Q: How many molecules of the sweetener saccharin can be prepared from 30 C atoms, 25 H atoms, 12 O…
A: The structure of the molecule is given as:
Q: Will the following reactions occur as written? 1. Nat OEt CH3CCH,COCH,CH3 CH;CH2CO2H 2. CH3B. 3.…
A: Active methylene compound: Compounds which possess a methylene bridge located between two strong…
Q: HNO 3, H2SO4 HNO 3, H2SO4 HNO 3, H2SO4 'N ZI
A: Nitration is a type of organic reaction for the introduction of nitro group into an organic…
Q: Reaction B. CI (CH3),COK (CH3),COH
A: Two questions based on substitution and elimination reactions, which are to be accomplished.
Q: Oxidation Reaction CH,-CH-C-H CH, CH3-C-CH3 Reduction Reaction CH;-CH–C-H CH; CH;-C-CH3
A:
Q: when 10.5 of HgOcss CMV 216.3 9g Imol is decomposedto Form Hg a) and o 2zcg) e Zcg) 8 the heat or…
A: Provided amount of HgO=10.5g Molar mass of HgO=216.39g/mol The reaction is 2HgO(s) → 2Hg(l) +O2(g)…
Q: What is the product of the following reaction? 1. LIAIH, (excess) ?? 2. H3O* Он N- YOH Он N- OH
A: Lithium aluminium hydride is a versatile reducing reagent. This reagent is used for the reduction of…
Q: Calculate the ?H (in kcal/mol) for the reactions (use table above): a)H2 + O2 → 2 H2O b) CH4 + 2 O2…
A: Enthalpy of the Reaction is the difference of sum of energies of Reactants and Products. It can be…
Q: -NH2 SOCI, HO,
A: The given reactant is a 3-carbon carboxylic acid known as propionic acid. It shows substitution…
Q: determine DHorxn for the reaction below: 2 H2S (g) + 3 O2 (g) --> 2 H2O (g) + 2 SO2 (g)
A: Delta H reaction is
Q: Consider a mixture of the compounds shown below dissolved in hexane (C,H;a) CH3(CH)10CH3 CH,COOH…
A: Note: According to our guidelines we are supposed to answer only one question. Kindly repost other…
Q: What is the milliequivalent weight (mEq wt.) of Sodium Bicarbonate (baking soda)?
A: Sodium bicarbonate = NaHCO3 Molar mass = 84 g/mol Acidity = 1 ( Acidic hydrogen per molecule). The…
Q: Which of the following compounds is generated by the reaction below? он (1) B2He, diglyme но. (2)…
A: Hydroboration–oxidation reaction: Alkene gives an electrophilic addition reaction with borane.…
Q: What is true for this reaction? CI4(1) + 4 HCI(g) CH4(g)+ 4 Cl2(g) O AHrxn 0 AHrxn 0; ASrxn 0;…
A: For the reaction ., CCl4(l) + 4HCl(g) ----> CH4(g) + 4Cl2(g) Bond energy ., For C-H bond = 414 KJ…
Q: Alcoholic fermentation converts glucose (C6H12O6) into ethanol (C2H5OH) and carbon dioxide (CO2).…
A:
Q: What si na limiting reactant of NaOH/0.05moles+HCl/0.05moles=
A: Given: To find limiting reactant of reaction of NaOH/0.05moles HCl/0.05moles
Q: C5H8 + 7O2 → 5CO2 + 4H2O type of reaction
A: The given reaction is combustion reaction because it involves oxygen. it is specially…
Q: Find AHxn for the reaction: 3C(s) + 4 H2(g) → C;Hs(g) Use the following reactions with known AH's:…
A: Given C3H8(g) + 5O2(g) → 3 CO2(g) + 4 H2O(g) ∆H = - 2043 KJ…
Q: The product P in the following chemical reaction will be 0= CI 1. NaN3 2. Heat 3. H₂O P
A:
Q: Study the reactions. 1. C2H2 (g) + 2H2 (g) → C2H6 (9) 2. C2H4 (9) + H2 (g) → C2H6 (9) 3. C2H2 (g) +…
A:
Q: Use the following reagent table for the reaction of: cyclohexanol + H2 SO4 → cyclohexene Chemical MW…
A: Limiting reagent is that react completely in the reaction. In this reaction, 1 mol of cyclohexane…
Q: OH CH KMNO,
A: Interpretation: The given reaction is to be completed. Given reaction:
Q: Give the correct organic product for the following reaction at – 78 °C: 1. Li*[AlH(OtBu),] CI 2. Н,О
A: Lithium tri-tert butoxyaluminum hydride (LiAlH(Ot-Bu)3) is a mild reducing agent that can produce…
Q: Br FeClz or AICI3 Br2 + HBr Sulfonation O Hydration O Halogenation O Hydrohalogenation
A:
Q: 3. Alcohol Starting Materials 1. methanol 2. ethanol 3. 1-propanol 4. 2-propanol 5. cyclohexanol a.…
A: In the Jones oxidation, secondary alcohols are oxidized to ketones while primary alcohols undergo…
Q: OH H2CrO4 PHCHO E F heating NaOH MeSH H2CrO4 H HO excess H*/MEOH MeONa J
A: The details solution for this reaction is provided below in attach image.
