Chemistry (1= T15HW Question 5 Homework Unanswered O Determine the percent by mass of both magnesium and phosphate in the compound magnesium phosphate. When you are ready match the lon (on the left) with the correct percentage (on the right). Drag and drop options on the right-hand side and submit. For keyboard navigation... SHOW MORE 27.74% Mg²+ 56.56% PO 79,61% 72.26% Fullscreen Topic 15 Homework Homework Due in 9 hours Mg PO E 9/14 answered E 27.74% 56.56% M 79.61% E 72.26%
Chemistry (1= T15HW Question 5 Homework Unanswered O Determine the percent by mass of both magnesium and phosphate in the compound magnesium phosphate. When you are ready match the lon (on the left) with the correct percentage (on the right). Drag and drop options on the right-hand side and submit. For keyboard navigation... SHOW MORE 27.74% Mg²+ 56.56% PO 79,61% 72.26% Fullscreen Topic 15 Homework Homework Due in 9 hours Mg PO E 9/14 answered E 27.74% 56.56% M 79.61% E 72.26%
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter5: Gases
Section: Chapter Questions
Problem 25QAP: Cyclopropane mixed in the proper ratio with oxygen can be used as an anesthetic. At 755 mm Hg and...
Related questions
Question
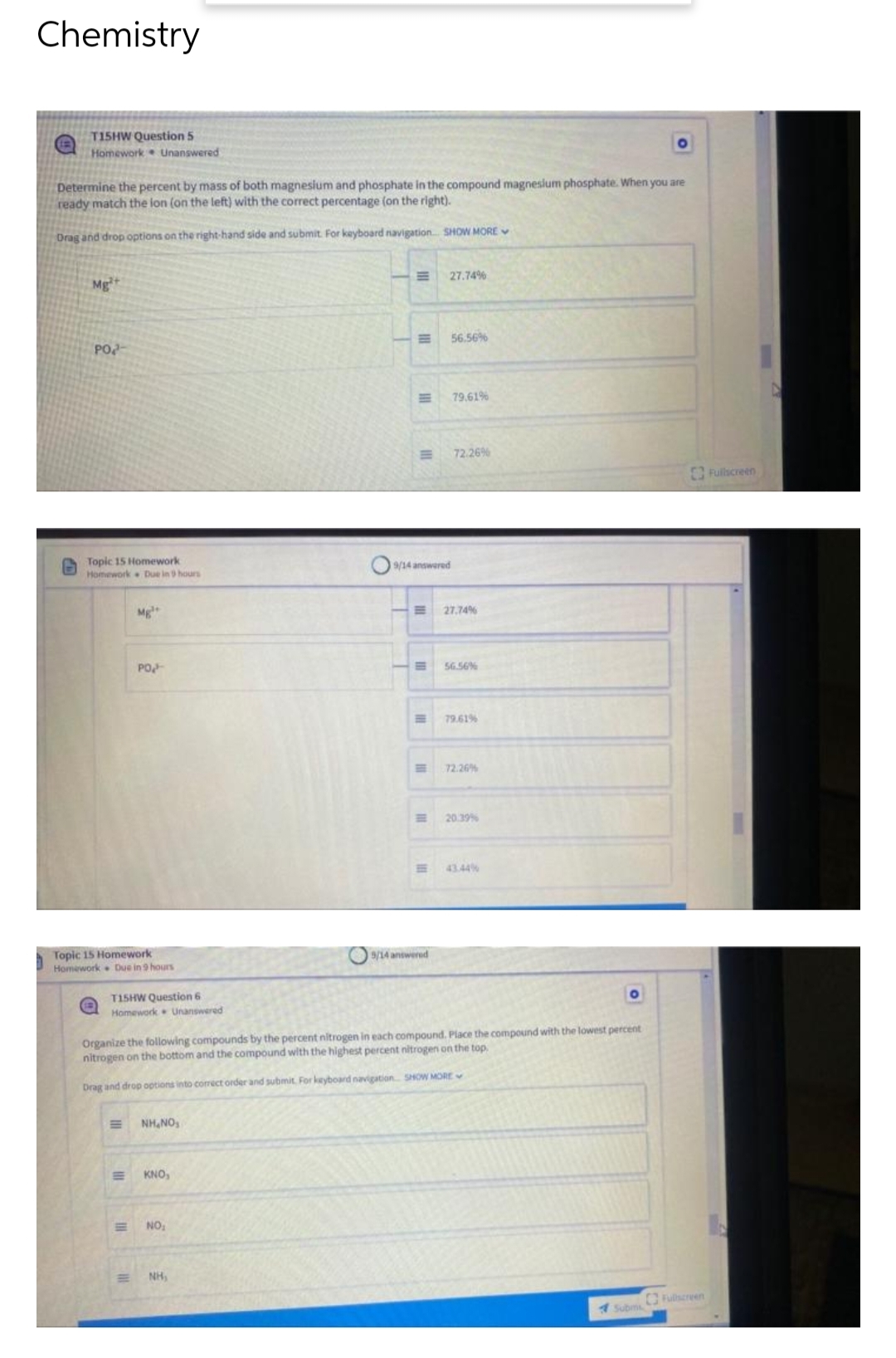
Transcribed Image Text:Chemistry
(1
T15HW Question 5
Homework Unanswered
O
Determine the percent by mass of both magnesium and phosphate in the compound magnesium phosphate. When you are
ready match the lon (on the left) with the correct percentage (on the right).
Drag and drop options on the right-hand side and submit. For keyboard navigation... SHOW MORE
27.74%
Mg²+
56.56%
PO
79.61%
72.26%
Topic 15 Homework
Homework Due in 9 hours
Mg
PO
Topic 15 Homework
Homework Due in 9 hours
E
E
NH₁
E
9/14 answered
9/14 answered
T15HW Question 6
Homework Unanswered
O
Organize the following compounds by the percent nitrogen in each compound. Place the compound with the lowest percent
nitrogen on the bottom and the compound with the highest percent nitrogen on the top.
Drag and drop options into correct order and submit. For keyboard navigation SHOW MORE
NHINGS
KNO,
NO₂
Fullscreen
Submi
27.74%
56.56%
= 79.61%
E
72.26%
20.39%
Fullscreen
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning