Q: Select the best answer or answers from the choices given: Molecules formed when electrons are shared…
A:
Q: 3. Describe (or draw) how NaCl salt is formed (hint: involves charges).
A: Compounds formed from the reaction of respective elements.
Q: 1) For the given elements, place them in order of INCREASING ELECTRONEGATIVITY: -Mn, N, Ga, P 2)…
A: Electronegativity : N > P > Ga > Mn. N belongs to second period. Therefore more…
Q: Write the name from the formula or the formula from the name for hydrated ionic compound. LiNO2.H2O
A: Hydrated molecules are those molecules which contains water molecule in its formula. Example:…
Q: O O O O The atoms in a water molecule are Polar 9. Hydrogen Oxygen Covalent
A: We know that, Molecular formula of water= H2O
Q: Which of the following is true about polar colavent compound: a. only polar covalent compounds will…
A: Polar covalent compounds are the ones in which the bonded atoms have difference in…
Q: Rank the lattice energy from high to low: BaO NaF SrO NaI
A: We have to predict the order of lattice energy.
Q: v Question Completion Status: QUESTION 15 In the picture below, what is the best description of the…
A:
Q: A look ahead: Suggest a neutral ionic compound that would be made of the magnesium ion (Mg2+) and…
A: The most common ion f by the nitrogen atom is nitride ion N-3 The charge on nitride ion is -3 . The…
Q: The ability of an atom to attract electrons in a bond with another atom istermed itsa.…
A: In an atom the core area is called nucleus which contain protons and neutrons. In an atom, electrons…
Q: Use the following elements to demonstrate electroneutrality? Na and O . Iron (TIT) and chlorine Ba…
A: To demonstrate the electroneutrality principle using the following elements: (i) Na and O (ii)…
Q: 1. Re-draw the structure of KHP into your laboratory notebook (Show all carbon atoms instead of just…
A: KHP - Potassium Hydrogen Phthalate is used as a primary standard in our laboratories. It is an…
Q: London force or induced dipole is a type of IMF that is observed in: polar molecules only ionic…
A: London forces or induced dipole are weakest intermolecular forces and it results in interaction…
Q: СООН 12 3 2 1
A:
Q: 16. Name the structure CI .CI 17. Name the structure OH
A: (13) In the given compound (13), parent chain is 2-hexene and methyl group is attached in 4th…
Q: ch3nh3+ what is the polarity and justification
A: It is asked to justify the polarity on CH3NH2.Lets discuss.
Q: Which of the following is chloric acid?
A: Given : Which of the following is chloric acid?
Q: Determine the formula of the compound formed from hydrogen ions and sulfate ions. options: H2SO4…
A: The answer is given as follows The reaction of hydrogen ions and sulfate ions produce H2SO4
Q: MCQ 153: The strength of an atom to attract electrons increases with the increase in A. polarity B.…
A: Solution: By the defination of electronegativity we can say as electronegativity increases, the…
Q: Water and the human body. How does a human being’s water content change throughout his life? (fetal…
A: a) Human being’s water content throughout his life - 1) Water is of major importance to all living…
Q: Select the best description for each compound, using each description only once. Na3PO4 : KOH:…
A:
Q: 6. Calculate the percentage of water in the compound CaSO,•6H,O. 7. Calculate the percentage of…
A: Answer: During the crystallization of certain compound, a few molecule remains the crystal of the…
Q: Determine the number of k+ and f-to form a neutral ionic compound
A: The elements in the left end of the periodic table are metals and the elements in the right end of…
Q: When the toluene layer (containing methyl benzoate and benzoic acid) was added to the aqueous…
A:
Q: Compare dissolving GCl(g) in water to dissolving CH3COOH(l) in water Part A: When HCl(g) is…
A: An acid is a substance that gives ions in its solution whereas a basic substance gives ions in its…
Q: Why would one use an aqueous solution of NaCl to prepare solutions of deionoized water?