Q: Reaction 1 Reaction 2 Reaction 3 Reaction 4 Reaction 5 Reaction 6 HF + HCOOH + + + NH₂* H₂PO H₂SO4…
A: As per the guidelines we are supposed to answer first three parts , to solve remaining parts please…
Q: A. COMBINATION or SYNTHESIS REACTION: A + B→AB 1. Fe +_O2 → _Fe;O3 2. Li+_d →_ Lid _C;H2 + _F…
A: Answer A. Combination reactions 1) 4Fe + 3O2 → 2Fe2O3 2) 2Li + Cl2 →2LiCl 3) C2H2 + 2F2 → C2H2F4 4)…
Q: Butene can be reduced to butane by the addition of hydrogen as shown in the following balanced…
A: Mass of butene = 28 g
Q: A + B ↔ AB is 3.793 What is the Keq for 2 + AB ↔ 2 A + 2 B Keq = ?
A: Given :- Keq for A + B ↔ AB is 3.793 To be calculated :- Keq for 2 AB ↔ 2 A + 2 B
Q: ow many equivalents of an acid could be reacted by 1.750 g Al(OH)₃?
A: To calculate equivalents of acid that can be reacted by 1.750 g Al(OH)₃.
Q: a) Water with 0=SeCl2 to form O=Se(OH)2 b) Water with O=SeCl2 to form SeO2 c) Methanol with POC13 to…
A: As you not specified so I am giving answer of first 3 subparts. First nucleophile attack on…
Q: 2 C5H10 (1)+15 O₂ (g) → 10 CO₂ (g) + 10 H₂O(l) a. Calculate 4H of the reaction. b. Calculate 4S of…
A: Dear student , since you have posted multiple parts questions we will allow to solve only first…
Q: 20 g of CH3CONHCH3 is formed in the following reaction from 30 g of acetic acid (CH3COOH). Calculate…
A: Given chemical reaction, CH3NH2 + CH3COCl ------> CH3CONHCH3 + HCl Molar mass of CH3COCl =…
Q: For the following reaction, 2H2S (g) + O2 (g) = 2s (s) + 2H2O (g) Explain what happen to a. [02] if…
A: According to Le Chatelier's principle state, when an equilibrium reaction experiences a disturbance…
Q: 2) If cobalt(II) sulfate is heated too strongly, the following reaction will occur COSO(s) → Co0(s)…
A: In the case of inorganic chemistry, where hydrate is a salt in which H2Ois included in the ionic…
Q: Consider the reaction sequence below to answer the following questions: L. NaOE, EOH CO NaO, OH…
A: Compound X has two ester groups connected by a -CH2 unit .The IUPAC name of X is diethyl…
Q: Through several successive reactions, a chemist uses carbon, Cao, HCI and 20 to prodluce CoHuClz…
A: Answer is explained below. Given that, percent yield = 75% Molar mass of C6H4Cl2 = 147.09 g/mol.…
Q: CH, c=c CH3 H (1) major organic product ROOR hv (3) process H20, Hgso, (2) enol product (4) keto…
A: The first substrate undergoes polymerization reaction in the presence of peroxide, ROOR and light.…
Q: what is the best reagent for the conversion shown below? OH H3C CCH,COCH3 H3C-CHCH,CH,OH Select one:…
A:
Q: 1. For the reaction: H2 +Cl2 -----> 2HCl, 3.7 moles of H2 are mixed with excess HCl. How many moles…
A: 1. For the reaction: H2 +Cl2 -----> 2HCl, 3.7 moles of H2 are mixed with excess HCl. How many…
Q: 1) NaBH, 2) H30* no reaction OH HO
A: It is reduction reaction
Q: 2. Given: 2H2S(g) + O2(g) 25(g) + 2H20(g) a) What happens to (H20] if O2 is added? b) [O2] if H2S is…
A: Le chatelier principle says that equilibrium will shift in such a way so as to undo the effect of…
