Q: How many Faraday is needed to deposit 6 moles of Zinc (Zn) from ZnCl2 solution using electrolysis pr...
A:
Q: 2) Which of the following has the lowest pKa? NH3 NH3 NH3 NH3 OMe NO2 CH3 B C A
A: Lowest pka means Strongest acid. Therefore, we have to find the strongest acid among these.
Q: ion 3: (a) What is the second law of thermodynamics? For any process, there are four possible sign c...
A: Given, Enthalpy of Vaporization = 58.51 KJ / mol ...
Q: [Refe Which of the following is the ground-state electron configuration of a Ct ion? O 1s 2s O 1s 2s...
A: Given, The ground-state electron configuration of C3+ ion is :
Q: What I Know Direction: Write P if the mixture can be separated by picking out, E i. Do it using your...
A: Decantation is a process for separation of mixture of immicible liquid and a solid mixture such as s...
Q: dont
A: An acidic substance has a greater amount of hydrogen ions and mathematically, the magnitude of hydro...
Q: Predict whether each of the following has a dipole moment. Show shifting of electron density. (a) ...
A: Overall dipole moment of a molecule is the vector sum of dipole moments of individual bonds and lone...
Q: Potassium bromide solution at a molar concentration of 0,0500 mol/ is titrated by silver nitrate wit...
A: Br-(aq) of potassium bromide, KBr solution reacts with Ag+(aq) of AgNO3 solution to form silver brom...
Q: consider the attached picture for reference: 1. What type of reaction is involved? a. Addition Rea...
A: Atoms or molecules are chemically combine or dissociate to form new atoms or molecules. this is know...
Q: Which has the maximum weight in the following? 10 moles of H2O2, 100 molecules of NH3, 1000 molecule...
A:
Q: 3. Standard white vinegar you can buy in the grocery store is 5% concentration. That means 5% of the...
A: A question based on calibration graph that is to be accomplished.
Q: the entropy of argon is given to a good approximation by the expression: Sm( in J/K-mole) = 36.36 +...
A: entropy and Gibbs free energy are related to each other
Q: но.
A: A detail solution is given below -
Q: Calculate pCl for the titration of 100.0 mL of 0.1000 M Cl− with 0.1000 M AgNO3 for the addition of ...
A: Given, Concentration of Cl- = 0.1000 M Volume of Cl- = 100.0 mL a). Concentration of AgNO3 = 0.10...
Q: Give some treatment methods for wastewater and explain.
A: We can make use of wastewater by wastewater treatment technologies. There are many methods for was...
Q: Given the hypothetical thermochemical equation: A + B —> C + D ΔH = - 430 kJ Which among the follow...
A:
Q: Two elements in Period 4 are adjacent to one another in the periodic table. The ground-state atom of...
A:
Q: For the complex [Co(H20)61*, is the magnetic moment, Pobs. O a. Temp. dep and Hobs<Hs.0 O b. almost ...
A:
Q: Which of the following is the number of microstates contained in an electronic state with term symbo...
A: A question based on electronic effect that is to be accomplished.
Q: Weaker the intermolecular forces of attraction larger will be the rate of evaporation Select one : T...
A:
Q: 1) HNO3, H2SO4 2) Fe, H3O* 3) NaNO2, HCI, < 5 deg C 4) CuCN
A: Given
Q: 1. Problem In analytical chemistry accuracy describes: (a) How wide is data spread (b) How close to ...
A:
Q: titrimetric determination of acetic acid (60.05 g/mol) in vinegar. Titration of a 5.00 mL vinegar sa...
A:
Q: (a) An ideal gas undergoes an adiabatic expansion into a vacuum. Are AS, ASsurrounding and AStotal p...
A: Hii there, As there are multiple question posted. we are answering first question. If you need furth...
Q: all of the above.
A:
Q: What is the molarity of Na+ in a solution of NaCl whose salinity is 5.6 if the solution has a densit...
A: Molarity of Na+ can be calculated using the moles of Na+ present in the one litre of solution.
Q: DI 2 H20 CH,Cl2 II
A: According to the question, we need to determine the product of the reaction. At first, alkene is tr...
Q: What is the concentration of 50.00 mL HCl solution prepared from 15.00 mL of 12.0 M HCl
A: Given, The concentration of 50.00 mL HCl solution prepared from 15.00 mL of 12.0 M HCl is :
Q: Question:- why do Chloride atoms form ions under biological conditions while other atoms (for examp...
A: According to octate rule all atoms wants to fill up the octate i e.wants to fill the valance cell by...
