Q: . A 0.57 gram sample aspirin (MM=180 g/mol) is crushed and dissolved in water. The sample is titrate...
A: Since you have posted multiple questions, we are entitled to answer the first only. 1) Given: Mass o...
Q: Determine the systematic name of the organic molecule in Figure A below.
A: Systematic name is also called IUPAC name.
Q: A compound has a normal boiling point of 82∘C and has a ΔHvap of 42 kJ. What is its vapor pressure a...
A:
Q: Question 28 of 28 If the density of an ideal gas at STP is found to be 0.0902 g/L, what is its mola...
A:
Q: Draw all possible resonance structures for NPO in your work. Then, assign formal charge to each atom...
A: We have to draw the all possible resonance structure yNPO and also circle the most stable resonance ...
Q: Draw the Newman projection (sighting down this bond) of the conformation that will be capable of an ...
A: E2 elimination is bimolecular elimination reaction.
Q: 5. Draw the energy diagram for the following reaction in the reaction coordinate drawn below. HCI + ...
A: In this reaction , HCl is a acid so it will readily donate H+ ion to water molecule to form H3O+ an...
Q: Write the net ionic equation for the reaction that occurs, if any, when a calcium chip is dropped in...
A:
Q: Two drops (0.1 mL) of 7.0 M HCl are added to water to make 1.0 L of solution. What is the pH of this...
A: Given: Concentration of HCl initially = 7.0 M Volume of initial HCl taken = 0.1 mL = 0.0001 L ...
Q: What is the molality of a solution prepared by dissolving 15.1 g of potassium nitrate in 250. g of w...
A:
Q: Calculate the amount. of heat required to completely evaporate 625 ml of water at 25C. the enthalpy ...
A:
Q: 1-pentanol Write "most" under the compound which can be hydrolyzed most easily. Write "least" under ...
A: Given : We have to arrange these compounds in order of hydrolysis .
Q: ng phase diagram of Substa
A:
Q: Slucose is an important sugar in a person's metabolic cycle. Identify the functional group circled. ...
A: Organic compounds with -CHO functional group are called aldehyde class of compound. Organic compoun...
Q: phase diagram
A:
Q: [References) Name the following compounds. (Include stereochemical terms where appropriate. Only use...
A: The compounds given are,
Q: Calculate the boiling point elevation and freezing point depression for a aluminum acetate solution ...
A:
Q: What is a selection rule for vibrational (IR) spectroscopy? The angular momentum must change by a v...
A: Given 1) Three of the term symbols that correspond to a nd2electron configuration are 1S, 3P, and 3...
Q: 5. Draw the major organic product of the following reaction? 1) KOI CH, 21A
A:
Q: Identify the MAJOR product that would be obtained from the following reaction. *MgBr Me Me then H20 ...
A:
Q: Which of these molecules or ions has a violation of the octet rule (other than H)? CO H20 PCI5 Br2 N...
A: Given :- molecular formula of compounds CO H2O PCl5 Br2 NH3 To identify :- species that vio...
Q: neously. After a certain amount of time, it was found that 6.71I mL of O, had passed of the unknown ...
A: Using Graham's law of diffusion, Rate of diffusion of a gas is inversely proportion of square root o...
Q: The electron configuration 1s 2s²2p 3s23p°4s23d104p 5s24d refers to the ground state of what element...
A:
Q: m largest to smallest atomic radius (that is, their physical size). Of the elements available in eac...
A:
Q: 11. For the following wavefunction p(x) = Ne4o² a) Normalize this wavefunction Answer Parts b) and c...
A:
Q: Titration is routine, useful laboratory technique that can be used to confirm the concentration of a...
A: a.) i.) As vinegar contains Acetic acid , so for titration of an acid , we need a base. ii) As most...
Q: Decide which intermolecular forces act between the molecules of each compound in the table below. in...
A: The compound given are,
Q: What is the name of TIO2 O titanium (i) oxide titanium oxide titanium (iv) oxide titanium (ii) oxide...