A: Deionized water contains no ions in it where as the sodium chloride solution having sodium(Na+) ion…
Q: H-C Select one: O 1 O 2 O 3 O 4 O 5
A: I Think its 2 just like benzene
Q: molecules are unlikely to dissolve in water easily. molecules with polar and nonpolar areas polar…
A: Answer .......... molecules are unlikely to dissolve in water easily…
Q: E hydroxide ions
A:
Q: Would you expect the attraction to be stronger between a hexane molecule and a water molecule or…
A:
Q: Write the formula of the compound more likely to form from: a) Cuprous ion and peroxide ion b)…
A:
Q: Unlike soap, which is ionic, some liquid laundry detergents are neutral molecules. Explain how the…
A:
Q: TRUE OR FALSE 1. A bond between two nonmetals always acts like a covalent bond 2. The bonding…
A: 1. A bond between two nonmetals always acts like a covalent bond 2. The bonding electrons in the N-H…
Q: MCQ 49: In excess water, Ca(OH), produces A. Ca 2 ions B. H2g) C. Calcium oxide (CaO) D. calcium…
A: When we add Ca(OH)2 in excess of water then we get Ca(OH)2(s) + H2O(aq) ⇌ Ca2+(aq) + 2 OH- (aq)
Q: B. Identify what kind of Bond is present in the following molecules. (You may show your solution 1.…
A: This question is related to chemical bonding. Bonds present in molecules may be of mainly three…
Q: Does NaCl(s) conduct electricity at room temperature? Yes/No and Explain why?
A: Electrolyte is a substance that is able to conduct electricity in its aqueous solution. This is due…
Q: Provide the structural formulas for A-D for the following reactions HCI B AICI3 1) THF:BH3 2) H2O2,…
A: Alkenes undergo electrophilic addition reaction with HCl to give alkyl chloride which undergo…
Q: A) Which of the following bonds would dissolve well (be soluble) in water? Explain your choices. a)…
A:
Q: (NH4);CO;(s)-→ H;O(g) + (8) + (8)
A: Ammonium carbonate decomposes to give ammonia ,carbon dioxide and water molecules
Q: Calculate the number of moles of Cl- ions in 1.75 L of 1.2 *10-3 M ZnCl2.
A: The number of moles of chloride ion in ZnCl2 is double the number of moles of ZnCl2 .
Q: Assuming that the effect of bond energy is more important than the effect of electronegativity, the…
A: Bond energy of HI is 295 kj per mole while bond energy of HCl is 428 kj per mole . having less bond…
Q: The morality of 0.1 N phosphoric acid is 0.3 False True
A:
Q: how would a molecular structure of salt affect the appearance of salt that you see with your eyes?
A: Salt crystals are translucent and cubic in shape; they normally appear white but impurities may give…
Q: How do I determine if a solvent is polar or nonpolar based on structure
A: We have to tell how we can identify whether given solvent is polar or non polar
Q: Write the chemical name for N2H4. Type of Bonding: Show Lewis Structure or Bonding in the solution…
A: The details about N2H4 is given below
Q: OH H,SO4 NH 2.
A: Solution : Beckmann rearrangement is the acid-induced rearrangement of oxime to an amide.…
Q: Write name for the compound
A: The given compound contains a benzene ring and two carboxyl groups on adjacent carbons. It is an…
Q: Calculate the number of oxygen atoms in a 120.0gsample of diphosphorus pentoxide P2O5
A:
Q: 35. Which of the following does have the highest boiling point? molar masses: C = 12.01 g/ mole,…
A: Among I2, SO2,HCl and CO which will have highest boiling point.
Draw a balanced chemical equation (including any necessary charges) for the formation of a hydrochloride salt below.
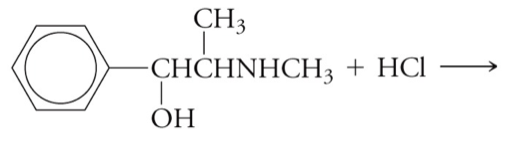
Step by step
Solved in 2 steps with 2 images


