Q: Propose an efficient synthesis for each of the following transformations OMe O;N.
A: The first synthesis pathways involved in the formation of an olefin intermediate, where as second…
Q: Draw the complete mechanism for the following transformation. 1. MgBr 2. H20 Draw the complete…
A:
Q: Which one of these compounds would be the best starting material for a malonic ester synthesis? CH3…
A:
Q: Which of the following reactions does not give the carbonyl product shown? a. Which of the reactions…
A: Carbonyl group chemistry is the most important chemistry in organic synthesis. There are some…
Q: Use a sheet of paper to answer the following question. Take a picture of your answers and attach to…
A: Enamine intermediate: An enamine intermediate is defined as an α,β-unsaturated amine compound which…
Q: Which of the following will NOT produce an ester with an acid chloride? O CH3CH2OH O (CH3)3COH O…
A: Let an acid chloride R-CO-Cl As we know that Cl is a leaving group .So when nucleophile will attack…
Q: What is the expected product A for the following intramolecular aldol reaction? CH3 O NaOH A H3C H.…
A:
Q: In reactions with an aldehyde or ketone to form an imine or an enamine (Section 16.8)
A: Solution : Aldehyde or ketone shows dehydration reaction with an amine to form imines. Imines…
Q: Он но он HO H* но HO HO но
A:
Q: 5. Draw the major organic product of the following reaction? 1) KOI CH, 21A
A:
Q: What is the major enolate formed in each reaction ?
A: For useful alkylation reactions of enolate anions, in the absence of other strong nucleophiles and…
Q: CH3NH2 H30* e) j) NaOEt Aldol Condensation
A: Given reactions,
Q: Would the following reaction form the product as shown? (CH3CH₂)2NH H A) Yes, it is easier to form…
A: Would the following reaction form the product shown? = A) Yes, it is easier to form the diketone…
Q: Define the following reagents or functional groups as being related to either aldehydes/ketone AK,…
A: Reagent have multiple character i.e selectivity and reactivity . Example :- LiAlH4 is a strong…
Q: What organolithium reagent and carbonyl compound can be used to prepare each of the following…
A: a.
Q: Which of the following nucleophiles will undergo conjugate addition in a Michael reaction? O lithium…
A: The Micheal reaction Or Micheal addition is a conjugate addition (1, 4-addition) of the carbanion to…
Q: Rank the carbonyl-containing compounds in order of reactivity towards nucleophilic attack. Most…
A:
Q: Dehydration via an E1cb mechanism Michael Reaction Intramolecular Aldol Reaction В KOH / ELOH / heat…
A: We know, Micheal reaction is basically the nucleuphilic addition reaction which is basically 1-4…
Q: 4. Give the structures of the aldol products that form when each of the following compounds or…
A: Hey, since there are subpart questions posted, we will answer three subparts. If you want any…
Q: Dehydration via an E1cb mechanism Michael Reaction Intramolecular Aldol Reaction А C KOH / ETOH /…
A:
Q: Which of the following statements about enolate anions is false? 1. Enolate anions can act as…
A: Enolate ion is formed when base can abstract alpha hydrogen . Hence, there must be alpha hydrogen…
Q: For the following reaction schemes, fill in the appropriate reagents and starting materials you…
A: For the formation of nabumetone, from acetoacetic ester and acetone following reagents are used, the…
Q: C. NaOCH3 d. NaOH Ph
A:
Q: Draw a structure for the major product in each of the following reactions DMSO OTs CI NaOCH;
A: An acyl substituted compound and an alkene will be the major products for these given reactions.
Q: The following molecule undergoes an intramolecular reaction in the presence of pyrro- lidinium…
A:
Q: Draw the structures A, B and C for the reaction sequence below. Michael Reaction A Dehydration via…
A: Given are 3 reactions. Michael reaction, aldol condensation and dehydration.
Q: Draw the enol tautomers for each of the following compounds. For compounds that have more than one…
A: a) Ketones can undergo a type of isomerism called tautomerism. The isomers/tautomers formed are…
Q: What is the name of the carbonyl compound that has served as common starting material for synthesis…
A: We have to find out the carbonyl which can form all the compound by reacting with different…
Q: Draw the major product formed when the given epoxide reacts with aqueous acid. Select Draw Rings…
A:
Q: D) 3) Which of the following reagents will quantitatively convert an enolizable ketone to its…
A: Introduction: For alkylation reactions of at enolizable aldehyde and ketone it is essential, to…
Q: Hydrolysis of an acetal in aqueous acid gives an aldehyde or ketone and two molecules of alcohol or…
A:
Q: i) Synthons: Synthetic equivalents: ii) Synthons: OH Synthetic equivalents:
A: By disconnection , the fragment ions i.e. cation and anion are formed that may be real or imaginary…
Q: Use a sheet of paper to answer the following question. Take a picture of your answers and attach to…
A: In this question, we have to synthesized the given carbonyl products a) and b) by using enamine…
Q: e following carbonyl-containing compounds in order of reactivity towards nucleophilic acyl…
A: ->For reactivity towards nucleophilic acyl substitution reaction we see electrophilicity of…
Q: 6.1.3 Draw the product(s) of the following transformation.
A: Mechanism
Q: When a 1:1 mixture of acetone and 2-butanone is treated with base, six aldol products are possible.…
A: Each pair should have two isomers so totally six products in the below the scheme have chiral…
Q: ) Which of the following compounds is the least reactive toward nucleophilic acyl substitution?…
A: Nucleophilic acyl substitution defines a subset of nucleophile and acyl compound substitution…
Q: (CHz) 2 CH3 doble
A: Given Product = To be determined
Q: (1] CH,CH,MgBr [2] H20
A:
Q: 2. Draw the product of the reaction of cyclopentanone with each of the following reagents: a.…
A: The correct answer is given below
Q: Draw the structure of the product that is formed when the compound shown below is treated with the…
A: ->reagent -1-PBr3,Br2 2-H2O
Q: Cardiolipins are found in heart muscles. Draw the products formed when a cardiolipin undergoes…
A: The mechanism is as follows:
Q: When a cyclic ketone reacts with diazomethane, the next larger cyclic ketone is formed. This is…
A:
Q: 3. Which structure represents an enolate? (4 pts) I3D II IV a. I b. II с. Ш d. IV e. None of these
A:
Q: how the r-group affects the elimination, specifically E-1 reaction and how a different r-group would…
A: E1 elimination reaction is unimolecular elimination reaction. This is two step reaction. First…
Q: Please draw out the 2 reaction schemes: Ethyl diacetoacetate + hydrazine = pyrazole Ethyl…
A:
Q: Show how each transformation may be accomplished by using a nitrile as an intermediate. You may use…
A: (a)
Q: Draw the product of treating the following anhydride with CH3OH.
A: The reaction of the given anhydride with methanol will give the product as given below.
Draw the structure of the major enolate and both resonance contributors for the given
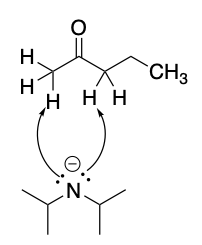
Step by step
Solved in 3 steps with 2 images