A: Name of the given compound--
Q: Which generalized equation represents a double replacement reaction? a- AB--->A+B b-AB+CD--->AD+...
A: Given :- Reactions : a- AB--->A+B b-AB+CD--->AD+CD c- A+B--->AB d-A+BC--->B+AC To ide...
Q: 2KCIO3 (s)→2KCI(s) + 302(g) and the following table of values: AG Substance (kJ/mol) KC103 (s) -296....
A: Given : We have to calculate the Gibbs free energy for the following reaction.
Q: A voltaic cell consists of a strip of cadmium metal in a solution of Cd(NO3)2 in one beaker, and in ...
A: Given : We have to tell which of the following will act as anode and cathode.
Q: What is the rate for the zero order reaction A → Products when [A] = 0.200 M? (k = 5.49 M/min)
A:
Q: Assuming that Coca-Cola has the same specific heat as water [4.18 J/(g C)], calculate the amount of ...
A: Given: Mass = 350g Temperature (initial) = 300C Final temperature = 90 C
Q: 1.Draw what your molecule would look like at a pH of 1 and clearly identify the overall charge
A: The name of the given molecule is atorvastatin which is used to treat cardiovascular disease. Both ...
Q: Would this reaction have more products or reactants
A: Bronsted Lowery acid-base theory : Bronsted acid is proton (H+) donar, where as Bronsted base is ...
Q: Study the following phase diagram of Substance X. A E D' F, temperature (K) А В Along which line wou...
A: The phase diagram given is,
Q: Use the reaction system below to answer the questions that follow. Predict the effect of changes in ...
A: Given that the reaction system is at equilibrium Since water is a reactant Here, stress applied : ch...
Q: On a sheet of paper 3.235 g of a compound was made to 375.0 mL solution with osmotic pressure of 4.7...
A: a. We know that : osmotic pressure π = CRT = (n/V)RT = (W/MV)RT Or, M = (WRT/πV) ...(1) Here, W =...
Q: 3. Use molecular orbital theory and the diagram below to predict which species has the strongest bon...
A: Given : F2+ , F2- , F2
Q: ism of ving eactions. а) OBr Br OtBu HOtBu b) HH H HOtBu OtBu CI CI c) H3O H20
A: Since you have posted question with multiple subparts as per guidelines we can solve only one per se...
Q: Consider this molecular-level representation of a mixture of gases. If the partial pressure of the d...
A: Mole fraction of gas= = Moles of gas /Total moles = 3/(6+5+3) = 0.214
Q: Use the phase diagram of Substance X below to find the pressure at which the boiling point of X is -...
A: The phase diagram given is,
Q: Question 25 of 28 > Three samples of an ideal gas, all at the same temperature, occupy identical con...
A: we know that for an ideal gas PV = nRT
Q: The empirical formula of a compound is C2H5O. Its molecular mass is 135.112. What is its molecular f...
A: Molecular mass = n × Empirical formula mass
Q: Identify the MAJOR product(s) that would be obtained from the following reaction. Oso, H,O2, H20 OH ...
A:
Q: Calculate the pH of solutions having the following [H+]: a) 1.69 M
A: Given : [H+] = 1.69M
Q: A gaseous mixture contains 434.0 Torr H, (g), 350.5 Torr N, (g), and 79.1 Torr Ar(g). Calculate the ...
A: the solution is as follows:
Q: Rank the following radicals in order of increasing stability.
A:
Q: In the reaction between carbon dioxide and water to produce carbonic acid, how many moles of the pro...