Q: If a given reaction is A + B --> 2C with ΔH = -8.8 lJ and the needed reaction is 2A + 2B -->…
A: Given:When 1 mole of A react with 1 mole of B, then ΔH = -8.8 KJ
Q: C₂H₂ + O₂ CO₂ + H₂O The chemical equation represents the combustion of propane. When correctly…
A: Given unbalanced chemical reaction of combustion is : C₂H₂ + O₂ --------------> CO₂ + H₂O…
Q: Give the minor product(s) of the following reaction. CH3CH2CI (1 mole) AICI3, heat U There is no…
A: The given reaction is represented as follows:
Q: What reagents are required for the below conversion? OH ? HO, (a) NBS; Br2, H,O; NaOH, H,O (b) Brz,…
A:
Q: 16) HNO, H;SO4(catalyst) NH2
A: Given reaction:
Q: Reaction 1 H,C-C- CH, CH, HC-C- CH,CH, он A Reaction 2 H ex cess conc, H, SO, H,C-C-CH,CH, tut-1-ene…
A: First we see about theoretical yield: Mw of compound A = 87.11 g/mol Mw of butanone = 72.11 g/mol It…
Q: Give the major product(s) of the following reaction. Cl2 (1 mole) OMe heat OMe CI CI OMe OMe CI CI…
A: The methoxy group, OCH3, is ortho-para directing group and thus the formation of meta substituted…
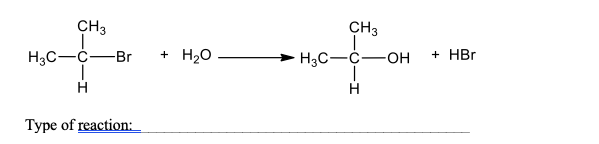
What type of reaction is shown?
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- When (CH3CH2)3CBr is added to CH3OH at room temperature, the product is(CH3O)C(CH2CH3)3. Propose a mechanism(s) for the reactions leading to theseproducts and use curved arrows to show the movement of electrons.Oximene and myrcene, two hydrocarbons isolated from alfalfa that have the molecular formula C10H16, both yield 2,6- dimethyloctane when treated with H2 and a Pd catalyst. Ozonolysis of oximene forms (CH3)2C = O, CH2 = O, CH2(CHO)2, and CH3COCHO. Ozonolysis of myrcene yields (CH3)2C = O, CH2 = O, (two equiv), and HCOCH2CH2COCHO. Identify the structures of oximene and myrcene.The alcohol compound güven the formula below is used in perfume making.bromine benzeme and -1 synthesis of this compound using the necessary organic and inor chemicals show
- Reagent 5 options; OsO4 NaBH4 Methanol NaOH, H2ODraw the structure(s) of the major organic product(s) of the following reaction. NH₂ O₂N NaNO₂ aqueous HCI at 0°The key step in a reported laboratory synthesis of sativene, a hydrocarbon isolated from the mold Helminthosporium sativum, involves the following base treatment of a keto tosylate. What kind of reaction is occurring? How would you complete the synthesis?
- Draw the products 1. (S)-2-chlorobutane and sodium acetate in DMSO 2. 1-bromopropane and methylamine in acetonitrile.Cyclohexene plus 1) Hg(OAc)2, H2O; 2) NaBH4; yields __________. HO2CCH2CH2CH2CH2CO2H a cyclic diketone cyclohexyne cyclohexanol OHCCH2CH2CH2CH2CHOStarting from acetone and methanol synthesize isobutylene (CH2=C(CH3)2)
- KMnO4, warm, conc'd reacts with hept-1-ene to yield __________. CO2, hex-1-ene CO2, hexanoic acid Formic acid, pentanoic acid Ethanoic acid, pentanal Formic acid, hexanoneComplete the reaction map by providing the answer from A-E. Write the IUPAC name of the products. conc HNO B conc H,SO, B, D FeBrs conc HNO, E AIC conc H2SO. conc H2SO4A reaction of an alkene plus H2O and excess acid.

