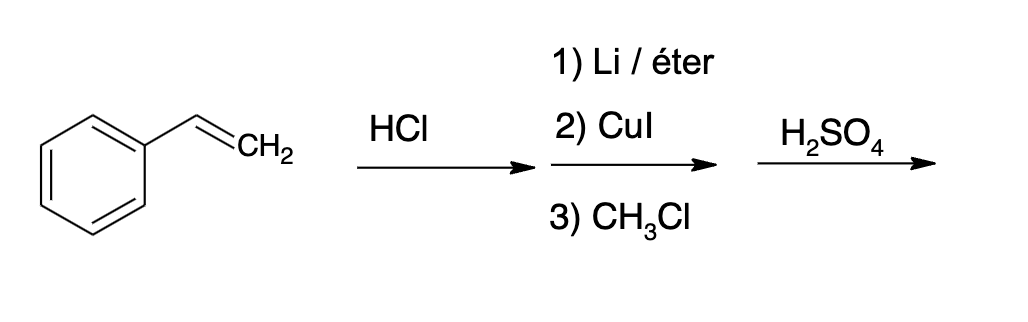
Q: 1. Provide the reagents necessary to carry out the following conversion. ہیں ملی
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Explain the basic way that each device detects radioactivity. Match the words in the left column to…
A: Given question is related to matching the words in the left column to the appropriate blanks in the…
Q: Br `H H₂CO H* + Ag(NH3)2* base
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Determine the pH change when 0.070 mol HNO3 is added to 1.00 L of a buffer solution that is 0.301 M…
A: The objective of the question is to determine the pH change.It is given that,The moles of are .The…
Q: 3 which one of the following bases is strong enough to efficiently deprotonate tert-butyl alcohol?…
A: Those base can deprotonate Acetic Acid whose conjugate acid is a weaker acid than acetic acid.That…
Q: What is the compound with the highest acidity in the following pairs? Explain why CO₂H CO₂H or
A: Acidity is defined as the ability of a molecule to release protons.If a compound easily donates…
Q: Which of the following is the strongest acid? 2018 -H (A) HH B OH R I-Z. (D)
A: In the question we have to find the most acidic compound for this We should know definition of…
Q: Determine the Eell of a voltaic cell composed of Ni(s) in a 1.00 M solution of Ni and Cd(s) in a…
A: E0 for Cd = -0.403 VE0 for Ni = -0.257 VT = 25oC[Ni2+]=1.00 M[Cd2+]=1.00 M
Q: The half life of silicon-32 is 710 years. If 60 grams is present now, how much will be present in…
A: The initial amount of silicon-32 is .The initial amount is .The time taken is .To find the final…
Q: its. te: Reference the Solubility product constants (K₁) table for additional infom
A: Given:-Mass of CaCl2 = 20gVolume, V= 3LFirst calculate the concentration is of CaCl2We know that…
Q: Which of the compounds shown most closely corresponds to the IR data: 2950-2850 cm ? Select the…
A:
Q: Draw the product formed when the structure shown below undergoes a reaction with diluted water…
A:
Q: 4. Draw the curved arrows and show the products formed for the following Lewis acid-base reac- tion:…
A: Lewis acid is a type of acid that is able to accept an electron pair in its vacant orbital while…
Q: This molecule, which is used in the manufacture of medications, would be best-named: Select one: O…
A: In aromatic compounds, if the substituents are present in the ring then we need to give numbers to…
Q: Methane gas and chlorine gas react to form hydrogen chloride gas and carbon tetrachloride gas. What…
A:
Q: H₂CO Na₂S₂04 H₂CO OH "OH R= ососн www
A: Given is a reduction reaction using Na2S2O4 reagent.To find the mechanism of the reaction
Q: Balance the chemical equation below using the smallest possible whole number stoichiometric…
A: Balancing of chemical equation is based on the fact that the same elements on the reactant side and…
Q: Br2 hv
A: the reactant is . It reacts with bromine in the presence of sunlight to give a brominated product.
Q: Draw the product of this reaction. Ignore inorganic byproducts. H₂/Pt
A: Alkynes can be completely hydrogenated into alkanes with hydrogen (H2) in the presence of a…
Q: 8. Assign R or S designations to each of the following compounds: CHO HO-C-H CH₂OH H CI-C-OH C(CH3)3…
A:
Q: Select the major product of the following reaction. OH J Z Z J OH ph ph ph ph I Oll DIV O No…
A:
Q: How many signals would you expect to see in the ¹³C NMR spectrum for the following compound? 13.