A:
Q: at what presure in atm does ethane (C2H6) have a density of 37.2 g/L
A: Ethane molar mass. 30.07 g/mol Density = 37.2g/L Temperature 55°C We have yo find pressure
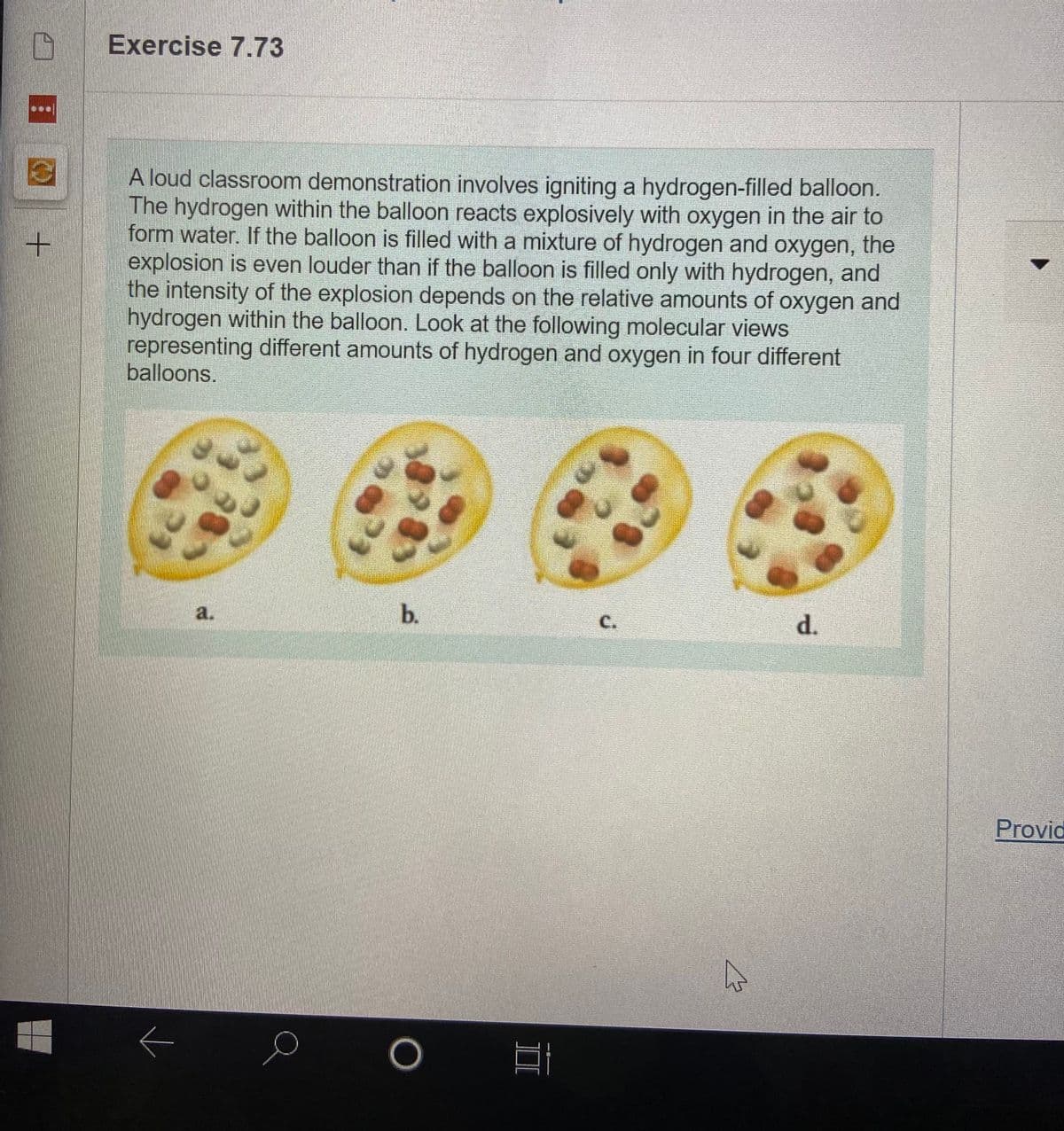
Based on the balanced chemical equation, which balloon will make the most quiet explosion?