Q: Determine the &ntropic contribution for polyethylene (C2Hu] that hoas a moleculas weight 200 / mol 9...
A: Given, M(H) = 1.008 g/mol M(C) =12.01 g/mol polyethylene molecular weight= 200 g/mol Entropic contri...
Q: A worker accidentally ingested a radioactive form of inorganic phosphate (32P;). 32P; could be incor...
A:
Q: A solution contains 1.20×102 M potassium sulfide and 7.43×103 M sodium hydroxide. Solid copper(II) a...
A:
Q: Which of the following statements is true concerning the decomposition of liquid water to form hydro...
A:
Q: Question 4: (a) What are redox reactions? Explain the difference between a gal- vanic and an electro...
A: In certain reactions, transfer of electrons takes place from one species to another. There are two ...
Q: Be as explicit as you can in describing how the covalent bond between an atom of Chlorine and an ato...
A: Given: Bond is forming between a chlorine and an iodine atom.
Q: Calculate the molarity of a solution containing 241 grams of NaCl in 1836 ml of solution? (Atomic ma...
A: Molarity: It is number of moles of solute present in 1 litre of solution. It is denoted by M and its...
Q: Using periodic trends, arrange the following elements by increasing the first ionization energy: Ar,...
A: Ionization enthalpy is the amount of energy required to remove an electron from valence shell of an ...
Q: A solution contains 1.19x102 M ammonium carbonate and 1.39×102M sodium sulfide. Solid nickel(II) nit...
A: Precipitation occurs when ionic product is greater than solubility product Thus among two substance...
Q: Question 2: (a) Under what conditions is a dA <0 condition that defines the spontaneity of a process...
A:
Q: Consider the following reactions: 1. 2 CH4(g) + O2(g) ⇄ 2 CO(g) + 4 H2(g) Kp1 = 9.34 × 1028 2. ...
A: Given Kp1 = 9.34 × 1028 Kp2 = 1.374
Q: (b) Why does the liquid-gas coexistence curve in a P-T phase diagram end at the critical point? Draw...
A: A substance can exist in three different states: solid, liquid, and gas. The existence of the substa...
Q: In the following disproportionation reaction, distinguish the element that undergoes oxidation and r...
A: Given :- 2H2O2(aq) → 2H2O(l) + O2(g) To identify :- Element that undergoes both oxidation and red...
Q: Baking soda is a pure substance, sodium bicarbonate. Vinegar is a mixture of acetic acid dissolved i...
A: Given that - Vinegar Solution in water and solid sodium bicarbonate react to give following balance...
Q: Given the following thermochemical data at 25°C and 1 atm pressure, 3/2 02(g) + 2B(s) = -1264 kJ; - ...
A:
Q: A solution contains 1.23×102 M potassium sulfide and 1.34x102 M sodium carbonate. Solid cobalt(II) a...
A:
Q: Br "OCH,CH3
A: Reaction of alkyl halides with nucleophile is termed as nucleophilic substitution. It is competed by...
Q: Question 1 (a) Distinguish between the differential rate law and the integrated rate law. Which of t...
A:
Q: ÇONH, iv) кон, в H20, A Br 87% ÇH2-CEN H,0 HCI 40°C CH,CH;OH vi) CH,=CH-C-OH CH;=CH-C-C ÇOCI он NaOH...
A:
Q: Which Keq value indicates the greatest concentration of products at equilibrium assuming similar sto...
A: Keq = ( concentration of product ) / (concentration of reactant) . As Keq is directly proportional ...
Q: Benzene is a common organic solvent that was previously used in gasoline; it is no longer used for t...
A: A hydrocarbon is a chemical compound that consists of only carbon and hydrogen in its molecules. For...
Propose an efficient synthesis for each of the following transformations:
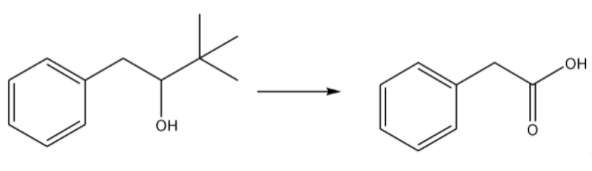
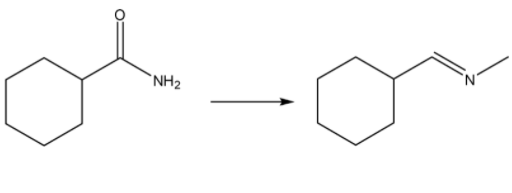
Step by step
Solved in 2 steps with 1 images