A: The number of signals in the carbon-13 spectrum is equal to the number of different types of carbon…
Q: Outline the mechanism showing both products formed for the reaction of ammonia (2 mole equivalent)…
A: The question is related to the conversion of haloalkane to alkylamine.The reaction takes places…
Q: a. d. e. Predict the major organic product(s) of each reaction below. For reagents over the arrow,…
A: HydrogenationHalohydrin formationHydroboration oxidationOxymercuration-demercurationWilliumson ether…
Q: Suggest a mechanism for the following reactions. Each will require multiple types of concerted…
A:
Q: 1A H2A 8A 3A 4A 5A 6A 7A He B C N O F Ne 18 28 Al Si P S Cl Ar Li Be Na Mg 38 48 58 68 7888 K Ca Sc…
A:
Q: What is the equilibrium constant at 25°C for the following reaction? Sn2+ (aq) + Fe(s) = Sn(s) +…
A:
Q: The bond angles in antimony trifluoride are 87°. Describe the bonding in SbF3, including a picture…
A: The bonding in a molecule can be explained as a result of overlapping interactions between the…
Q: Q) How many grams of AICI, are in a 2 mEq solution? [AICI, MW 133.5]
A: In the given question-> Milliequivalents of AlCl3 = 2 mEqMolar mass of AlCl3 = 133.5 gm/ mol
Q: 2. The following reactions, as drawn, have problems with them. Explain why each of these reactions…
A: Reaction of alkene with HI proceeds through the formation of carbocation intermediate. The most…
Q: For many purposes we can treat ammonia (NH3) as an ideal gas at temperatures above its boiling point…
A: The objective of this question is to find the final pressure of a sample of ammonia gas when the…
Q: Provide the IUPAC name for the structural condensed formula shown here. CH3 CH₂ CH₂ CH₂ CH₂ CH₂ CH3
A: The objective of the question is to find the IUPAC (International Union of Pure and Applied…
Q: C,H,Br: 8 2.0 (d, 3H), 5.3 (q, 1H), 7.6 (m, 5H).
A: Given molecular formula is, C8H9Br
Q: Identify the chiral carbon atoms by selecting each atom and assigning it a map n until all atoms are…
A: Chiral carbons are those which have 4 different groups attached to it, hence do not possess any…
Q: Draw a curved arrow mechanism for the reaction, adding steps as necessary. Be sure to include all…
A: The representation of the path followed in a chemical reaction using the curved arrows showing the…
Q: A galvanic cell at a temperature of 25.0°C is powered by the following redox reaction:…
A: T = 25oC=25+273=298K[Ca2+] = 6.08M[Cr3+] = 1.96 Mn=6 as 6 electrons are involved in the reaction
Q: What volume of 1.5 M AgNO3 is required to precipitate the chloride from 30.0 mL of a 0.45 M HCI…
A:
Q: of Cl is greater than that of Si + sume pulovity develops goving polar. 3. Predict the enthalpy of…
A: Standard enthalpy of formation is defined as the energy change taking place during formation of one…
Q: Balance the following redox reaction in acidic solution. NO(g)+Cl2(g) ---> NO3^-(aq)+Cl-(aq)
A: Given,
Q: Draw the product of this reaction. Ignore inorganic byproducts. Br Br 1. NaNH2, A 2. H3O+ Drawing Q
A: Gem dihalides on treatment with strong base like sodamide forms terminal alkynes.
Q: Methane gas and chlorine gas react to form hydrogen chloride gas and carbon tetrachloride gas. What…
A: Given:A balanced chemical equation is a representation of a chemical reaction using chemical…
Q: 1. Predict the product of the following reactions. Ph -CC13 Et NO₂ HCI HBr DBr HI
A:
Q: OH 1. 00
A: Ketones can be prepared from carboxylic acid by treating it with suitable reactants. One suitable…
Q: H 3. What is the relationship between the two molecules? CI CI CH3 OH Н3С....... HO I. Diastereomers…
A: Isomers having same molecular formula are two types -a. Constitutional isomers : They have same…
Q: Curved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and…
A: The objective of the question is to draw the product of the reaction.
Q: Determine the equilibrium constant, K, for the voltaic cell composed of the following 2 half…
A:
Q: A student used 26.28 mL of hydrochloric acid to titrate a 0.1166 gram sample of primary standard…
A: The question is based on the concept of titrations.We need to calculate molarity of hydrochloric…
Q: What is the more stable radical in each pair? A B Pair 1 Pair 2 A C A. B. C. Pair 1 A A B D. B B D…
A: A radical is an atom, ion, or molecule that contains a minimum of one unpaired valence electron.…
Step by step
Solved in 3 steps with 1 images