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Butane C4H10 is burned with 16% excess air. An analysis of the combustion gases revealed that all hydrogen in the fuel burns to H20 but only 88.9% of the burns to CO2 with the remaining C forming Co. What is the volumetric fraction in % of the of CO2 in the products?using the following chemical reaction Mg+O2=MgO, 50.0 g of magnesium and 50.0 L of oxygen gas will produce how many grams of magnesium oxide? initial change endAstatine Iodide, AtI, is the heaviest known interhalogen compound. To produce AtI, astatine should reacts with Iodine, where the mole ratio between AtI to polyatomic At is 2:1. If the yield is 100%, 321 g of polyatomic At will yield 515 g of AtI.How many atoms does a single molecule of polyatomic At has? (MW, g/mol: At = 210, I = 126.9) *round off your answer to whole number
- Complete the following reaction and balance the reaction. Select all of the following that are correct coefficients for the following reaction when balanced (If no coefficient is listed, assume a value of 1) H2SO4 + NaOH ⟶ Group of answer choices H2O 2 H2O NaSO4 Na2SO4 Na(SO4)2 NaOH 2 NaOH 3 NaOH H2SO4 2 H2SO4Consider the reaction of a mixture of NaHCO3 and Ca(HCO3)2 with excess HCl. The net ionic reaction observed is H+(aq) + HCO3-(aq) → H2O(l) + CO2(g) The data for this problem is for 25 oC 7.701 g of a sample containing a mixture of NaHCO3 and Ca(HCO3)2 is mixed with an excess of HCl to produce CO2 gas. When all of the (dried) gas is collected in a 1.00 L vessel, the total pressure is 1.452 atm. How many g of NaHCO3 were present in the initial sample?3. Propane(C3H8) burns in oxygen to produce CO2 and water C3H8 + 5O2 → 3 CO2 + 4H2O (a) How many moles of oxygen are required to burn 4 moles of propane? (b)Calculate the moles of CO2 produced (c) Calculate the volume of CO2 produced by burning of 5moles propane at rtp (d) How many moles of water is produced (e) Calculate the mass in grams of water produced
- The following graph shows the concentration vs. time for the reaction of nitrogen with hydrogen to form ammonia: N2 (g) + 3H2 (g) → 2NH3 (g) Which curve is hydrogen? Question 2 figure Group of answer choices a b c None of the above is correct.Detonation of nitroglycerin proceeds as follows:4 C3H5N3O91l2¡12 CO21g2 + 6 N21g2 + O21g2 + 10 H2O1g2(a) If a sample containing 2.00 mL of nitroglycerin 1density =1.592 g>mL2 is detonated, how many moles of gas are produced?(b) If each mole of gas occupies 55 L under the conditionsof the explosion, how many liters of gas are produced?(c) How many grams of N2 are produced in the detonation?A reaction of 0.028 g of magnesium with excess hydrochloric acid generated 31.0 mL of hydrogen gas. The gas was collected by water displacement in a 22 °C water bath. The barometric pressure in the lab during the experiment was 746 mm Hg. What is the theoretical amount (mol) of hydrogen gas that should have formed from the reaction of 0.028 grams of magnesium with an excess of hydrochloric acid? ??(?) + 2???(??) → ????2(??) + ?2(?)
- 1. For a reaction in which the reactants are gases, which of the following would increase the rate of chemical reaction? (Select all Correct Answers) 2. For a reaction in which one of the reactants is a solid, what would increase the rate of reactionWhich equation below represents the reaction that is taking place between ammonia and oxygen shown in Graph 1?. Question 12 options: 2NH3 + 4O2 à 2NO + 3H2O 4NH3 + 5O2 à 4NO +6 H2O NH3 + O2 à NO + H2O 4NO + 6H2Oà 4NH3 + 5O2Calculate the number of grams in the following samples. Show all work.a. 0.20 mol of WO2b. 0.067 mol of Ru(NH3)6Cl3 How is the %P in your sample of calcium phosphate related to the %P in one mole of calcium phosphate? Would you expect that relationship to always be true? Why or why not?

